Understanding the management of brachial plexus birth palsy and the roles of nerve transfers
Abstract
Early recognition and management of neonatal brachial plexus injury (NBPP) is key to optimizing outcomes and surgical options. Because up to a third of patients with birth palsy may require surgical intervention, the multidisciplinary team must follow up on the function and recovery of the entire upper extremity, from shoulder to fingertips, within the first few months of life. Options include neuroma resection and primary nerve grafting and/or nerve transfers, which can be intra- or extra-plexal, as adjunctive or standalone procedures. When limited proximal nerve roots are available for nerve grafting, or when too many nerve roots are involved, exceeding the supply of nerve graft, extra-plexal nerve transfers are then indicated. While early intervention is preferred (between 3 and 6 months for most authors), older children presenting late (> 1 year since birth) with partial nerve recovery may still potentially benefit from nerve transfers, which can be offered to late-presenting infants. Here we discuss the anatomy, indications, work-up, options, complications, and outcomes for such nerve transfers for upper, lower, and global plexus palsy in neonates. Nerve transfers are a common procedure done for the treatment of various types of birth plexus palsies. Currently, while there is an abundance of clinical reports and evidence, there is still a lack of clarity regarding best practices or in terms of the type of procedure and technique for the treatment of birth plexus palsy. Our hope is that this chapter provides a concise source after an extensive cumulative review of the evidence of best practices for nerve transfers in the case of birth plexus palsy.
Keywords
INTRODUCTION
Nerve damage is common morbidity across the global population and is a particularly common complication in birth. Nerve transfers as a treatment for such disorders have seen an increase in use as well as an increase in the number of techniques in recent years. However, there is a lack of concise literature and standardization regarding best practice protocols, as proven by evidence/research. Much of the evidence seen in the literature is scattered, and while valid, the magnitude of variety in opinion on the best practices indicates a need for a single concise source serving as a review of the most up-to-date evidence on the topic. Neonatal brachial plexus injury (NBPP) or Brachial plexus birth palsy (BPBP) presents as weakness or paralysis of the upper extremity at birth. Early recognition and management in the newborn period can significantly impact outcomes. NBPP is not uncommon, occurring in approximately 1-4 children per 1,000 live births[1]. Despite advancements in obstetric care, this incidence seems to be increasing, due to increasing birth weights[2].
While many of the existing management options provide good clinical evidence, there is a great lack of clear and available information regarding long-term outcomes and the overall morbidity from complications. This is compounded by the lack of good outcome measures to examine patients and the nerve transfer patient population in the long term. Our hope is that our chapter provides more extensive options for examining long-term clinical outcomes in the case of nerve transfers.
The initial management of NBPP focuses on conservative management. Physical therapy is instituted immediately once a diagnosis is made in an attempt to recover flexion. In cases where spontaneous recovery does not occur after conservative management, surgical intervention may be warranted at 3-8 months of age to prevent further nerve damage and permeant sequelae such as strength loss, decreased size of the extremity, and range of motion deficits.
Risk factors primarily include shoulder dystocia, macrosomia, and maternal diabetes or maternal weight gain. Affected roots commonly involve C5 or C6 (upper plexus palsy), but occasionally involve C7 (extended upper plexus palsy), or even C8 and T1 (total/global palsy). Lower plexus palsy (C8 and T1) has been reported in the literature, but is usually associated with total brachial plexus injuries with avulsion of the lower roots and subsequent recovery in the upper roots [Table 1].
Types of brachial plexus injuries and associated findings[15]
| Pattern (Incidence) | Nerve roots involved | Primary deficiency |
| Erb-Duchenne lesion Upper brachial plexus (50%) | C5 and C6 | Shoulder abduction and external rotation Elbow flexion |
| Extended Erb palsy Upper & middle plexus (30%) | C5 through C7 | As above, plus elbow and finger MP joint extension |
| Dejerine-Klumpke paralysis Lower brachial plexus | C8 and T1 | Hand intrinsic muscles Finger flexors |
| Total or global lesion Entire brachial plexus (20%) | C5 through T1 | Entire extremity |
There is an abundance of literature and evidence regarding the use of nerve transfers in the setting of both plexus palsy. However, there is a lack of conciseness in terms of cumulative review of the evidence in order to provide best practice guidelines, and as such, there is a lack of standardization or protocols for nerve transfers in the setting of birth plexus palsy. Our aim is to provide a review of the most up-to-date evidence regarding nerve transfers in the setting of birth plexus palsy, hopefully allowing for a more concise view of the evidence as a whole.
INDICATIONS
Plexus injuries can be self-limiting (80%-90%) and resolve over the course of the first few weeks of life without significant long-term morbidity[3]. Physical therapy is instituted immediately after birth and continues as part of the multidisciplinary care plan. Recovering elbow flexion by two to three months is a positive prognostic sign overall, including as a sign of potential recovery at the shoulder. When spontaneous or complete recovery does not occur, surgical intervention is required to prevent or limit permanent sequelae from denervation, such as decreased strength, decreased size and girth of the extremity, range of motion limitations, joint dysplasia, joint contractures, or persistent neurologic deficits[4]. Shoulder anatomy and function can be rather challenging to reconstruct, especially shoulder abduction, for which there are no good secondary options. Abnormal muscle innervation can lead to abnormal muscle development, changes to biomechanics, and joint development, with worsening progression over time, resulting in stiffness of the glenohumeral joint, with compensatory winging of the scapula. Elbow flexion contractures are also extremely common[5].
ASSESSMENT
Based on serial physical examinations within the first few months of life, which can be challenging in a newborn, the decision is made on whether surgical intervention is indicated. In addition to general medical clearance, imaging and nerve studies can be helpful, particularly when root avulsion is suspected. In root ruptures (postganglionic), neuromas develop. In mild forms, some conduction through the neuroma may permit recovery to near-normal function; in severe forms, extensive paralysis and stiffness can develop. In root avulsions (preganglionic), no improvement is expected, and there is little value in delaying operative reconstruction.
Computed Tomography (CT) Myelography or, more likely, magnetic resonance (MR) myelography has a high (> 90%) true positive rate for diagnosing avulsion injuries[6-8]. In 2017, in a small cohort, Bauer et al. described a new technique using three-dimensional (3D) volumetric proton density MRI to evaluate nerve roots in neonates without the need for radiation, contrast agents, or sedation/anesthetic risk[9]. The sequence allowed visualization of preganglionic pseudomeningoceles with and without nerve root avulsion, postganglionic neuromas, and nerve root thickening, aiming to grade the severity of injury earlier than serial clinical examinations. Ultrasound alone, even high-resolution sonography, is not reliably comprehensive enough to be used alone, but can be used as an adjunct to MRI[10]. Electrodiagnostic studies (EDS) can be used when there is doubt surrounding avulsion injuries, but because they can sometimes be misleading, EDS are not used for prognosis[11]. Diaphragmatic ultrasound examination should also be performed to assess whether cervical nerve injury has affected diaphragmatic innervation and could impact safety for surgery.
OPTIONS FOR SURGICAL INTERVENTION
Up to a third of patients with birth palsy may require surgical intervention[12]. The approach to surgical repair varies depending on the surgeon and the nature of the injury. The timing of surgery is controversial but is typically done between 3 to 8 months of age. Options include neuroma resection and primary nerve grafting and/or nerve transfers, which can be intra- or extra-plexal, as adjunctive or standalone procedures[13].
When limited proximal nerve roots are available for nerve grafting, or when too many nerve roots are involved, exceeding the supply of nerve graft, extra-plexal nerve transfers are then indicated[14,15] nerve transfer in BPBP follows all the traditional principles- selection depends on cataloging what nerves are deficient, what nerves are available/expendable, their anatomic proximity, and agonist function (caliber, motor vs. sensory function)[16-18]. Compared to excision and grafting alone, by performing transfers as close as possible to the motor endplates, nerve transfers bring more specific axonal inflow (pure motor donors), and offer quicker reinnervation, allowing for faster recovery. While early intervention is preferred (between 3 and 6 months for most authors), older children presenting late (> 1 year since birth) with partial nerve recovery may still potentially benefit from nerve transfers, which can be offered to late-presenting infants[15,19].
UPPER PLEXUS PALSY (C5-6) AND EXTENDED UPPER PLEXUS PALSY (C5-7)
Upper plexus palsies most likely occur as ruptures. When occurring as sequelae of breech presentations, there should be a high index of suspicion for nerve avulsion and the need for earlier intervention. In managing upper plexus palsy, establishing the stability of the shoulder is of paramount importance[20].
The shoulder is a complex joint and has the most range of motion of any joint in the body. It is believed to have played an important role in evolution, allowing for vertical climbing and, with the ability to carry things, creating an incentive for bipedal locomotion. In order to position the hand in a wide range of space, the shoulder requires stability and mobility, conferred by the glenohumeral joint, the scapulothoracic-gliding plane, and the muscles that surround it. The rotator cuff muscles and deltoid are innervated by branches of C5-6: the suprascapular nerve (supraspinatus, infraspinatus), upper and lower subscapular nerves (subscapularis), and axillary nerve (teres minor, deltoid)[21]. The supraspinatus and deltoid are abductors, the infraspinatus and teres minor external rotators, and the subscapularis is an internal rotator.
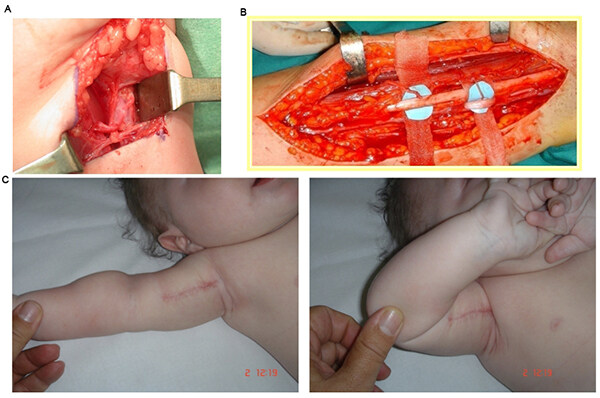
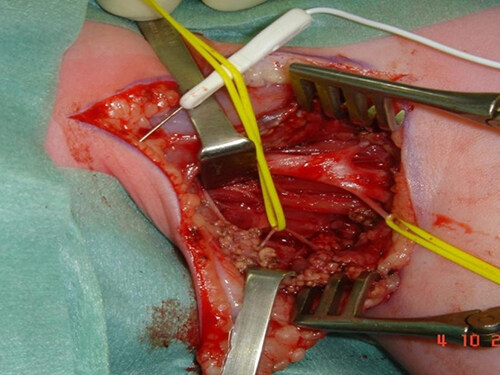
As such, the targets for nerve transfer are the suprascapular nerve and/or the axillary nerve. Kozin has previously discussed his indications, with a preference to target both nerves for flail shoulders and isolated nerve transfer to the suprascapular nerve when adequate deltoid function exists without external rotation[22,23]. The most common options to reconstruct these nerves are the spinal accessory to suprascapular nerve transfer and the radial to axillary nerve transfer. Specifically, when addressing the suprascapular nerve, some have recommended decompressing the suprascapular nerve at the suprascapular notch to obviate the double crush phenomenon[24,25]. As for the axillary nerve, the long or lateral head of the triceps branch of the radial nerve is transferred to the anterior division of the axillary nerve (Somsak transfer, Figure 1)[26].
Figure 1. Somsak transfer: Radial nerve to axillary nerve transfer. (Photos courtesy of Dr. Gharboui, senior author).
Previous studies comparing grafting vs. transfer for shoulder reconstruction showed no significant difference, but patients were not always matched based on injury severity[27]. In a 2020 multi-center, retrospective cohort study, Manske et al. matched Narakas types [Table 2], preoperative Active Movement Scale (AMS) scores, and shoulder subluxation to compare outcomes after grafting vs. transfer[28-30]. They found regardless of surgical technique, shoulder external rotation recovery was disappointing, but more children undergoing spinal accessory nerve (SAN) to suprascapular nerve (SSN) transfers achieved functional shoulder external rotation (AMS > 5) and fewer secondary shoulder procedures, such as tendon transfers or osteotomies. They concluded SAN to SSN transfer should be performed in all infants undergoing brachial plexus reconstruction. These results were corroborated in a 2021 meta-analysis[31]. Likewise, Segal et al. noted regardless of starting point, SAN-SSN transfer improved shoulder function[32].
Narakas Classification of Obstetric Palsy[29]
| Group | Name | Roots injured | Site of weakness/paralysis | Likely outcome |
| 1 | Upper Erb’s | C5, C6 | Shoulder abduction/external rotation, elbow flexion | Good spontaneous recovery in over 80% of cases |
| 2 | Extended Erb’s | C5, C6, C7 | As above, with drop wrist | Good spontaneous recovery in over 60% of cases |
| 3 | Total palsy with no Horner syndrome | C5, C6, C7, C8, T1 | Complete flaccid paralysis | Good spontaneous recovery for shoulder and elbow in |
| 4 | Total palsy with Horner syndrome | C5, C6, C7, C8, T1 | Complete flaccid paralysis with Horner syndrome | The worst outcome. Without surgery, severe defects throughout the limb are expected |
Subsequent to shoulder stability, elbow motion is critical to upper extremity function and warrants high-priority consideration during nerve reconstruction[16]. The elbow is involved in forearm flexion, extension, supination, and pronation. Not only is the basic function of “hand-to-mouth” essential (“cookie test”), the absence of anti-gravity elbow flexion at key milestones (3, 5-6, or 9 months) is a primary indicator of poor functional recovery and the need for surgical intervention[33]. For elbow flexion, the targets for reconstruction are the brachialis muscle (the primary elbow flexor) and the biceps muscle (the primary forearm supinator and a secondary elbow flexor). The musculocutaneous nerve (C5-7) innervates both muscles, and the brachialis receives an additional contribution from the radial nerve (C5-6).
Donor nerve selection is dependent upon the extent of the palsy: For infants with upper (C5-6)/middle trunk (C7) deficits and an intact lower trunk (C8-T1), the surgeon can use local ulnar and/or median nerve donors to the musculocutaneous nerve.
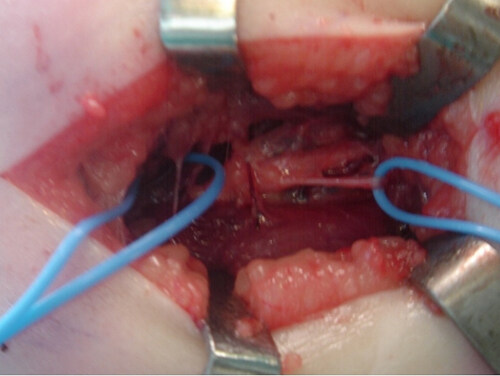
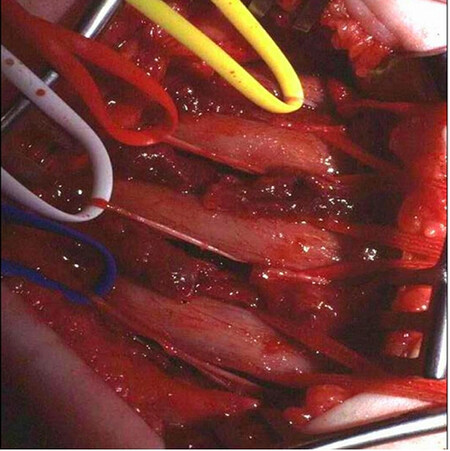
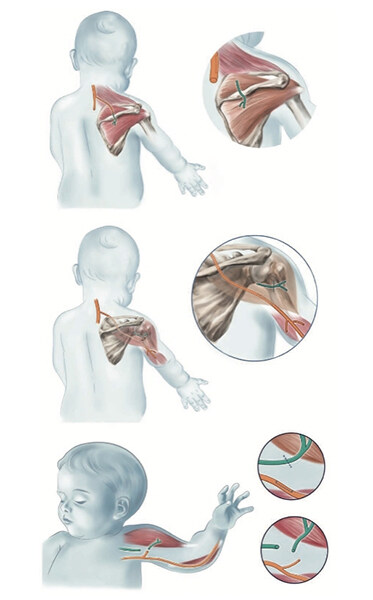
Specifically, the Oberlin procedure is one of the most commonly performed transfers for recovery of elbow flexion. The ulnar nerve fascicles to the Flexor Carpi Ulnaris (FCU) can be transferred to the biceps branch (Oberlin transfer, Figure 2), and the median nerve fascicles to the Flexor Carpi Radialis (FCR) can be transferred to the brachialis branch[34,35]. Other commonly used options include the intercostal nerves [Figure 3], thoracodorsal nerve, medial pectoral nerve, and long thoracic nerve. The phrenic nerve, routinely used in adults, is rarely used in obstetric cases. Finally, ipsilateral and contralateral C7 roots allow the addition of a large number of axons without deficits for the donor site[36]. A review of the literature finds small sample sizes, varying outcomes, and inconsistent comparison to traditional nerve grafting[37]. The consensus is that the procedure is safe and, sometimes, effective in restoring elbow flexion[38-41]. Compared to traditional nerve grafting alone, O’Grady et al. have reported triple nerve transfer with nerve grafting to have better functional shoulder external rotation and forearm supination, faster recovery, and lower cost[42] [Figure 4].
Figure 2. Oberlin Procedure for reconstruction of musculocutaneous nerve for elbow flexion: (A) Ulnar nerve fascicle to FCU transfer to brachialis branch. (B) Double Oberlin procedure, with ulnar nerve to brachialis, and median nerve fascicles to FCR transferred to biceps branch. (C) Elbow flexion after Oberlin procedure. (Photos courtesy of Dr. Gharboui, senior author).
Figure 3. Intercostal nerves identified as donors in preparation for nerve transfer. (Photos courtesy of Dr. Gharboui, senior author).
Figure 4. Distal nerve transfers. (Top) Posterior approach transferring a distal branch of the spinal accessory nerve to the suprascapular nerve and also releasing the scapular ligament. (Center) Transferring a motor branch of the radial nerve to the lateral triceps to reinnervate the axillary nerve close to its neuromuscular junction in the deltoid muscle. (Below) Using a motor branch of the ulnar nerve to the flexor carpi ulnaris muscle to reinnervate the biceps muscle. Shared with permission by authors[41].
TOTAL/GLOBAL PLEXUS PALSY
In global plexus palsy, not only is the shoulder and elbow compromised, but also the hand, to varying degrees (complete vs. near complete). Global plexus palsies often occur as a rupture of the upper roots and avulsion of the lower plexus. As such, with an avulsive component, surgery is to be expected within the first few months of infancy[43]. If possible, intra-plexal transfers are performed, with the C5 root as a donor to the two lower avulsed roots, the C6 root as a donor to the lateral and posterior cords, and, if C7 is available, motor fibers can be used to neurotics C8 and T1 roots via end-to-end repair. However, in true global palsy, intra-plexal transfers are not possible.
In the unfortunate case of complete palsy, due to the likelihood of pre-ganglionic avulsions, in addition to attempting nerve grafts, extra-plexal nerve transfers are needed to restore shoulder stability and elbow flexion. Common donors in this setting include the spinal accessory nerve (transfer to suprascapular nerve, Figure 5), and intercostal nerves (3rd to 6th, transfer to the musculocutaneous, radial nerve). The unaffected, contralateral C7 root can also be used (partial/posterior division or complete)[44-47]. The hypoglossal nerve has been used in the past, with poor functional results[48,49]. Of note, in a 2020 series of qualitative interviews, Lanier et al. noted that most surgeons in North America avoided extraplexal donors other than the intercostal nerve (ICN) or SAN due to concerns for safety and morbidity[50].
Figure 5. Spinal accessory nerve to suprascapular nerve transfer (SAN to SSN). (Photos courtesy of Dr. Gharboui, senior author).
Finally, though they have been described, sensory nerve transfers are generally not part of the primary brachial plexus reconstruction.
Complications
In a 2021 systematic review of nerve transfers for brachial plexus birth injuries, Hinchcliff et al. reported complications for various nerve transfers[51]. Overall, complications were relatively uncommon and transient. Transfers most commonly associated with adverse events included intercostal nerve transfers and hypoglossal nerve transfers.
For intercostal nerve transfer to various targets, there were reports of basilar atelectasis (9 out of 9) complicated by pneumonia (2 out of 9), and mild growth retardation of the donor site ribs (in patients who underwent surgery at < 5 months of age), without changes in chest wall sensation, thoracic cage deformity, or scoliosis.
For ulnar/median nerve to biceps/brachialis transfers, complications were minimal, with one report of transient anterior interosseous nerve (AIN) palsy and no loss of function after nerve transfer. There were no motor deficits, functional deficits, sensory deficits, or growth deformities.
For the largely obsolete hypoglossal nerve (partial or complete) to various targets, atrophic change in the tongue occurred in 0%-100% of patients, but only 16.6%-21.4% with serious atrophy. All patients had early feeding difficulties, with 50% requiring speech therapy intervention. Phonologic speech development was affected, including delay or disturbance, and associated movements of the ipsilateral arm with tongue movement were observed.
Scapular winging was seen in 37.5% of patients who underwent spinal accessory nerve (SAN) to various targets and for the dorsal scapular nerve to suprascapular nerve (SSN) transfer. For contralateral C7 to various targets, complications included synchronous movements and sensations (44.7%-55%), transient
Outcomes assessment
Assessing outcomes after nerve transfer for birth palsy is difficult- injury pattern and severity are varied, reports are limited, and physical exam (motor and sensory) in infants is challenging. Outcome reports are subject to the surgeon’s reporting bias (conscious or unconscious), variability of follow-up duration, and quality of the study (small sample size, retrospective studies), and there is a lack of consistency in how clinicians assess, treat, and report functional outcomes, making direct comparisons troublesome[52]. The Toronto Test Score, Hospital for Sick Children Active Movement Scale (AMS), the Mallet score, and the Medical Research Council grading system are accepted tools for assessment[14,30,51-53]. Outcomes assessments must also include quality of life measures, impact on the family, and psychosocial factors[51,52].
CONCLUSION
Neonatal birth plexus palsy is a varied and complicated diagnosis requiring multidisciplinary care. From physical examination and treatment to psychosocial issues and family impact on the patient and caregivers, NBPP remains a complex and challenging problem. In addition to primary nerve grafting, nerve transfers should be considered essential tools in the surgical reconstruction of NBPP. Nerve transfers allow for faster, more targeted recovery, and can be used in older children, and more research is needed to better characterize, understand, and compare outcomes.
DECLARATIONS
Authors’ contributionsMade substantial contributions to the conception and design of the article and preparation: Chen W, Gharbaoui I, Keyho R
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestWendy Chen- paid consultant for Allergan LIMITLESS Women’s Surgeons initiative. All authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2023.
REFERENCES
1. McLaren RA Jr, Chang KW, Ankumah NE, Yang LJ, Chauhan SP. Persistence of neonatal brachial plexus palsy among nulliparous versus parous women. AJP Rep 2019;9:1-5.
2. Jobe MT. Peripheral nerve injuries. In: Cnalae ST, Beaty JH, editors. Campbells’s operative orthopedics, ed 12. Philadelphia: Mosby; 2013. p.3062-123.
3. Gordon M, Rich H, Deutschberger J, Green M. The immediate and long-term outcome of obstetric birth trauma. I. Brachial plexus paralysis. Am J Obstet Gynecol 1973;117:51-6.
4. Hems T. Nerve transfers for traumatic brachial plexus injury: advantages and problems. J Hand Microsurg 2011;3:6-10.
5. Russo SA, Kozin SH, Zlotolow DA, et al. Scapulothoracic and glenohumeral contributions to motion in children with brachial plexus birth palsy. J Shoulder Elbow Surg 2014;23:327-38.
6. Chow BC, Blaser S, Clarke HM. Predictive value of computed tomographic myelography in obstetrical brachial plexus palsy. Plast Reconstr Surg 2000;106:971-7; discussion 978.
7. Steens SC, Pondaag W, Malessy MJ, Verbist BM. Obstetric brachial plexus lesions: CT myelography. Radiology 2011;259:508-15.
8. Tse R, Nixon JN, Iyer RS, Kuhlman-Wood KA, Ishak GE. The diagnostic value of CT myelography, MR myelography, and both in neonatal brachial plexus palsy. AJNR Am J Neuroradiol 2014;35:1425-32.
9. Bauer AS, Shen PY, Nidecker AE, Lee PS, James MA. Neonatal magnetic resonance imaging without sedation correlates with injury severity in brachial plexus birth palsy. J Hand Surg Am 2017;42:335-43.
10. Gunes A, Bulut E, Uzumcugil A, Oguz KK. Brachial plexus ultrasound and mri in children with brachial plexus birth injury. AJNR Am J Neuroradiol 2018;39:1745-50.
11. Kawai H, Tsuyuguchi Y, Masada K, et al. Identification of the lesion in brachial plexus injuries with root avulsion: a comprehensive assessment by means of preoperative findings, myelography, surgical exploration and intraoperative electrodiagnosis. Neuro-orthopedics 1989;15-23. Available from: https://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=19275066.
12. Squitieri L, Steggerda J, Yang LJ, Kim HM, Chung KC. A national study to evaluate trends in the utilization of nerve reconstruction for treatment of neonatal brachial plexus palsy [outcomes article]. Plast Reconstr Surg 2011;127:277-83.
13. Bjorklund KA, West JM, Nopkhun W, Moore AM. Surgical innovations to restore function in pediatric peripheral nerve conditions. Pediatrics 2021;148:e2021052556.
14. Davidge KM, Clarke HM, Borschel GH. Nerve transfers in birth related brachial plexus injuries: where do we stand? Hand Clin 2016;32:175-90.
15. Tse R, Kozin SH, Malessy MJ, Clarke HM. International Federation of Societies for Surgery of the Hand Committee report: the role of nerve transfers in the treatment of neonatal brachial plexus palsy. J Hand Surg Am 2015;40:1246-59.
16. Kozin SH. Nerve transfers in brachial plexus birth palsies: indications, techniques, and outcomes. Hand Clin 2008;24:363-76, v.
17. Hinchcliff KM, Pulos N, Shin AY, Stutz C. Morbidity of nerve transfers for brachial plexus birth injury: a systematic review. J Pediatr Orthop 2021;41:e188-98.
18. Pondaag W, Malessy MJ, van Dijk JG, Thomeer RT. Natural history of obstetric brachial plexus palsy: a systematic review. Dev Med Child Neurol 2004;46:138-44.
19. Smith BW, Chang KWC, Koduri S, Yang LJS. Nerve graft versus nerve transfer for neonatal brachial plexus: shoulder outcomes. J Neurosurg Pediatr 2020;27:87-92.
20. Terzis JK, Papakonstantinou KC. Management of obstetric brachial plexus palsy. Hand Clin 1999;15:717-36.
21. Veeger HE, van der Helm FC. Shoulder function: the perfect compromise between mobility and stability. J Biomech 2007;40:2119-29.
22. Kozin SH. Correlation between external rotation of the glenohumeral joint and deformity after brachial plexus birth palsy. J Pediatr Orthop 2004;24:189-93.
23. Clarke SE, Kozin SH, Chafetz RS. The biceps tendon as a measure of rotational deformity in residual brachial plexus birth palsy. J Pediatr Orthop 2009;29:490-5.
24. Tse R, Marcus JR, Curtis CG, Dupuis A, Clarke HM. Suprascapular nerve reconstruction in obstetrical brachial plexus palsy: spinal accessory nerve transfer versus C5 root grafting. Plast Reconstr Surg 2011;127:2391-6.
25. Seruya M, Shen SH, Fuzzard S, Coombs CJ, McCombe DB, Johnstone BR. Spinal accessory nerve transfer outperforms cervical root grafting for suprascapular nerve reconstruction in neonatal brachial plexus palsy. Plast Reconstr Surg 2015;135:1431-8.
26. Witoonchart K, Leechavengvongs S, Uerpairojkit C, Thuvasethakul P, Wongnopsuwan V. Nerve transfer to deltoid muscle using the nerve to the long head of the triceps, part I: an anatomic feasibility study. J Hand Surg Am 2003;28:628-32.
27. Pondaag W, de Boer R, van Wijlen-Hempel MS, Hofstede-Buitenhuis SM, Malessy MJ. External rotation as a result of suprascapular nerve neurotization in obstetric brachial plexus lesions. Neurosurgery 2005;57:530-7; discussion 530.
28. Manske MC, Kalish LA, Cornwall R, Peljovich AE, Bauer AS. Reconstruction of the suprascapular nerve in brachial plexus birth injury: a comparison of nerve grafting and nerve transfers. J Bone Joint Surg Am 2020;102:298-308.
29. Al-Qattan MM, El-Sayed AA, Al-Zahrani AY, et al. Narakas classification of obstetric brachial plexus palsy revisited. J Hand Surg Eur Vol 2009;34:788-91.
30. Curtis C, Stephens D, Clarke HM, Andrews D. The active movement scale: an evaluative tool for infants with obstetrical brachial plexus palsy. J Hand Surg Am 2002;27:470-8.
31. Nickel KJ, Morzycki A, Hsiao R, Morhart MJ, Olson JL. Nerve transfer is superior to nerve grafting for suprascapular nerve reconstruction in obstetrical brachial plexus birth injury: a meta-analysis. Hand (N Y) 2021:Epub ahead of print.
32. Segal D, Cornwall R, Little KJ. Outcomes of spinal accessory-to-suprascapular nerve transfers for brachial plexus birth injury. J Hand Surg Am 2019;44:578-87.
33. Waters PM. Comparison of the natural history, the outcome of microsurgical repair, and the outcome of operative reconstruction in brachial plexus birth palsy. J Bone Joint Surg Am 1999;81:649-59.
34. Bertelli JA, Ghizoni MF. Reconstruction of C5 and C6 brachial plexus avulsion injury by multiple nerve transfers: spinal accessory to suprascapular, ulnar fascicles to biceps branch, and triceps long or lateral head branch to axillary nerve. J Hand Surg Am 2004;29:131-9.
35. Tung TH, Novak CB, Mackinnon SE. Nerve transfers to the biceps and brachialis branches to improve elbow flexion strength after brachial plexus injuries. J Neurosurg 2003;98:313-8.
36. Blaauw G, Slooff AC. Transfer of pectoral nerves to the musculocutaneous nerve in obstetric upper brachial plexus palsy. Neurosurgery 2003;53:338-41; discussion 341.
37. Ladak A, Morhart M, O'Grady K, et al. Distal nerve transfers are effective in treating patients with upper trunk obstetrical brachial plexus injuries: an early experience. Plast Reconstr Surg 2013;132:985e-92e.
38. Rde M, Grechi G, Gepp Rde A. Oberlin’s procedure in children with obstetric brachial plexus palsy. Childs Nerv Syst 2016;32:1085-91.
39. Siqueira MG, Socolovsky M, Heise CO, Martins RS, Di Masi G. Efficacy and safety of Oberlin's procedure in the treatment of brachial plexus birth palsy. Neurosurgery 2012;71:1156-60; discussion 1161.
40. Al-Qattan MM. Oberlin’s ulnar nerve transfer to the biceps nerve in Erb’s birth palsy. Plast Reconstr Surg 2002;109:405-7.
41. Shigematsu K, Yajima H, Kobata Y, Kawamura K, Maegawa N, Takakura Y. Oberlin partial ulnar nerve transfer for restoration in obstetric brachial plexus palsy of a newborn: case report. J Brachial Plex Peripher Nerve Inj 2006;1:3.
42. O'Grady KM, Power HA, Olson JL, et al. Comparing the efficacy of triple nerve transfers with nerve graft reconstruction in upper trunk obstetric brachial plexus injury. Plast Reconstr Surg 2017;140:747-56.
43. Terzis JK, Papakonstantinou K. Surgical treatment of obstetrical brachial plexus paralysis: the norfolk experience. Semin Plast Surg 2004;18:359-75.
44. Gu YD. Contralateral C7 root transfer over the last 20 years in China. Chin Med J (Engl) 2007;120:1123-26.
45. Lin H, Hou C, Chen D. Modified C7 neurotization for the treatment of obstetrical brachial plexus palsy. Muscle Nerve 2010;42:764-8.
46. Lin H, Hou C, Chen D. Contralateral C7 transfer for the treatment of upper obstetrical brachial plexus palsy. Pediatr Surg Int 2011;27:997-1001.
47. Chen L, Gu YD, Hu SN, Xu JG, Xu L, Fu Y. Contralateral C7 transfer for the treatment of brachial plexus root avulsions in children - a report of 12 cases. J Hand Surg Am 2007;32:96-103.
48. Malessy MJ, Hoffmann CF, Thomeer RT. Initial report on the limited value of hypoglossal nerve transfer to treat brachial plexus root avulsions. J Neurosurg 1999;91:601-4.
49. Blaauw G, Sauter Y, Lacroix CL, Slooff AC. Hypoglossal nerve transfer in obstetric brachial plexus palsy. J Plast Reconstr Aesthet Surg 2006;59:474-8.
50. Lanier ST, Hill JR, James AS, Rolf L, Brogan DM, Dy CJ. Approach to the pan-brachial Plexus injury: variation in surgical strategies among surgeons. Plast Reconstr Surg Glob Open 2020;8:e3267.
51. Mallet J. Paralysie obstétricale du plexus brachial. 3. Conclusions [Obstetrical paralysis of the brachial plexus. 3. Conclusions]. Rev Chir Orthop Reparatrice Appar Mot 1972;58:201-4.
52. Mallet J. Obstetrical paralysis of the brachial plexus. II. Therapeutics. Treatment of sequelae. Results of different therapeutic technics and indications. Rev Chir Orthop Reparatrice Appar Mot 1972;58:192-6.
53. Compston A. Aids to the investigation of peripheral nerve injuries. Medical Research Council: Nerve Injuries Research Committee. His Majesty’s Stationery Office: 1942; pp. 48 (iii) and 74 figures and 7 diagrams; with aids to the examination of the peripheral nervous system. By Michael O'Brien for the Guarantors of Brain. Saunders Elsevier: 2010; pp. [8] 64 and 94 Figures. Brain 2010;133:2838-44.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Chen W, Keyho R, Gharboui I. Understanding the management of brachial plexus birth palsy and the roles of nerve transfers. Plast Aesthet Res 2023;10:19. http://dx.doi.org/10.20517/2347-9264.2022.46
AMA Style
Chen W, Keyho R, Gharboui I. Understanding the management of brachial plexus birth palsy and the roles of nerve transfers. Plastic and Aesthetic Research. 2023; 10: 19. http://dx.doi.org/10.20517/2347-9264.2022.46
Chicago/Turabian Style
Chen, Wendy, Ryan Keyho, Idris Gharboui. 2023. "Understanding the management of brachial plexus birth palsy and the roles of nerve transfers" Plastic and Aesthetic Research. 10: 19. http://dx.doi.org/10.20517/2347-9264.2022.46
ACS Style
Chen, W.; Keyho R.; Gharboui I. Understanding the management of brachial plexus birth palsy and the roles of nerve transfers. Plast. Aesthet. Res. 2023, 10, 19. http://dx.doi.org/10.20517/2347-9264.2022.46
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 9 clicks
Cite This Article 9 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.