Effect of neoadjuvant chemotherapy on skin-sparing mastectomy and breast reconstruction modalities in 409 patients
Abstract
Aim: While skin-sparing mastectomy (SSM) can be performed in patients with stage II-III breast cancer, the impact of neoadjuvant chemotherapy (NAC) on SSM rates and reconstructive modalities in these patients is not known.
Methods: Between January 2007 and December 2009, 409 immediate breast reconstructions (IBRs) were performed in patients with Stage II-III breast cancer. Data were collected on preoperative, operative, and postoperative factors.
Results: There was a statistically significant relationship between clinical stage of disease and the utilization of SSM or non-SSM (P < 0.0001). Seventy-five percent of all patients with stage II disease and 50% of patients with stage III disease underwent SSM; similarly, 75.5% of patients with stage II and 49.1% of patients with stage III disease who received NAC underwent SSM with immediate reconstruction, in spite of having a greater proportion of stage III patients (P < 0.01). In addition, patients who received NAC followed by SSM with IBR had larger tumors (mean, 3.5 cm vs. 3.1 cm, P < 0.001). The type of IBR, and size of skin defect was significantly affected by whether the patient underwent SSM or non-SSM (P = 0.001, P < 0.01, respectively).
Conclusion: We are increasingly considering NAC to be an important tool to potentially reduce the morbidity of mastectomy, including the need to resect breast skin, which can subsequently enhance reconstructive outcomes in patients with clinical stage II and III breast cancer. Specifically, our data suggest that NAC patients with stage II and III breast cancer and larger tumors can reliably and safely undergo SSM in nearly half of cases, thus improving reconstructive outcomes and patient well-being.
Keywords
Introduction
The increased use of neoadjuvant chemotherapy (NAC) has altered the therapeutic management of patients with clinical stage II and III breast cancer. For patients with large breast tumors, NAC has been shown to significantly reduce tumor size in > 90% of cases, thus increasing the proportion of patients eligible for breast conservation surgery (BCS).[1] Conversely, in women undergoing mastectomy for early-stage breast cancer, skin-sparing mastectomy (SSM) followed by immediate breast reconstruction (IBR) has been shown to result in acceptable oncologic and esthetic outcomes and good patient satisfaction.[2-5] There is increasing evidence that NAC followed by SSM and postmastectomy radiation therapy results in favorable long-term local control and survival rates.[6,7] It is generally accepted that NAC does not increase complication rates after SSM and IBR but that NAC patients undergo IBR and delayed breast reconstruction with decreased frequency.[8,9] However, it is unclear whether the use of NAC increases the likelihood that patients with stage II and III breast cancer will receive SSM with immediate reconstruction, or if it changes the use of reconstructive modality.
Given the clear preference for skin-preserving mastectomy with IBR in the majority of patients undergoing mastectomy, the interaction of these therapeutic options and their impact on outcomes needs to be elucidated.[10-13] In light of the advantages of NAC on improving breast conservation rates, if applied to patients undergoing mastectomy, it could both allow for more skin preservation and improve the reconstructive options that can be offered to these patients.[14,15] For patients with clinical stage II-III breast cancer who would otherwise not be candidates for SSM, conversion from non-SSM to SSM allows reconstructive surgeons to optimize outcomes due to the preservation of the three-dimensional skin envelope, the key component of an aesthetically acceptable breast reconstruction.
The purpose of this study was to compare the clinical characteristics and outcomes of patients with large primary and locally advanced breast cancer (stages II and III) with or without NAC and IBR after mastectomy. Objectives of this study were to determine the impact of NAC and other clinical factors on the rate of SSM and the choice of the reconstructive modality in these patients.
Methods
We searched the plastic surgery, breast surgical oncology and breast medical oncology databases for patients with stage II-III breast cancer who underwent IBR. We excluded patients whose records lacked information about the type of primary surgery or whether the patient had received NAC. All patients were treated at the same tertiary referral center. American Joint Committee on Cancer clinical disease stage, patient demographic information, and the side of the affected breast were recorded for all patients. Data were collected from clinic notes, patient charts, operative reports, and prospectively entered plastic surgery, medical oncology, and breast surgical oncology databases.
For statistical analyzes, patients who underwent IBR were separated into two groups: patients who underwent SSM and patients who underwent non-SSM. Clinical and pathological data were tabulated for each of these groups. For comparison of all categorical variables, Chi-square analysis or Fisher’s exact test (when sample sizes were small) was used. For continuous variables, Student’s t-test or the rank sum test (when variances from comparison groups were not equal) was used. All P values were two-tailed, and we considered P ≤ 0.05 to be significant. Stata statistical software (StataSE 10, StataCorp LP, College Station, TX) was used for all statistical analyzes.
Results
We identified 409 patients with stage II-III breast cancer who met study criteria for inclusion. Table 1 shows the clinical characteristics of patients who underwent SSM vs. those who underwent non-SSM. There was a statistically significant relationship between clinical stage of disease and the utilization of SSM or non-SSM (P < 0.0001). Seventy-five percent of patients with stage II disease and 50% of patients with stage III disease underwent SSM. Tumor size also had a significant impact on the utilization of SSM (P = 0.017): patients who underwent SSM had a mean tumor size of 3.1 cm (range, 0.5-14 cm) vs. a mean tumor size of 3.9 cm (range, 0.8-20 cm) for patients who underwent non-SSM. The authors found a significant difference in the size of excised skin in non-SSM vs. SSM patients (56.2 cm2 vs. 22.3 cm2, P < 0.01). As a consequence of the need to replace breast skin, the type of IBR was significantly affected by whether the patient underwent SSM or non-SSM (P = 0.001). Fifty-one-point-four percent of SSM patients ultimately had implant-based reconstruction, 41.4% had autologous reconstruction, and only 7.2% had a latissimus dorsi flap plus a breast implant, vs. 36.8%, 44.4%, and 18.8% for non-SSM patients, respectively.
Comparisons of clinical characteristics between patients who underwent SSM and patients who did not (n = 409)
| Characteristics | Non-SSM (n = 117) | SSM (n = 292) | P |
|---|---|---|---|
| Age | |||
| Mean | 48.1 | 47.3 | 0.4 |
| Median (range) | 47 (29-76) | 48 (23-75) | |
| Race | |||
| White | 89 (29.1) | 217 (70.9) | 0.7 |
| Other | 28 (27.2) | 75 (72.8) | |
| Clinical TNM stage | |||
| Stage II | 87 (24.9) | 262 (75.1) | < 0.0001 |
| Stage III | 30 (50.0) | 30 (50.0) | |
| Tumor size (cm) | |||
| Mean | 3.9 | 3.1 | 0.017* |
| Median (range) | 3 (0.8-20) | 2.9 (0.5-14) | |
| Neoadjuvant chemotherapy | |||
| No | 43 (25.9) | 123 (74.1) | 0.3 |
| Yes | 74 (30.4) | 169 (69.6) | |
| Year of surgery | |||
| 2007 | 20 (32.3) | 42 (67.7) | 0.7 |
| 2008 | 35 (29.9) | 82 (70.1) | |
| 2009 | 62 (27.0) | 168 (73.0) | |
| Reconstruction type | |||
| Tissue expander followed by implant | 43 (36.8) | 150 (51.4) | 0.001 |
| Autologous | 52 (44.4) | 121 (41.4) | |
| Latissimus dorsi flap | 22 (18.8) | 21 (7.2) | |
Despite the findings that 57.8% of the SSM patients received NAC and 69.5% of the patients who had NAC underwent SSM, NAC was not shown to have a significant impact on whether a patient underwent SSM or non-SSM (P = 0.3). Table 2 compares the clinical characteristics of SSM and non-SSM patients who underwent NAC. Similar to the listing in Table 1 (which includes all study participants), 75.5% of patients with stage II and 49.1% of patients with stage III disease who received NAC underwent SSM with immediate reconstruction (P < 0.0001). Among the patients who received NAC, the mean clinical tumor size for the patients who underwent SSM was 3.5 cm (range, 0.5-14 cm) compared with 4.6 cm (range, 0.8-20 cm) for those who underwent non-SSM (P = 0.025). Of the patients who received NAC followed by SSM, 54.4% had implant-based reconstruction, 39.6% had autologous tissue flap reconstruction only, and 5.9% had a latissimus dorsi myocutaneous flap plus a breast implant, vs. 27%, 46%, and 27% for non-SSM patients, respectively.
Comparisons of clinical characteristics of patients who received neoadjuvant chemotherapy with or without SSM (n = 243)
| Characteristics | Non-SSM (n = 74) | SSM (n = 169) | P |
|---|---|---|---|
| Age | |||
| Mean | 46.8 | 45.8 | 0.4 |
| Median (range) | 47 (29-69) | 47 (25-75) | |
| Race | |||
| White | 53 (29.0) | 130 (71.0) | 0.4 |
| Other | 21 (35.0) | 39 (65.0) | |
| Clinical TNM stage | |||
| Stage II | 46 (24.5) | 142 (75.5) | < 0.0001 |
| Stage III | 28 (50.9) | 27 (49.1) | |
| Tumor size (cm) | |||
| Mean | 4.6 | 3.5 | 0.025* |
| Median (range) | 4 (0.8-20) | 3 (0.5-14) | |
| Year of surgery | |||
| 2007 | 15 (36.6) | 26 (63.4) | 0.3 |
| 2008 | 23 (34.3) | 44 (65.6) | |
| 2009 | 36 (26.7) | 99 (73.3) | |
| Reconstruction type | |||
| Tissue expander followed by implant | 20 (27.0) | 92 (54.4) | < 0.0001 |
| Autologous | 34 (46.0) | 67 (39.6) | |
| Latissimus dorsi flap | 20 (27.0) | 10 (5.9) | |
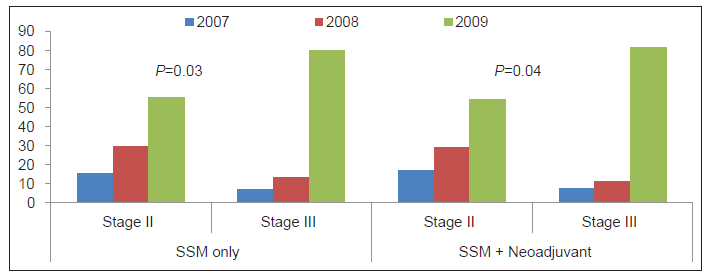
Figure 1 displays by year the percentages of patients with stage II and III disease who underwent SSM, with or without NAC. In the latter years of this study, a statistically significant increase occurred in the percentage of patients with both stage II and III disease who underwent SSM with immediate reconstruction. This increase in SSM with immediate reconstruction was most notable in patients with stage III disease, especially between the time periods 2007 and 2009.
Discussion
In this report, we present our experience with patients with clinical or pathological Stage II and III breast cancer who underwent IBR. We found that approximately 75% of patients with stage II disease and about half of patients with stage III disease underwent SSM. Patients who received NAC followed by SSM with IBR had larger clinical tumors than those who did not. More than half of these patients ultimately had implant-based reconstruction, without the need for additional skin from either a latissimus dorsi flap or other autologous tissue flap. Immediate reconstruction with an autologous tissue flap was affected by the availability of the breast skin envelope as seen in the significantly increased utilization of flaps in non-SSM as opposed to SSM patients. Preservation of the breast skin envelope thus appeared most beneficial for immediate reconstruction with a tissue expander followed by an implant.
Although the use of NAC was not associated with an increase in the use of SSM and IBR (71.4% in all patients, vs. 69.5% in NAC patients, P = 0.3), it was associated with the choice of reconstruction (P < 0.0001). NAC had a moderate effect on the proportion of patients who underwent implant-based or autologous tissue flap reconstruction, and a larger proportional difference in patients who underwent reconstruction with a latissimus dorsi flap plus breast implant. The authors suspect that less breast skin was sacrificed during mastectomies in the NAC cohort, resulting in this difference. However, while fewer SSM patients who had NAC had a latissimus dorsi flap plus breast implant than those who did not have NAC (5.9% vs. 7.2%), this finding was not statistically significant (P = 0.765, Chi-square).
While one purpose of NAC is to facilitate the conversion of mastectomy to BCS, the authors hypothesized that in patients with locally-advanced breast cancer (i.e. stage II and III) it can also reduce the morbidity of mastectomy by converting patients who would otherwise receive non-SSM to SSM. There was a significantly higher percentage of Stage III breast cancer patients in the NAC cohort (22.6% vs. 14.7%, P < 0.05). NAC patients furthermore had larger tumors on average (3.5 cm, vs. 3.1 cm for non-neoadjuvant patients), potentially allowing the use of SSM in more patients who would otherwise not be candidates. Additionally, a greater percentage of SSM patients who had NAC had implant-based breast reconstruction than those who did not have NAC, which may indicate that more mastectomy skin was preserved at the time of SSM. Indeed, the authors found a significant difference in the size of excised skin in non-SSM vs. SSM patients (56.2 cm2vs. 22.3 cm2, P < 0.01). It is our current practice to reconstruct non-SSM patients with either a latissimus dorsi flap + implant or autologous tissue, thus highlighting the role of mastectomy skin preservation in shaping reconstructive choices.
Breast conservation surgery has become an integral part of the management of breast cancer patients. It provides effective locoregional management and reduces the negative psychosocial impact related to mastectomy.[16,17] Its oncologic safety is now documented; it does not increase local or distant recurrence, nor does it adversely affect disease-free or overall survival. Furthermore, there is no significant delay in detection of cancer recurrence.[18-21] However, many patients still require mastectomies as standard treatment for breast cancer.[8,22,23] Many institutions have adopted the use of NAC to facilitate the conversion of mastectomy to breast-conserving surgery or inoperable tumors to operable tumors in women with locally advanced breast cancer.[7] In patients with skin involvement or frank T4b disease, NC has enabled an opportunity for SSM in patients where skin involvement regresses clinically.[20] However, little has been studied regarding the effects of NAC on IBR. Patients who receive IBR have better esthetic results, better psychosocial outcomes, and lower costs of surgery compared to patients who undergo delayed reconstruction or no reconstruction.[24] In these patients, SSM has clear advantages over non-SSM in that it preserves the breast’s three dimensional architecture. Furthermore, many studies have shown that SSM with IBR does not increase local or distant recurrence, demonstrating the oncological safety of this technique.[2,18] IBR after mastectomy is now routinely recommended for selected patients according to the National Comprehensive Cancer Network Guidelines.[19] Compared with non-SSM, SSM is far superior as regards cosmetic outcomes and is expected to remarkably reduce the emotional trauma due to the sense of loss of a breast that is perceived by the patient just after surgery.[25]
Although there have been some studies on the usefulness of SSM in locally advanced breast cancer,[21,26] its application is still debated. It is commonly approved that local control, prognosis, and risk of complications are the same for SSM and NSSM, at least in stages 0, I, and II. SSM is still considered to be contraindicated for inflammatory breast cancer and breast cancer with skin invasion.[25] So far, no study had addressed the issue of the influence of clinical characteristics on the type of breast reconstruction, especially for patients with advanced breast cancer. We found similar utilization of reconstructive modalities between non-NAC and NAC cohorts, despite the increased rate of stage III patients in the NAC cohort. The authors argue that NAC facilitates higher stage patients to undergo SSM-IBR, thus optimizing care in these patients.
Previous reports have documented a decreased receipt of breast reconstruction in NAC patients. Hu et al.[27] revealed that recipients of neoadjuvant therapy are less likely to undergo immediate reconstruction, even after controlling for age, disease stage, and receipt of radiotherapy. They assumed that, the average NAC recipient has a 28% chance of undergoing immediate reconstruction compared with 40% for the average patient who receives only adjuvant chemotherapy. These neoadjuvant recipients, however, are not more likely to progress to delayed reconstruction. Only younger age and lower BMI seem to predict delayed reconstruction among patients who do not undergo immediate reconstruction.[27] Conversely, some patients undergoing NAC may develop treatment fatigue and may be unwilling to undergo elective breast reconstruction.[28]
There are a number of limitations of our study. Since tissue expansion with two-stage reconstruction can be used for patients short of breast skin, a more logical comparison assessing the impact of NAC on SSM-IBR would evaluate direct-to-implant, one-stage implant reconstruction. Since we were only beginning to use this technique at the time of the study, there were not sufficient patients in the study cohort to examine this subject. In addition, our comparison of breast skin specimens (as noted above) makes this examination unnecessary. Direct comparison between NAC and non-NAC groups was confounded because tumor size was statistically larger in the NAC group (3.5 cm vs. 3.1 cm for SSM patients), and there was a statistically higher proportion of stage III patients in the NAC cohort. Patient cohort size did not permit us to confine statistical analysis only to patients with tumors larger than 3.5 cm or stage III patients. In addition, most patients with stage III disease already receive NAC, thus confounding this relationship. Of note, the authors found an increasing percentage of patients undergoing SSM over the course of the study, most notably in Stage III breast cancer patients [Figure 1]. While this is certainly multifactorial in nature, the use of NAC is a likely contributor to this phenomenon. However, a full analysis of this relationship is beyond the scope of this article.
Figure 1. Percentage of patients receiving skin‑sparing mastectomy (SSM) only or SSM with neaoadjuvant chemotherapy, vs. stage of breast cancer, for years 2007‑2009
Thus, far NAC has mainly been considered by medical oncologists as a predictor of response to chemotherapy and by breast surgeons as a means to increase eligibility for partial mastectomy instead of mastectomy. However, its use to reduce the morbidity of mastectomy in patients with Stage II and III breast cancer, including the need to resect breast skin and subsequently enhance reconstructive outcomes, is an intriguing possibility. In this large single-center study, we found that NAC did not statistically increase the use of SSM, but did affect the types of reconstruction used. Further work will help elucidate the role of NAC in IBR.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Prati R, Minami CA, Gornbein JA, Debruhl N, Chung D, Chang HR. Accuracy of clinical evaluation of locally advanced breast cancer in patients receiving neoadjuvant chemotherapy. Cancer 2009;115:1194-202.
2. Monrigal E, Dauplat J, Gimbergues P, Le Bouedec G, Peyronie M, Achard JL, Chollet P, Mouret-Reynier MA, Nabholtz JM, Pomel C. Mastectomy with immediate breast reconstruction after neoadjuvant chemotherapy and radiation therapy. A new option for patients with operable invasive breast cancer. Results of a 20 years single institution study. Eur J Surg Oncol 2011;37:864-70.
3. Alderman AK, Kuhn LE, Lowery JC, Wilkins EG. Does patient satisfaction with breast reconstruction change over time? Two-year results of the Michigan Breast Reconstruction Outcomes Study. J Am Coll Surg 2007;204:7-12.
4. Hu ES, Pusic AL, Waljee JF, Kuhn L, Hawley ST, Wilkins E, Alderman AK. Patient-reported aesthetic satisfaction with breast reconstruction during the long-term survivorship Period. Plast Reconstr Surg 2009;124:1-8.
5. Lee C, Sunu C, Pignone M. Patient-reported outcomes of breast reconstruction after mastectomy: a systematic review. J Am Coll Surg 2009;209:123-33.
6. Fernández-Frías AM, Aguilar J, Sánchez JA, Merck B, Pi-ero A, Calpena R. Immediate reconstruction after mastectomy for breast cancer: which factors affect its course and final outcome? J Am Coll Surg 2009;208:126-33.
7. Rastogi P, Anderson SJ, Bear HD, Geyer CE, Kahlenberg MS, Robidoux A, Margolese RG, Hoehn JL, Vogel VG, Dakhil SR, Tamkus D, King KM, Pajon ER, Wright MJ, Robert J, Paik S, Mamounas EP, Wolmark N. Preoperative chemotherapy: updates of national surgical adjuvant breast and bowel project protocols B-18 and B-27. J Clin Oncol 2008;26:778-85.
8. Song J, Zhang X, Liu Q, Peng J, Liang X, Shen Y, Liu H, Li H. Impact of neoadjuvant chemotherapy on immediate breast reconstruction: a meta-analysis. PLoS One 2014;9:e98225.
9. Mieog JS, van der Hage JA, van de Velde CJ. Preoperative chemotherapy for women with operable breast cancer. Cochrane Database Syst Rev 2007; doi: 10.1002/14651858.CD005002.pub2.
10. Yedibela S, Elad L, Wein A, Dimmler A, Merkel S, Hohenberger W, Meyer T. Neoadjuvant chemotherapy does not increase postoperative complication rate after resection of colorectal liver metastases. Eur J Surg Oncol 2005;31:141-6.
11. Turner II, Russell GB, Blackstock AW, Levine EA. Impact of neoadjuvant therapy on postoperative complications in patients undergoing resection for rectal adenocarcinoma. Am Surg 2004;70:1045-9.
12. Valenti V, Hernandez-Lizoain JL, Baixauli J, Pastor C, Aristu J, Diaz-Gonzalez J, Beunza JJ, Alvarez-Cienfuegos JA. Analysis of early postoperative morbidity among patients with rectal cancer treated with and without neoadjuvant chemoradiotherapy. Ann Surg Oncol 2007;14:1744-51.
13. Milman S, Kim AW, Warren WH, Liptay MJ, Miller C, Basu S, Faber LP. The incidence of perioperative anastomotic complications after sleeve lobectomy is not increased after neoadjuvant chemoradiotherapy. Ann Thorac Surg 2009;88:945-50.
14. Mehrara BJ, Santoro TD, Arcilla E, Watson JP, Shaw WW, Da Lio AL. Complications after microvascular breast reconstruction: experience with 1195 flaps. Plast Reconstr Surg 2006;118:1100-9.
15. Mitchem J, Herrmann D, Margenthaler JA, Aft RL. Impact of neoadjuvant chemotherapy on rate of tissue expander/implant loss and progression to successful breast reconstruction following mastectomy. Am J Surg 2008;196:519-22.
16. Foster JA, Abdolrasulnia M, Doroodchi H, McClure J, Casebeer L. Practice patterns and guideline adherence of medical oncologists in managing patients with early breast cancer. J Natl Compr Canc Netw 2009;7:697-706.
17. Al-Ghazal SK, Sully L, Fallowfield L, Blamey RW. The psychological impact of immediate rather than delayed breast reconstruction. Eur J Surg Oncol 2000;26:17-9.
18. Taylor CW, Horgan K, Dodwell D. Oncological aspects of breast reconstruction. Breast 2005;14:118-30.
19. NCCN.org. 2012 NCCN clinical practice guidelines in oncology: breast cancer. Ver. 1. 2012. Available from: http://www.nccn.org/professionals/ physician_gls/pdf/breast.pdf. [Last accessed on 2014 Oct 23].
20. Prabhu R, Godette K, Carlson G, Losken A, Gabram S, Fasola C, O'Regan R, Zelnak A, Torres M. The impact of skin-sparing mastectomy with immediate reconstruction in patients with Stage III breast cancer treated with neoadjuvant chemotherapy and postmastectomy radiation. Int J Radiat Oncol Biol Phys 2012;82:e587-93.
21. Lim W, Ko BS, Kim HJ, Lee JW, Eom JS, Son BH, Lee TJ, Ahn SH. Oncological safety of skin sparing mastectomy followed by immediate reconstruction for locally advanced breast cancer. J Surg Oncol 2010;102:39-42.
22. Langstein HN, Cheng MH, Singletary SE, Robb GL, Hoy E, Smith TL, Kroll SS. Breast cancer recurrence after immediate reconstruction: patterns and significance. Plast Reconstr Surg 2003;111:712-20.
23. Agrawal A, Grewal M, Sibbering DM, Courtney CA. Surgical and oncological outcome after skin-sparing mastectomy and immediate breast reconstruction. Clin Breast Cancer 2013;13:478-81.
24. Mustonen P, Lepistö J, Papp A, Berg M, Pietiläinen T, Kataja V, Harma M. The surgical and oncological safety of immediate breast reconstruction. Eur J Surg Oncol 2004;30:817-23.
25. Kinoshita S, Nojima K, Takeishi M, Imawari Y, Kyoda S, Hirano A, Akiba T, Kobayashi S, Takeyama H, Uchida K, Morikawa T. Retrospective comparison of non-skin-sparing mastectomy and skin-sparing mastectomy with immediate breast reconstruction. Int J Surg Oncol 2011;2011:876520.
26. Foster RD, Esserman LJ, Anthony JP, Hwang ES, Do H. Skin-sparing mastectomy and immediate breast reconstruction: a prospective cohort study for the treatment of advanced stages of breast carcinoma. Ann Surg Oncol 2002;9:462-6.
27. Hu YY, Weeks CM, In H, Dodgion CM, Golshan M, Chun YS, Hassett MJ, Corso KA, Gu X, Lipsitz SR, Greenberg CC. Impact of neoadjuvant chemotherapy on breast reconstruction. Cancer 2011;117:2833-41.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Goubran S, Halen JV. Effect of neoadjuvant chemotherapy on skin-sparing mastectomy and breast reconstruction modalities in 409 patients. Plast Aesthet Res 2015;2:17-21. http://dx.doi.org/10.4103/2347-9264.149369
AMA Style
Goubran S, Halen JV. Effect of neoadjuvant chemotherapy on skin-sparing mastectomy and breast reconstruction modalities in 409 patients. Plastic and Aesthetic Research. 2015; 2: 17-21. http://dx.doi.org/10.4103/2347-9264.149369
Chicago/Turabian Style
Goubran, Sameh, Jon Ver Halen. 2015. "Effect of neoadjuvant chemotherapy on skin-sparing mastectomy and breast reconstruction modalities in 409 patients" Plastic and Aesthetic Research. 2: 17-21. http://dx.doi.org/10.4103/2347-9264.149369
ACS Style
Goubran, S.; Halen JV. Effect of neoadjuvant chemotherapy on skin-sparing mastectomy and breast reconstruction modalities in 409 patients. Plast. Aesthet. Res. 2015, 2, 17-21. http://dx.doi.org/10.4103/2347-9264.149369
About This Article
Copyright
Data & Comments
Data

 Cite This Article 1 clicks
Cite This Article 1 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.