Robotic-assisted microsurgery for lymphedema treatment
Abstract
The recent development of robotic-assisted microsurgery and supermicrosurgery has raised great expectations to support some of the most demanding microsurgical procedures, which are applied in lymphatic reconstructive surgery to restore lymphatic vascular integrity and treat lymphedema. Procedures such as the establishment of lymphovenous anastomosis (LVA), the harvest of lymph nodes from anatomic locations that reduce donor-side morbidity and the transplantation of the vascularized lymph node flaps (VLNT) present procedures necessitating extreme precision and dexterity in often difficult-to-reach areas, thus pushing the physical limitations of the performing microsurgeon. Despite being limited in number, recent preclinical and clinical studies of independent groups using different robotic systems demonstrate the feasibility of robotic technology to perform supermicrosurgical procedures successfully. The robotic assistance offers unparalleled precision, refining the surgical techniques and minimizing potential side effects, with clinical outcomes comparable to the conventional techniques. Although the relative disadvantages of robotic assistance mostly appear to be related to adequate training and the prolonged learning curve, the technology promises to revolutionize the field of supermicrosurgery and improve the clinical outcomes of lymphatic reconstructive surgery.
Keywords
INTRODUCTION
Lymphedema is defined as the insufficiency of the lymphatic system to efficiently drain interstitial fluid from the periphery, resulting in edema. It is classified as primary or secondary, depending on the cause of the lymphatic disorder; primary lymphedema is a rare genetic disorder, while secondary lymphedema may occur following infection, trauma or iatrogenic intervention[1]. Secondary lymphedema as a result of surgical oncology is one of the most common yet underestimated side effects of the oncologic treatment. It is estimated that approx. 20% of the patients receiving lymphadenectomy as part of their oncologic regime, e.g., breast cancer, other gynecological tumors, urological malignancies, melanomas and sarcomas, will develop lymphedema, with potentially increased risk for lower extremity lymphedema[2,3]. While the gold standard remains conservative decongestive therapy, lymphedema is increasingly treated surgically, as lymphatic reconstructive surgery aims to reestablish the lymphatic network integrity using microsurgical and supermicrosurgical techniques. Recent studies report improved outcomes when lymphedema is treated earlier, as the fibroadipose tissue alterations contribute to the irreversible character of the disease[4,5].
Lymphatic reconstructive surgery for lymphedema includes the installation of lymphovenous anastomosis (LVA) or transfer of vascularized lymphatic tissue (VLNT)[6]. While LVAs redirect lymph into the venous system, the transfer of vascularized lymph nodes supports lymphangiogenesis and allows lymph to drain through the venous system[7]. The efficacy of these surgical approaches in reducing the edema of the affected extremities and improving the quality of life for the treated patients has been assessed in a number of clinical studies, which have been summarized in comprehensive systematic reviews[8,9].
The progress in the development of novel surgical approaches has been supported by the improvement of the surgical armamentarium and the use of robots in surgery has pushed the boundaries of medical innovation. From the first reported use of the daVinci® Surgical Robotic System in a robotic-assisted cholecystectomy twenty years ago[10], the daVinci® technology has been implemented in many surgical specialties to accomplish highly complex minimally invasive interventions[11]. The three-dimensional stereoscopic vision, instruments with increased motion of freedom, scalable movements and elimination of tremor offered by the robotic technology found a number of applications in plastic and reconstructive surgery quickly[12]. Despite these advantages, experimental studies indicated the drawbacks of this technology in microsurgery due to the absence of dedicated, refined instruments of small size and subtle handing that this type of surgery requires[13]. The special and refined needs of reconstructive microsurgery led to the development of specialized robotic systems for microsurgery and supermicrosurgery, which have been found particularly useful in lymphatic reconstructive surgery[14,15].
In this review, we will address the use of robotic surgery in the field of lymphatic reconstructive surgery. We will provide an overview of the various robotic applications, their advantages and disadvantages, as well as the future directions in robotic-assisted supermicrosurgery.
ROBOTIC-ASSISTED PLASTIC AND RECONSTRUCTIVE SURGERY
Robotic technology has been introduced into the field of plastic and reconstructive surgery with a number of applications, ranging from flap harvest to nerve surgery and trans-oral robotic surgery[12].
In a constant effort to improve flap harvesting, robotic-assisted surgery was a promising tool. Decreasing scaring, attempting a less traumatic dissection and increasing the pedicle length have been the driving incentives. Muscle flap harvest has been attempted by different groups for isolated cases, suggesting the feasibility of the method[12]. In the particular case of the DIEP flap harvest for breast reconstruction, the usage of a robot was found to enable a minimally invasive intra-abdominal dissection of the entire pedicle length, reducing the fascial incision to 1.5-3cm while achieving a pedicle length of 10-15cm[16]. Given its well-known abdominal donor site morbidity[17], this suggests an attractive approach to improve outcomes after DIEP flap harvest.
ROBOTIC-ASSISTED HARVEST OF VASCULARIZED LYMPHATIC TISSUE
Autologous lymph node transplantation has widely gained acceptance in lymphedema reconstructive surgery, particularly in advanced and primary lymphedema. In the process of refining the procedure, an effort was paid to decrease the donor site morbidity and thus reduce the risk of causing lymphedema at the harvesting site. Among the different options, the omentum presents an ideal donor for autologous vascularized lymph node transfer. It is abundant in lymphatic tissue, offers a broad surface area and reliable vascularity and eliminates the risk of donor-site lymphedema[18,19].
The use of the omentum was initially limited due to the concern of complications related to intra-abdominal manipulation and the need for laparotomy. The development of laparoscopic techniques clearly improved the harvest and significantly reduced the associated complications. Reduced blood loss, reduced post-operative pain, faster recovery and improved cosmesis are counting among the major benefits of this less invasive technique. But the visualization is still imperfect, restricting the ability of fine dissection. The inclusion of robotic harvest enabled a leap in the omentum flap harvest technique. The robotically assisted harvest offers an unparalleled visualization of the tissue, thus supporting very precise tissue dissection and pedicle preparation. What is more, the risk of damaging adjacent anatomical structures is minimized due to the tremor amortization and increased motion of freedom. The inclusion of additional imaging tools, such as fluorescent optics to visualize the blood and lymphatic vascular patterns, allows for improving the flap design and harvest[20,21].
Despite the longer operating times in comparison to the laparoscopically assisted surgery and the specialized training needed, the robotic harvest presents a promising approach in lymph node harvest for lymphatic reconstructive surgery.
ROBOTIC-ASSISTED MICROSURGERY AND SUPERMICROSURGERY IN LYMPHEDEMA
It is without a doubt that the development and establishment of the VLNT and LVA techniques have drastically changed lymphedema treatment, particularly given that no pharmacological treatment is still currently available and the conservative measurements cannot correct the underlying lymphatic vascular compromise. Many prospective and retrospective studies highlight the positive outcomes of lymphatic reconstructive surgery, namely volume and circumference reduction, improved quality of life and reduction of compression garment use[18,22]. Both techniques are extremely refined and technically demanding, with strong physical demand for the performing microsurgeon. A significant level of experience is necessary, along with the acquisition of challenging surgical skills[23]. Thus, technical improvements in the surgical armamentarium used are needed to improve surgical outcomes.
With the urge to constantly improve and refine surgical techniques, the development of robotic-assisted supermicrosurgery was introduced into lymphatic reconstructive surgery. Lymphatic microsurgeons are confronted with the anastomosis of vessels with a diameter between 0.3 to 0.8 mm for the reconstruction of lymphatic flow and the transplantation of pedicled lymph nodes in often hard-to-reach areas, e.g., the axilla. In particular, for the performance of LVAs, extremely fine nylon sutures (11-0 or 12-0) on a 50 µm needle are required, defining undoubtedly extremely technically demanding circumstances. Even for experienced surgeons with outstanding skills and experience, the surgical performance is still limited by the precision and dexterity of the human hands[12,24].
Robotic supermicrosurgery facilitates these procedures, helping microsurgeons overcome these limitations. Robotic assistance provides complete tremor amortization and motion scaling up to 20x. This leads to increased precision and unparalleled steadiness, particularly when handling or preparing extremely small and fragile lymphatic vessels or performing anastomosis with size mismatch or in deeper body cavities. The presence of flexible, free-moving robotic arms and seven degrees of freedom enables the deployment of the robot even in deeper and less accessible anatomic locations. While the microsurgery robots are compatible with existing operation microscopes, three-dimensional visualisations systems, also referred to as exoscopes, may contibute in a better spacial vision in light of the the absent “haptic” feedback. Additionally, the recent development of robotic systems without fixed joysticks[15] but with a remote console further improves the surgeon’s ergonomic position and endurance performance.
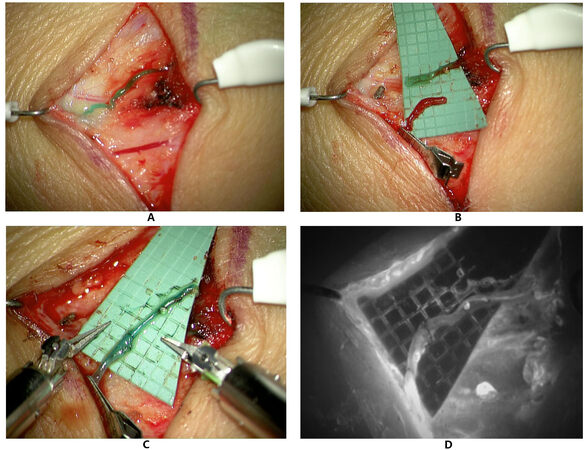
Currently, there are two robotic microsurgery systems available. The robotic system MUSA® (MicroSure, Eindhoven, The Netherlands) developed in 2014 is the first available system of its kind[25]. It is equipped with dedicated supermicrosurgical instruments. However, it is mounted to the surgical table with fixed joysticks. Its feasibility for microsurgery has been demonstrated in both preclinical and clinical models[14,25]. The second available system is the Symani® Surgical System [Medical Microinstruments (MMI), Pisa, Italy] which was designed the second available system is the Symani® Surgical System (Medical Microinstruments (MMI), Wilmington, DE, USA) which was designed to provide movable manipulators istead of fixed handling joysticks [Figure 1]. In the system, the specialized microsurgical instruments are connected to flexible robotic arms, which are guided through freely movable forceps-like joysticks. The system also allows teleoperation, and the forceps-like joystick’s similarity to conventional micro-instruments has been reported to enhance the robot-assisted experience[15].
Figure 1. Presentation of a robotic-assisted lymphovenous anastomosis performed with the Symani Surgical System®. (A): 0.5 mm lymphatic vessel (above) and 0.5 mm vein (below) after intradermal injection of 0.2 mL Indocyanine green (ICG)/patent blue. (B): Proximal transection of the lymphatic and distal transection of the vein for end-to-end anastomosis. An intravascular stent (IVAS) was used for vessel stabilization during anastomosis. (C): Robot-assisted lymphovenous anastomosis with Nylon 11-0 showing good patency. (D): Fluorescent mode confirming lymphatic flow of ICG into the vein.
The available but limited literature reporting the first experiences of the robotic system application in lymphatic reconstructive procedures[15,25], including the personal experience of the senior author of this manuscript, suggests the technical feasibility of the technique, with clinical outcomes comparable between robotic-assisted and conventional lymphatic surgery[26,27] [Table 1]. However, potential drawbacks of these initial applications of the new technology definitely exist and are analyzed below.
Summary of clinical studies using robotic-assisted lymphatic surgery
| Publication | Type of robot | Type of surgery | Total Nr of patients | Nr of patients with robotic anastomosis | Nr of robotic anastomosis | Time (min) for robotic anastomosis | Time (min) for manual anastomosis | Year of publication |
| van Mulken | MUSA | LVAs | 20 | 8 | 14 | 25 ± 6 min and a range 16-33 min | 9 ± 6 min and range 4-36 min | 2020 |
| van Mulken | MUSA | LVAs | 20 | 8 | 14 | 2021 | ||
| Lindenblatt | Symani | LVA & VLNT | 5 | 5 | 10 | 2022 | ||
| Barbon, Lindenblatt | Symani | LVA & VLNT | 22 | 22 | 32 | 25.3 ± 12.3 min | 14.1 ± 4.3 min | 2022 |
THE CHALLENGES OF ROBOTIC-ASSISTED (SUPER)MICROSURGERY
Despite the obvious advantages of using robotic-assisted supermicrosurgery, a number of limiting factors have to be acknowledged as well. The major obstacle in the broad integration of robotic technology in the surgical routine is the learning curve and the initially increased operating times. The published literature indicates increased anastomosis times using the robot versus the manual technique, even for very experienced microsurgeons. However, the learning curve was found to be steep, with the quick improvement in the operating times. The frequency of practice and level of microsurgical experience were found to support faster improvement and significantly decrease anastomosis time[27,28].
Furthermore, the absence of haptic feedback and the need for the performing surgeon to develop a “see-feel” concept during the performance of the anastomosis is a relative limiting factor. The use of adequate imaging support and training has been reported to significantly and rapidly improve the absence of sensorial feedback, especially among already experienced surgeons[15,29].
Lastly, the increased costs to purchase and maintain the robot, the expensive robotic consumables and instruments, as well as the need to have an appropriately educated operating room team to maintain time efficiency have to be taken into consideration and may limit the accessibility and adoption of the technology.
FUTURE PERSPECTIVES
As research continues, further improvement of the robotic systems available is expected, as well as the development of new robots for specific indications. This progress is expected to enhance surgical precision and enable the expansion of surgical procedures.
The expansion of microsurgical instruments to improve the degree of articulation and meet different size demands will decisively influence the increase of robotic deployment. Additionally, the development of new instruments or miniaturized versions of existing ones will augment the surgeon’s capabilities. Instruments to dissect vessels and tissue will be introduced. The inclusion of biosensors and improvement in the haptic feedback may restore one of the main drawbacks of robotic surgery, even if visual cues can mimic the perception of tactile feedback. This is particularly crucial in supermicrosurgery, as the surgeon is unable to sense the forces applied to the fragile lymphatic capillaries. Thus, the inclusion of haptic feedback could further improve surgical precision and atraumatic handling[26,29].
The optimal visualization of the operating field presents another topic of intense research that decisively influences supermicrosurgery. The development, refinement or integration of imaging modalities such as three-dimensional imaging, high-spectral imaging or near-infrared fluorescence imaging could facilitate the intraoperative anatomical navigation, support the functional assessment of anastomosis patency and partially compensate for the absence of haptic feedback[29,30].
Last but not least, the inclusion of artificial intelligence systems in robot-assisted systems promises to bring surgical techniques to a new level. The recording and analyzing of the surgical movements used in intelligent robots will result in the development of cognitive skills and a process of “self-learning”, thus leading to semi-automated surgical applications. The possibilities for improving surgical techniques and training can be breathtaking[31].
CONCLUSION
Following the establishment of lymphatic reconstructive surgery as the only means currently available to at least partially restore lymphatic integrity, the effort is now placed on refining the techniques used, improving surgical outcomes and minimizing potential side effects.
Due to the extreme nature of lymphatic surgery, surgeons face technical and physical limitations. However, robotic-assisted supermicrosurgery enables the performance of this delicate surgery beyond the physical capabilities of the human hands, offering unprecedented dexterity, accuracy and endurance. In addition robotic systems will make access to the central part of the lymphatic sytsem, e.g., the thoracic duct more accessible[32].
The literature available so far demonstrates the feasibility of the technology and favorable clinical outcome, with a considerable but steep learning curve effect. Undoubtedly tremendous potential is available, empowering the growth and hopefully the accessibility of robotic-assisted supermicrosurgery, identifying novel applications for the patient`s needs and optimizing surgical outcomes.
DECLARATIONS
Authors’ contributionsReview conception and design: Gousopoulos E, Grünherz L, Lindenblatt N
Manuscript preparation: Lindenblatt N
Critical revisions: Gousopoulos E, Grünherz L, Giovanoli P, Lindenblatt N
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestNicole Lindenblatt acts as a symposium speaker and clinical advisor for Medical Microinstruments.
All other authors have no conflict of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationWritten consent to publish the intraoperative photos has been given by the patient.
Copyright© The Author(s) 2023.
REFERENCES
1. Lee B-B, Bergan JJ, Rockson SG. Lymphedema : a concise compendium of theory and practice. London: Springer;2011.
2. DiSipio T, Rye S, Newman B, Hayes S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol 2013;14:500-15.
3. Hayes SC, Janda M, Ward LC, et al. Lymphedema following gynecological cancer: Results from a prospective, longitudinal cohort study on prevalence, incidence and risk factors. Gynecol Oncol 2017;146:623-9.
4. Felmerer G, Sattler T, Lohrmann C, Tobbia D. Treatment of various secondary lymphedemas by microsurgical lymph vessel transplantation. Microsurgery 2012;32:171-7.
5. Grünherz L, Angst F, Barbon C, et al. Cultural adaption and multicenter validation of the German version of the LYMPH-Q Upper Extremity Module. J Vasc Surg Venous Lymphat Disord 2022;10:922-928.e2.
6. Uyulmaz S, Scaglioni MF, Lindenblatt N. Chirurgische Therapie des Lymphödems - Möglichkeiten und Grenzen. Praxis (Bern 1994) 2017;106:1331-8.
7. Garcés M, Pons G, Mirapeix R, Masià J, et al. Intratissue lymphovenous communications in the mechanism of action of vascularized lymph node transfer. J Surg Oncol 2017;115:27-31.
8. Chun MJ, Saeg F, Meade A, et al. Immediate lymphatic reconstruction for prevention of secondary lymphedema: a meta-analysis. J Plast Reconstr Aesthet Surg 2022;75:1130-41.
9. Kong X, Du J, Du X, Cong X, Zhao Q. A Meta-analysis of 37 studies on the effectiveness of microsurgical techniques for lymphedema. Ann Vasc Surg 2022;86:440-451.e6.
10. Ballantyne GH. Robotic surgery, telerobotic surgery, telepresence, and telementoring. Review of early clinical results. Surg Endosc 2002;16:1389-402.
12. Dobbs TD, Cundy O, Samarendra H, Khan K, Whitaker IS. A systematic review of the role of robotics in plastic and reconstructive surgery-from inception to the future. Front Surg 2017;4:66.
13. der Hulst R, Sawor J, Bouvy N. Microvascular anastomosis: is there a role for robotic surgery? J Plast Reconstr Aesthet Surg 2007;60:101-2.
14. van Mulken TJM, Boymans CAEM, Schols RM, et al. Preclinical experience using a new robotic system created for microsurgery. Plast Reconstr Surg 2018;142:1367-76.
15. Lindenblatt N, Grünherz L, Wang A, et al. Early experience using a new robotic microsurgical system for lymphatic surgery. Plast Reconstr Surg Glob Open 2022;10:e4013.
17. Lindenblatt N, Gruenherz L, Farhadi J. A systematic review of donor site aesthetic and complications after deep inferior epigastric perforator flap breast reconstruction. Gland Surg 2019;8:389-98.
18. Scaglioni MF, Arvanitakis M, Chen YC, Giovanoli P, Chia-Shen Yang J, Chang EI. Comprehensive review of vascularized lymph node transfers for lymphedema: Outcomes and complications. Microsurgery 2018;38:222-9.
19. Forte AJ, Cinotto G, Boczar D, Huayllani MT, McLaughlin SA. Omental lymph node transfer for lymphedema patients: a systematic review. Cureus 2019;11:e6227.
20. Frey JD, Yu JW, Cohen SM, Zhao LC, Choi M, Levine JP. Robotically assisted omentum flap harvest: a novel, minimally invasive approach for vascularized lymph node transfer. Plast Reconstr Surg Glob Open 2020;8:e2505.
21. Özkan Ö, Özkan Ö, Çinpolat A, Arıcı C, Bektaş G, Can Ubur M. Robotic harvesting of the omental flap: a case report and mini-review of the use of robots in reconstructive surgery. J Robot Surg 2019;13:539-43.
22. Nacchiero E, Maruccia M, Elia R, Robusto F, Giudice G, Manrique OJ, Campisi CC, Giudice G. Lymphovenous anastomosis for the treatment of lymphedema: a systematic review of the literature and meta-analysis. Lymphology 2020;53:172-94.
23. Chang EI, Skoracki RJ, Chang DW. Lymphovenous anastomosis bypass surgery. Semin Plast Surg 2018;32:22-7.
24. Ibrahim AE, Sarhane KA, Selber JC. New frontiers in robotic-assisted microsurgical reconstruction. Clin Plast Surg 2017;44:415-23.
25. Mulken TJM, Schols RM, Scharmga AMJ, et al. MicroSurgical Robot Research Group. First-in-human robotic supermicrosurgery using a dedicated microsurgical robot for treating breast cancer-related lymphedema: a randomized pilot trial. Nat Commun 2020;11:757.
26. van Mulken TJM, Wolfs JAGN, Qiu SS, et al. MicroSurgical Robot Research Group. One-year outcomes of the first human trial on robot-assisted lymphaticovenous anastomosis for breast cancer-related lymphedema. Plast Reconstr Surg 2022;149:151-61.
27. Barbon C, Grünherz L, Uyulmaz S, Giovanoli P, Lindenblatt N. Exploring the learning curve of a new robotic microsurgical system for microsurgery. JPRAS Open 2022;34:126-33.
28. Alrasheed T, Liu J, Hanasono MM, Butler CE, Selber JC. Robotic microsurgery: validating an assessment tool and plotting the learning curve. Plast Reconstr Surg 2014;134:794-803.
29. van Mulken TJM, Schols RM, Qiu SS, et al. Robotic (super) microsurgery: feasibility of a new master-slave platform in an in vivo animal model and future directions. J Surg Oncol 2018;118:826-31.
30. Cornelissen AJM, van Mulken TJM, Graupner C, et al. Near-infrared fluorescence image-guidance in plastic surgery: A systematic review. Eur J Plast Surg 2018;41:269-78.
31. Bhandari M, Zeffiro T, Reddiboina M. Artificial intelligence and robotic surgery: current perspective and future directions. Curr Opin Urol 2020;30:48-54.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Gousopoulos E, Grünherz L, Giovanoli P, Lindenblatt N. Robotic-assisted microsurgery for lymphedema treatment. Plast Aesthet Res 2023;10:7. http://dx.doi.org/10.20517/2347-9264.2022.101
AMA Style
Gousopoulos E, Grünherz L, Giovanoli P, Lindenblatt N. Robotic-assisted microsurgery for lymphedema treatment. Plastic and Aesthetic Research. 2023; 10(1): 7. http://dx.doi.org/10.20517/2347-9264.2022.101
Chicago/Turabian Style
Gousopoulos, Epameinondas, Lisanne Grünherz, Pietro Giovanoli, Nicole Lindenblatt. 2023. "Robotic-assisted microsurgery for lymphedema treatment" Plastic and Aesthetic Research. 10, no.1: 7. http://dx.doi.org/10.20517/2347-9264.2022.101
ACS Style
Gousopoulos, E.; Grünherz L.; Giovanoli P.; Lindenblatt N. Robotic-assisted microsurgery for lymphedema treatment. Plast. Aesthet. Res. 2023, 10, 7. http://dx.doi.org/10.20517/2347-9264.2022.101
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 17 clicks
Cite This Article 17 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.