Pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring: power and pitfalls
Abstract
Outpatient electrodiagnostic studies and intraoperative monitoring are essential tools for the surgical management of nerve injury. Decisions are based on the diagnostic certainty afforded by outpatient electrodiagnostic studies, which are more sensitive and specific than clinical examinations regarding the nature and localization of a nerve lesion. Intraoperative neurophysiologic monitoring detects changes in neurologic function during surgery. It provides significantly better information than visual inspection of the operative field, minimizing postoperative neurologic deficits due to surgical manipulation (e.g., stretching, compression, heating from electrocauterization, constriction, or clamping of local blood vessels). These techniques exploit similar neurophysiologic principles to afford enhanced diagnostic and real-time functional data during surgery. However, an understanding of their limitations is critical for the interpretation of these data. This review discusses these techniques, including their use, advantages, and disadvantages in diagnosing and managing three essential nerve lesions amenable to surgical management-radiculopathy, mononeuropathy, and brachial plexopathy.
Keywords
INTRODUCTION
Undergoing surgery introduces significant inherent risks to patients, particularly those risks involving the nervous system[1]. Surgical manipulation, including stretching, compression, heating from electrocauterization, and constriction or clamping of local blood vessels causing local ischemia, can pose risks to local neural structures, potentially resulting in irreversible neurologic injury. Since the surgeon’s visual inspection of the operative field provides limited information, pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring (IONM) can significantly improve surgical outcomes. Pre-operative electrodiagnostic studies allow for more precise diagnosis and localization of nerve injury by providing information about the duration, severity, and outcomes[2-6]. Intraoperative neurophysiologic studies provide real-time functional data that allows intervention to eliminate or minimize intraoperative injury[7]. IONM also provides the opportunity to test segments of nerve inaccessible to surface testing and, therefore, the ability to supplement the pre-operative electrodiagnostic data.
The physiologic elements of pre-operative testing include nerve conduction studies paired with electromyography. IONM includes free-running electromyography, stimulated electromyography, and intraoperative direct nerve stimulation. In this review, we will briefly describe these techniques in the context of three essential types of nerve injury: (1) radiculopathy; (2) brachial plexopathy; and
NEUROPHYSIOLOGIC TECHNIQUES
Pre-operative diagnostic nerve conduction studies and electromyography
Pre-operative electrodiagnostic testing should be considered to allow a better understanding of the nature and extent of the nerve injury to facilitate surgical planning. Outpatient electrodiagnostic studies include two complementary techniques, nerve conduction studies, and needle electromyography; these techniques provide distinct types of data regarding pathophysiologic changes at different levels of the peripheral neuraxis.
Nerve conduction studies
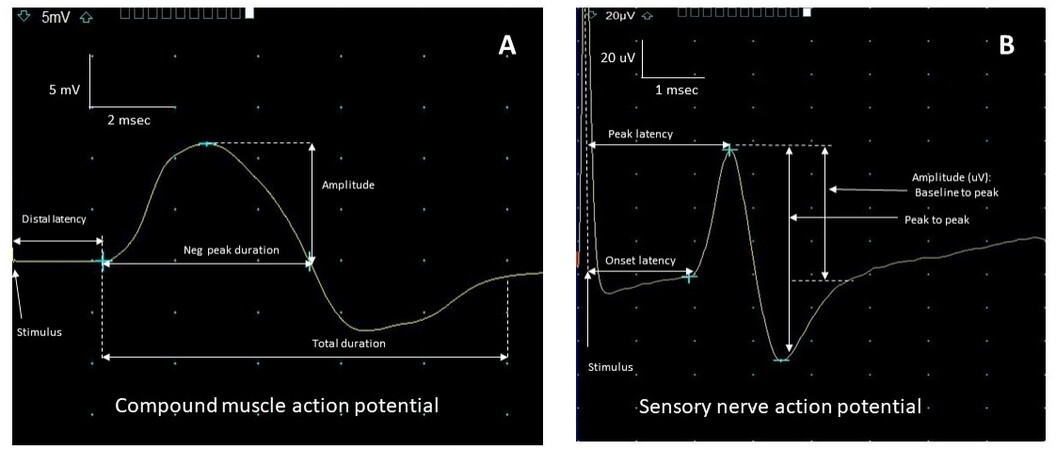
Nerve conduction studies (NCS) are performed using peripheral nerve stimulation to record an evoked sensory or motor potential. This is achieved by applying a brief electrical pulse to the skin overlying the nerve where it becomes superficial. A motor, sensory or mixed potential is obtained by recording electrodes on the skin over a muscle, cutaneous sensory, or mixed nerve[2,3,8]. The result is a recorded compound muscle (CMAP) or sensory nerve action potential (SNAP), the former representing a summated potential derived from muscle fibers and the latter a summated response from nerve fibers [Figure 1]. The amplitude of the response reflects the electrical activity of all the individual muscle or nerve fiber action potentials; a reduced amplitude is, therefore, indicative of axonal loss of nerve or muscle fibers. In the case of the CMAP, the low amplitude can also result from a drop out of signal between the stimulation site and recorded muscle, or conduction block, due to segmental demyelination[8]. Conduction block is essential in the electrodiagnosis of nerve entrapments not only as a diagnostic hallmark but also because of the implication for better outcomes[9,10]. The CMAP amplitude can also be affected early on by local or distal sprouting from adjacent and preserved nerve axons to the recorded muscle. This can account for amplitude improvement in the first year after injury[11]. Recognition of the effect of distal sprouting on CMAP is essential because it does not reflect an effect of axon regrowth from the proximal site of injury. Therefore, CMAP amplitude in the months after an injury may increase without meaning that axons are being regenerated from the injury site, whereas CMAP amplitude increases in a chronic injury that must occur because of regrowth from the original injury. This timing is essential in distinguishing true nerve recovery from nerve compensation for injury[11,12].
Figure 1. Nerve conduction responses. A: Stimulation of motor axons recording over the muscle with detecting a compound muscle action potential (CMAP). Amplitude reflects the number of muscle fibers contributing to the response; distal latency reflects the time (msec) it takes for a stimulus to pass along a nerve to produce the CMAP. B: Stimulation of a sensory nerve recording a sensory nerve action potential (SNAP). Amplitude reflects the number of nerve axons contributing to the response. Distal latency is equivalent to conduction velocity.
The conduction speed of an impulse (velocity, m/s) between two points on a nerve, on the other hand, is generally a reflection of myelin integrity. This is affected early in the setting of focal compression, even before evidence of axonal loss. Myelin loss due to compression can slow the response early on and block the impulse’s conduction to individual fibers. In this case (conduction block), the proximal CMAP is reduced, but the distal CMAP is not, and there is clinical weakness without distal loss of amplitude (axonal loss) or other indicators of axonal loss[9,10]. This is because weakness is due to a drop out of signal in otherwise intact nerve fiber axons. Conduction block is an essential source of weakness that carries much better outcomes than axonal injury; therefore, its detection on NCS can be very informative for surgical planning[4]. Nerve conduction studies, therefore, allow for identifying the fiber type involved (motor, sensory or mixed), the pathophysiology of the injury (axonal or demyelinating), localization, and outcomes. The nerves selected for the study will depend on the clinical context derived from the initial clinical question.
Needle electromyography
Needle electromyography (EMG) is subsequently performed on selected muscles to localize a lesion further and determine its age[13]. This is achieved by detecting aberrant electrical activity from muscle fibers and morphological changes in the structure and recruitment of the motor unit action potential (MUAP) during voluntary contraction. In this portion of electrodiagnostic testing, a needle electrode is placed into a muscle of interest. Movement of the electrode through the muscle at rest results in a burst of muscle fiber potentials that generally last no longer than 300 ms after needle movement. In denervated muscle, single muscle fibers spontaneously twitch at rest due to denervation hypersensitivity; this is detected as fibrillation potentials and positive sharp waves that reflect the activity of denervated muscle fibers due to nerve fiber axonal loss[2,4,8,13,14].
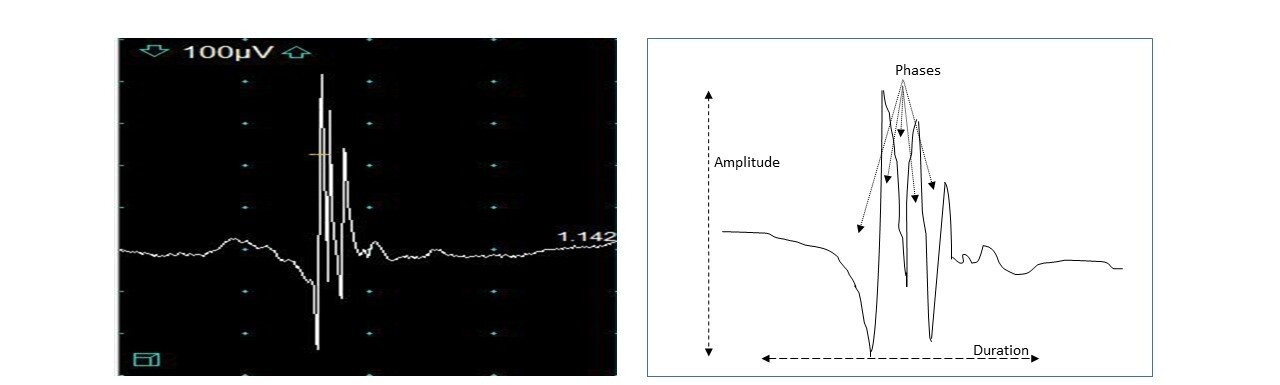
When the muscle is contracted, the morphology of individual MUAPs and the recruitment of those potentials are studied [Figure 2]. Recruitment reflects the gradual addition of more MUAPs as increasing force is generated in a muscle. Reduced recruitment occurs when an insufficient number of MUAPs are recruited for a given muscle contraction; this occurs in the setting of a neurogenic lesion or with sufficient demyelination to cause conduction block (drop out of the nerve action potential due to demyelination). Changes in the duration, number of phases (polyphasia), or amplitude of a MUAP reflect chronic changes resulting from reinnervation. These changes can reflect the reinnervation of muscle fibers as collateral nerve fibers sprout from adjacent intact axons. The conduction speed of sprouting nerve fibers is slower concerning mature branches due to their size and incomplete or absent myelination; therefore, the potential from the muscle fibers innervated by new sprouts is slightly delayed increasing in phases, turns, and the duration of the MUAP [Figure 2]. This reflects the asynchronous firing of potentials from a reinnervated muscle[2,4,8,13-15]. Therefore, polyphasia can occur in the setting of early reinnervation with poorly myelinated axonal sprouts that eventually become more established, allowing for the polyphasia to resolve (see “Time course of MUAP changes”).
Figure 2. Motor unit action potential. Amplitude reflects the number of muscle fibers contributing to a potential elicited from a single nerve fiber. Duration reflects dispersion in neuromuscular activation of branches to muscle fibers. Phases reflect baseline crossings and relate to the increased complexity of motor units due to reinnervation. Amplitude is 100 µV per division. The sweep speed is 5 msec per division.
Time course of MUAP changes
A predictable pattern of MUAP changes is seen after nerve injury resulting in axonal loss. Within the first week to 2 weeks, MUAP morphology is normal, but recruitment is reduced, reflecting drop out of MUAPs innervated by injured axons. By 4 weeks after injury, distal sprouting to denervated muscle fibers occurs from adjacent axons; this results in polyphasic MUAPs because of unstable neuromuscular transmission and asynchronous conduction along immature, poorly myelinated/unmyelinated nerve fiber sprouts branching from intact axons, as described above. These are referred to as “nascent units”. Because reinnervation is still early and nascent units have reinnervated a few muscle fibers, motor units can appear small. As more distant muscle fibers are reinnervated over the subsequent several months, the MUAP size and duration grow while remaining polyphasic. After ~6 months, the degree of polyphasia will diminish as nascent units mature and develop more stable neuromuscular transmission and conduction. However, MUAPs will continue to appear long in duration and large amplitude, reflecting the more significant number of muscle fibers taken over by the reinnervating motor unit[2,4,13-15].
Intraoperative electrodiagnostic techniques
Electrodiagnostic techniques, including NCS, free-running, and stimulated EMG, have been used in the surgical setting for over five decades. These techniques have been helpful for various surgeries, particularly involving the spine, nerve roots, and peripheral and cranial nerves.
EMG can be monitored intraoperatively in any muscle accessible by paired surface or needle electrodes, which provide a better signal-to-noise ratio. After the patient is anesthetized and sedated, multiple EMG electrodes are placed into or over the limb or cranial muscles of interest based on which neural structures are at risk during the surgery. EMGs are recorded continuously during the surgical procedure with low-noise differential amplifiers. Due to significant electrical noise emanating from the many electronic devices within the operating room, neurophysiologic signals must undergo analog and digital processing, i.e., amplification and filtering. The recordings are visually displayed and can also be broadcast from a speaker so changes in muscle activity can be heard, providing auditory feedback to the intraoperative neurophysiologic monitoring (IONM) team and surgeons. Bursts and runs of neurotonic discharges have characteristic high-frequency visual appearances and sounds, which can alert the team of perturbation or impending injury to neural structures if correlated to the surgical procedure. We would expect such discharges to occur in the setting of nerve perturbation. Alterations of EMG activity and nerve conduction can result not only from the surgical procedure itself but also due to other intraoperative factors, e.g., neuromuscular blockade, level of sedation, temperature, ambient electrical noise, neurologic conditions, and technical issues. Therefore, false positive and false negative changes should be considered and identified.
Muscle selection to maximize coverage for the relevant anatomic region is the best IONM strategy. Careful pre-operative planning is essential since it may not be possible to place additional EMG recording electrodes after the surgery is in progress. The most accessible muscles for IONM lie superficially within the cranial, cervical, lumbar, and sacral myotomes. Common EMG recording sites utilized for monitoring specific spinal nerve root levels and peripheral nerves are shown in Table 1[16,17]. Most muscles are innervated by at least two adjacent nerve roots, so monitoring multiple muscles is essential for accurate localization of neural irritation and injury. For example, irritation of the L4 nerve root might result in abnormal EMG activity solely in the tibialis anterior, whereas surgical manipulation of the L5 nerve root may cause abnormal discharges in the tibialis anterior and biceps femoris muscles. Technical issues can also affect the intraoperative EMG, so simultaneously recording from uninvolved remote muscles to serve as controls can be of great utility, e.g., troubleshooting[3-7].
Common EMG muscles for monitoring specific peripheral nerves and spinal nerve root levels[16,17]
| Upper limb muscles | Nerve | Spinal roots |
| Trapezius | Spinal accessory | C3, C4 |
| Deltoid | Axillary | C5, C6 |
| Biceps | Musculocutaneous | C5, C6 |
| Triceps | Radial | C6, C7, C8 |
| Brachioradialis | Radial | C5, C6 |
| Extensor carpi radialis longus | Radial | C6, C7 |
| Extensor carpi ulnaris | Posterior interosseous | C7, C8 |
| Pronator teres | Median | C6, C7 |
| Flexor carpi radialis | Median | C6, C7 |
| Abductor pollicis brevis | Median | C8, T1 |
| Pronator quadratus | Anterior interosseous | C7, C8 |
| Flexor pollicis longus | Anterior interosseous | C8, T1 |
| Flexor carpi ulnaris | Ulnar | C7, C8, T1 |
| Abductor digiti minimi | Ulnar | C7, C8, T1 |
| First dorsal interosseous | Ulnar | C7, C8, T1 |
| Iliopsoas | Femoral | L3, L4 |
| Vastus medialis and lateralis | Femoral | L3, L4, L5 |
| Tibialis anterior | Deep fibular | L4, L5 |
| Biceps femoris | Sciatic | L5, S1 |
| Extensor hallucis longus | Deep fibular | L5, S1 |
| Gastrocnemius | Tibial | S1, S2 |
| Abductor hallucis | Medial plantar | S1, S2, S3 |
| Anal sphincter | Pudendal | S2, S3, S4 |
Free-running spontaneous electromyography
Continuous electromyography is monitored from muscles innervated by nerves or nerve roots at risk for injury during surgery. Usually, the recorded EMG signals will consist of a broad mixture of motor units. Surgical manipulation, such as stretching or compression of peripheral nerves, branches, or nerve roots, can result in high-frequency bursts or trains of neurotonic discharges in the corresponding muscle(s). Such changes in muscle activity provide valuable instantaneous feedback to the monitoring team and the surgeons regarding the surgical manipulation and actions and their proximity to neural structures. In this manner, EMG monitoring is beneficial for preventing severe or irreversible nerve injury from inadvertent trauma.
Irritation related to the surgical procedure activates motor units in various patterns that several factors, such as the nerve condition, the degree of surgical manipulation, and the neuromuscular integrity, can influence. Ischemia from compression of the local blood supply can be another cause of such changes in neural activity. Simultaneous activation of multiple motor units can result in EMG “bursts”, which are brief periods of polyphasic EMG activity [Figure 3]. EMG bursts can often inform the surgeon of proximity to a peripheral nerve or nerve root. Continuous compression or traction of nerves or nerve roots can result in the repetitive firing of one or more motor units for seconds to minutes producing an EMG “train” [Figure 4]. The higher amplitude or frequency trains are often due to significant nerve fiber recruitment from the excessive local force, which could be a harbinger of nerve injury if sustained. In general, the degree of motor nerve irritation correlates roughly with the intensity of EMG activity, i.e., the duration, frequency, and complexity of neurotonic activity are often associated with its severity. Despite the utility of neurotonic discharges, their presence does not necessarily indicate nerve damage, and their absence does not exclude this possibility. Mechanical stimulation can be sufficient to induce EMG discharges but not permanent neural injury. In addition, damaged nerves are less likely than healthy nerves to produce discharges[18-20]. However, persistent neurotonic EMG activity, despite cessation of the surgical factor(s), can indicate the degree of local nerve injury. The EMG may become quiescent after the transection of a nerve with or without an initial large burst or train of neurotonic activity. In addition, manipulation and stimulation of the distal nerve stump may still activate the muscle, leading the surgical team to erroneously conclude that the motor nerve remains intact[18-20].
Figure 3. Spontaneous EMG recording demonstrating two large bursts of neurotonic activity in the orbicularis oris muscle during facial nerve retraction.Amplitude is 20 µV per division. The sweep speed is 400 msec per division.
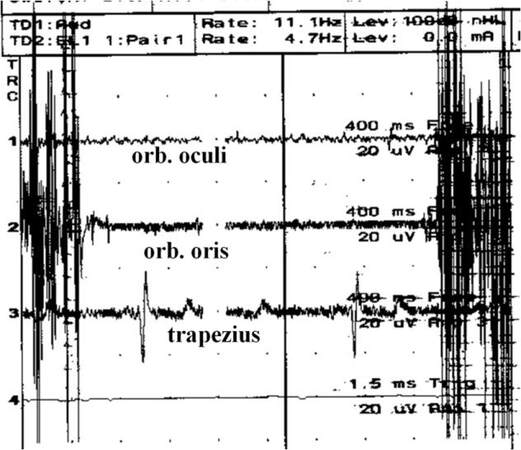
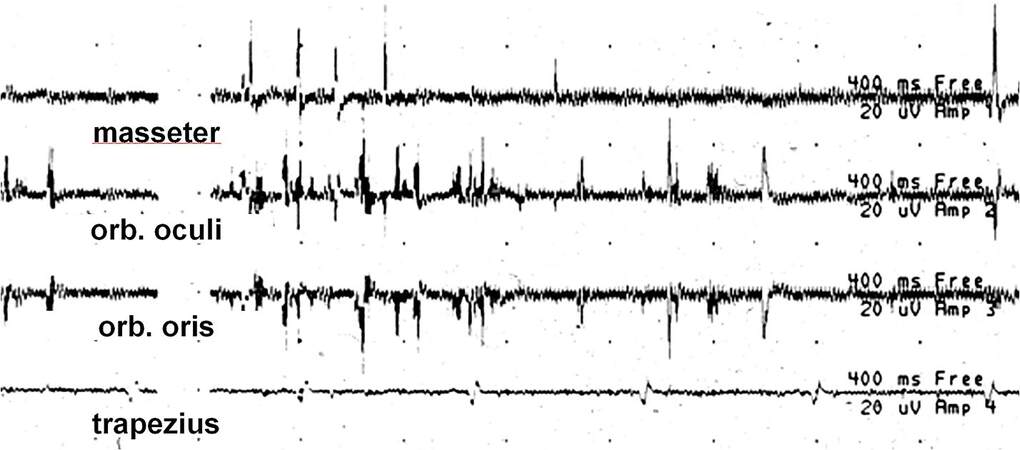
Figure 4. Spontaneous EMG recording demonstrating trains of neurotonic activity from several motor units in the orbicularis oculi and multiple motor units in the orbicularis oris muscles during facial nerve retraction. Note the superimposed electrocardiogram artifact in the trapezius muscle recording. Amplitude is 20 µV per division. The sweep speed is 400 msec per division.
It can be determined whether or not the change in EMG activity is related to a surgical action by temporal and anatomic correlation. If there is no relation of the activated muscles to the timing and location, e.g., spinal nerve root or peripheral nerve, of the surgical manipulation, it is unlikely that there is a causative relationship. For example, increased EMG activity in the abductor pollicis brevis muscle (C8, T1) cannot be explained by surgical manipulation near the ipsilateral C5 spinal nerve root. Other potential causes of neurotonic discharges include temperature changes related to local cool saline irrigation, heat from electrocautery, or local osmotic changes. Determining mechanical versus non-mechanical causes is essential since it is less common for the latter to result in neurotonic activity or neural damage. However, mechanical irritation can pose a significant risk of injury, particularly with repetitive trauma.
Electrodes detect interference or ambient electrical noise from various sources within the operating room, such as bed warmers, pumps, electrocautery devices, drills, or microscopes. It is essential to distinguish such artifacts from EMG activity [Figure 5]. In addition, various neuromuscular conditions and treatments can interfere with EMG recording, e.g., muscular dystrophy, myasthenia gravis, chronic neurogenic conditions, and botulinum toxin. EMG activity can also change due to several non-surgical factors, including the level of sedation and pharmacologic neuromuscular blockade. Motor unit potentials may saturate the recording due to irritation of multiple axons or voluntary muscle activation as the patient begins to awaken from the sedated state [Figure 6]. This EMG activity is usually bilateral and often involves the axial muscles. In addition, it is an anesthetic requirement that no paralytic agents are administered when monitoring intraoperative EMG. The absence of neuromuscular blockade can be verified with train-of-four (TOF) testing. Four successive supramaximal electrical stimuli at 2 Hz are applied to a peripheral nerve (e.g., ulnar nerve), and the resultant CMAPs are recorded from a corresponding muscle (e.g., abductor digiti minimi). Four robust CMAPs of equal amplitude should be induced for the EMG recording to be of sufficient sensitivity to detect intraoperative changes reliably. For short-acting agents, the neuromuscular blockade effects can dissipate during surgical exposure. However, if TOF testing indicates that neuromuscular blockade is still present, this can be resolved by administering reversal agents such as sugammadex[18,20-23].
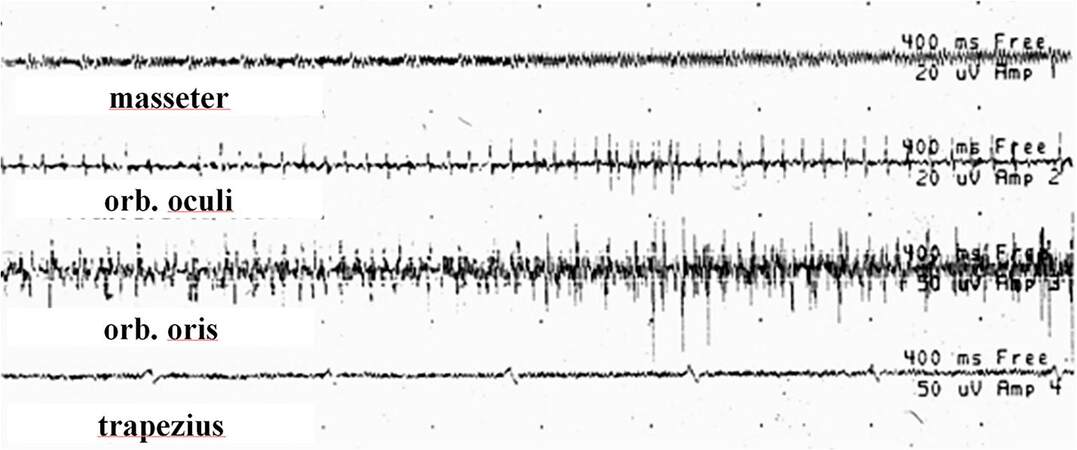
Figure 5. EMG recording demonstrating EMG activity with a superimposed artifact in the masseter, orbicularis oculi, and orbicularis oris muscles during the surgical procedure. Amplitude is 20 µV per division. The sweep speed is 400 msec per division.
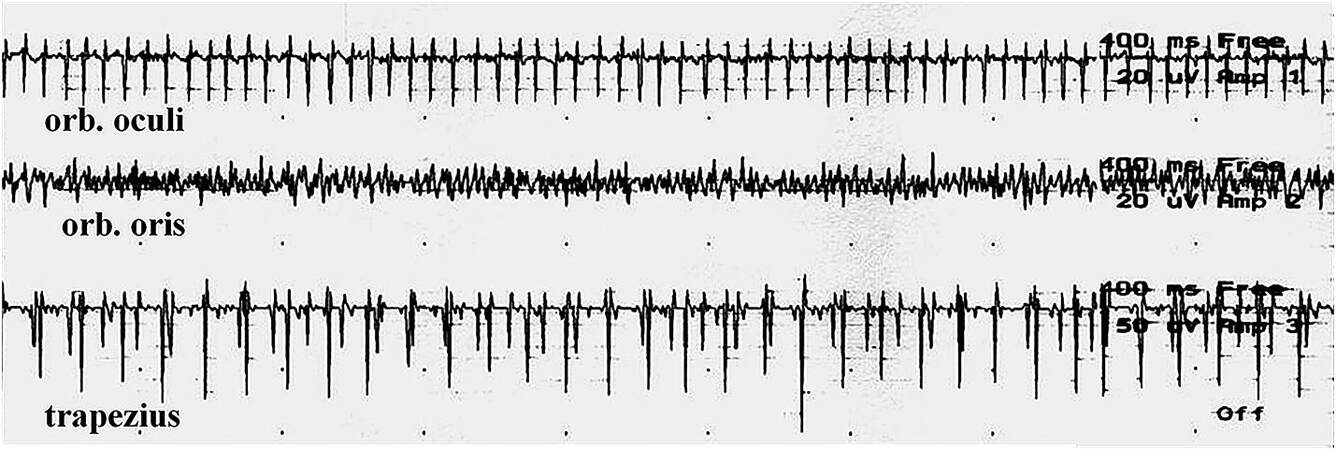
Figure 6. EMG recording demonstrating a train of increased spontaneous EMG activity in the orbicularis oris and trapezius (control) muscles during wearing off of pharmacologic sedation. Amplitude is 20 µV per division (orbicularis oculi and oris) and 50 µV per division (trapezius). The sweep speed is 400 msec per division.
Stimulation-triggered electromyography
Polyphasic CMAPs in innervated muscles can be evoked with motor nerve stimulation, either directly or through intervening tissue, e.g., tumor or bone [Figure 7]. To localize a nerve or nerve root intraoperatively, a sterile handheld monopolar or bipolar stimulating probe is used within the operative field by the surgeon. Typically, the surgeon will use the monopolar stimulator during dissection for stimulation through tissue, such as bone or tumor, to determine if a neural structure, i.e., nerve root, plexus, or peripheral nerve, is nearby. The bipolar stimulator is used when the neural structure is in clear view. An absent CMAP may indicate the absence of motor nerve function, technical problems (poor nerve contact, current shunting, subthreshold stimulation intensity, erroneous recording settings), or pharmacologic neuromuscular blockade. Figure 8 illustrates how the surgeon can sweep the stimulator across the surgical field to locate the peripheral nerve precisely; the stimulation location corresponding to the largest amplitude CMAP would be closest to this neural structure. In addition to providing information regarding localization, the integrity of the nerve or nerve root can be quantified by determining the stimulation threshold and how consistently this is maintained during the procedure. Another way to quantify nerve function is to apply supramaximal stimulation to the motor nerve: the size of the CMAP generated is correlated with the number of functioning axons between the stimulation and recording locations. Stimulation can be performed intermittently during surgery to monitor the integrity of the nerve. Preservation of the response and consistency of the threshold with proximal stimulation provides objective evidence that no significant neural injury has occurred. If a decrement of the CMAP amplitude or increase in the stimulation threshold occurs, the surgeon should consider altering the surgical approach. A significant reduction or complete disappearance of the response often correlates with the severity of the postoperative neural deficit in the short-term and potentially long-term[18,19].
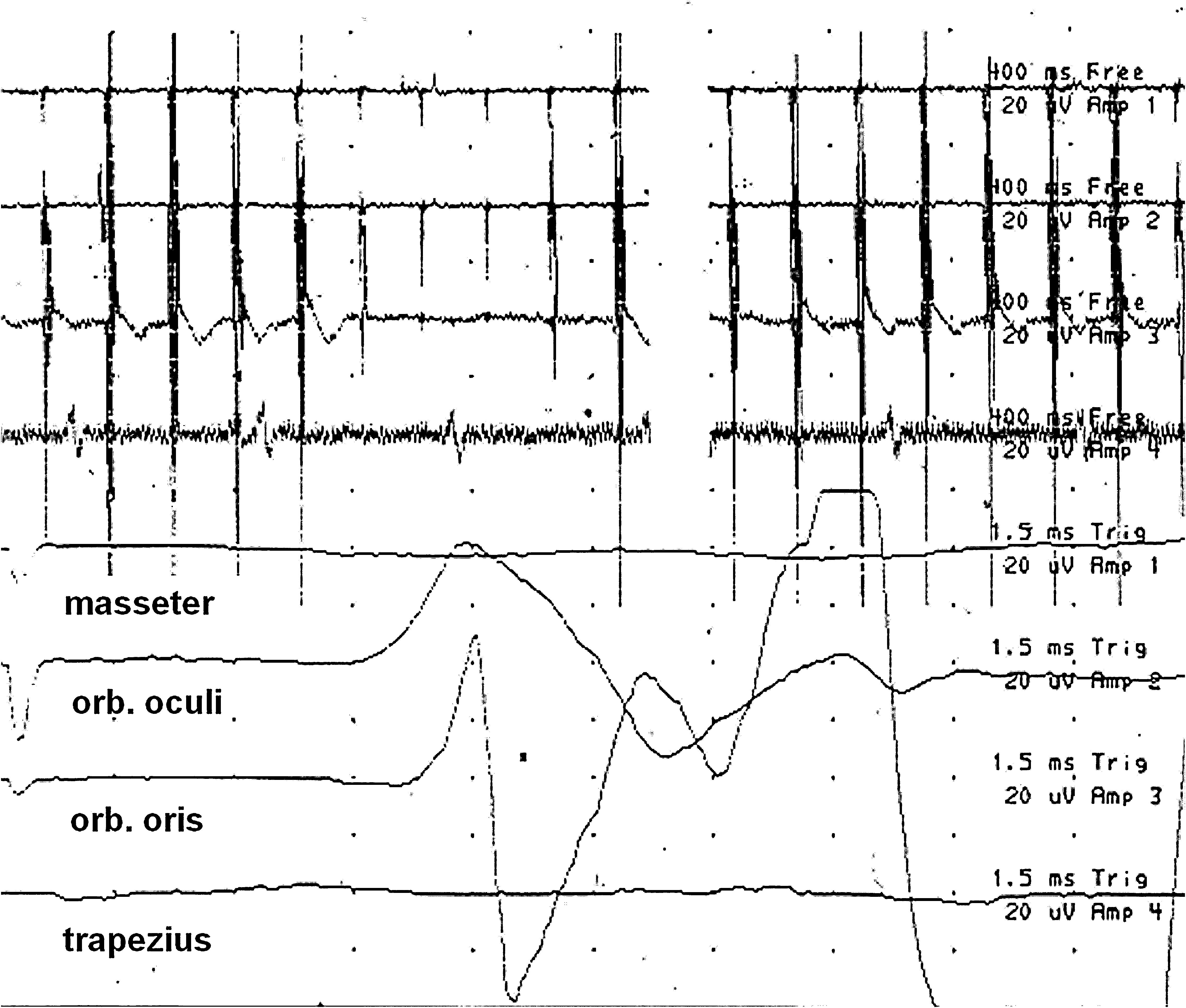
Figure 7. Bottom four tracings - Triggered EMG recordings demonstrating large amplitude, polyphasic compound muscle action potentials (CMAPs) in the orbicularis oculi and oris muscles during facial nerve stimulation within the surgical field. The top four tracings show the corresponding free-running EMGs with superimposed stimulation artifacts. Please note the different time bases for the recordings, i.e., 400 ms/division for the four top tracings and 1.5 ms/division for the bottom four tracings.Amplitude is 20 µV per division.
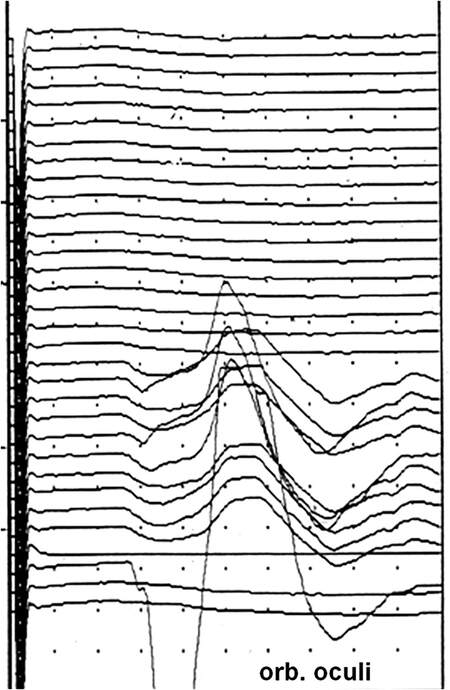
Figure 8. Triggered EMG recordings demonstrating large amplitude, polyphasic compound muscle action potentials (CMAPs) in the orbicularis oculi muscle during facial nerve branch stimulation, sweeping across the surgical field. The largest amplitude response corresponds to the location closest to the facial nerve branch. Amplitude is 20 µV per division.
Bipolar probe utilization results in localized current stimulation that minimizes undesired current spread to adjacent nerves. This can be advantageous when multiple nerves are nearby, such as in the brachial or lumbosacral plexus. However, current shunting between the two electrodes can occur within conductive fluids, e.g., blood in the surgical field, resulting in a false negative motor nerve response. This can be avoided by ensuring that the surgical field is sufficiently dry to avoid this aberrant local conduction. For monopolar stimulation, a reference anode electrode is placed remotely at least several centimeters away from the stimulation site, which results in a broader stimulation field. Monopolar stimulation is not prone to current shunting and can help localize deep neural structures during dissection[18,21-23].
Intraoperative nerve conduction studies
Peripheral and cranial nerves and nerve roots can be electrically stimulated intraoperatively, recording the compound nerve action potential (NAP) at another location along the course of the sensory or motor nerve. Plastic surgeons often use intraoperative stimulation to identify fascicles before nerve transfer. Handheld bipolar electrodes are utilized within the surgical field for stimulation and recording. Such recordings can assess conduction across the nerve’s scarred, injured, or compressed regions. Motor or sensory nerve conduction changes can occur during the procedure as indicated by alterations in the latency, amplitude, or conduction velocity of the NAP [ Figure 1][18,20,22]. An absent NAP may be due to the lack of motor or sensory nerve function or could be related to technical problems (poor nerve contact, current shunting, subthreshold stimulation intensity, erroneous recording settings). As with the CMAP, nerve function can be quantified during surgery using supramaximal sensory or motor nerve stimulation, where the amplitude of the NAP is proportional to the number of functioning axons between the stimulating and recording electrodes[19].
NEUROPHYSIOLOGIC STUDIES IN SPECIFIC DISEASE STATES
Outpatient electrodiagnosis of radiculopathy
NCS are typically normal in the electrodiagnosis of root disorders. However, they are routinely performed to exclude an alternative cause, such as neuropathy or plexopathy. The diagnosis is instead critically dependent on the needle EMG portion of the study, in which distal, proximal, and paraspinal muscles are sampled in the affected extremity. Isolated radiculopathy is based on the presence of neurogenic changes restricted to a specific myotome [Table 1][16,17]. The type of abnormality will depend on the chronicity and severity of the radiculopathy. In the acute period, early axonal loss or conduction block (due to demyelination) reduces the recruitment of motor unit action potentials. After approximately 14 days, active denervation of muscle, as evidenced by increased abnormal spontaneous activity, fibrillation potentials, and positive sharp waves, appears in a proximal to a distal pattern, initially occurring in paraspinal muscles.
It should be noted, however, that denervation potentials require up to 6 weeks to appear in distal muscles; therefore, the sensitivity of electrodiagnosis can be reduced in acute or even subacute radiculopathy. Over the next 3-18 months, unstable, long duration and polyphasic motor units are seen, reflecting early reinnervation and the instability of neuromuscular transmission in nascent nerve fiber branches. As these new fiber connections mature, transmission becomes faster and more stable, resulting in large, reinnervated motor units that also follow a proximal to distal pattern. If this evolution matches well with the reported duration of symptoms, this allows for more diagnostic certainty. The anatomic distribution of these changes is also correlated with clinical exam findings to establish the likely localization. However, an advantage of needle EMG is that even slight axonal loss or motor unit changes will be detected, making it much more sensitive than a clinical exam for subtle weakness. Furthermore, since clinical testing of specific movements can involve multiple muscles, needle EMG is more specific as it isolates a single muscle. Therefore, needle EMG detects more subtle neurogenic changes and offers the advantage of providing information regarding a lesion’s acuity.
Errors in electrodiagnosis of radiculopathies
EMG results that do not correlate well with clinical features or inconsistencies in electromyographic data underscore the need to understand potential sources for error in electrodiagnostic studies. Errors can result in underdiagnosis (type 2 error or false negative), overdiagnosis (type 1 error or false positive), or misdiagnosis [Table 2]. One of the more important limitations is the nerve lesion’s age. Studies performed during the hyperacute or acute period after injury (0-3 weeks) are likely to be normal. Reduced recruitment of MUAPs can be seen if there is sufficient root demyelination to cause conduction block, but this is not sensitive; conduction block requires significant demyelination, and the dual innervation of muscles by more than one nerve root further dilutes the impact. Rarely, very brief trains of positive sharp waves can be seen in the acute period reflecting motor axon loss. Increased abnormal spontaneous activity, fibrillation potentials, and positive sharp waves appear in the acute to the subacute period (> 3 weeks post-injury), and this is the phase in which studies can be most helpful. If fibrillation potentials and positive sharp waves are seen without associated changes in motor unit action potential morphology, the lesion represents active injury and is termed acute, active radiculopathy. The presence of changes in motor unit architecture with increased duration, phases, and amplitude establishes the radiculopathy as chronic; if fibrillation potentials and positive sharp waves are present, it becomes a chronic, active radiculopathy.
Types of errors in electrodiagnostic testing
| Type 1 error (overdiagnosis) | Type 2 error (underdiagnosis) | Error (misdiagnosis) | |
| Radiculopathy | Diagnosis based on polyphasia rather than active denervation | Lack of consideration of lesion duration in the interpretation of NP data | Insufficient number of muscles sampled to allow for confident localization |
| Patchy or fascicular involvement leading to localization errors | Sensory only radiculopathy | Sampling from the same nerve in addition to the same myotome | |
| Anatomic variants (e.g., Martin-Gruber anastomosis, accessory nerve branches, variability in nerve root myotomes) | Demyelinating injury to a nerve that is intermittent or mild | Overlapping innervation of paraspinal and limb muscles: the degree can be unpredictable and vary from one person to another | |
| Poor activation/effort, precluding analysis of motor unit architecture or recruitment | Disk protrusions/ spondylosis that damages only a small number of traversing fibers | Intraspinal DRG resulting in SNAP abnormalities | |
| Inability to test paraspinal muscles due to poor tolerance or concomitant anticoagulation | |||
| Plexopathy | Diagnosis based on polyphasia rather than active denervation | Lack of consideration of lesion duration in the interpretation of NP data | Insufficient number of muscles sampled to allow for confident localization |
| Patchy or fascicular involvement leading to localization errors | Demyelinating injury to the nerve that is intermittent, mild, or proximal | Overlapping innervation of limb muscles: the degree can be unpredictable and vary from one person to another | |
| Anatomic variants (e.g., Martin-Gruber anastomosis, accessory nerve branches, variability in nerve root myotomes) | Plexus lesions that damage a small number of plexus axons | Intraspinal DRG resulting in SNAP abnormalities | |
| Poor activation/effort, precluding analysis of motor unit architecture or recruitment | Inability to test paraspinal muscles due to poor tolerance or concomitant anticoagulation | ||
| Mononeuropathy | Diagnosis based on polyphasia rather than active denervation | Lack of consideration of lesion duration in the interpretation of NP data | Insufficient number of muscles sampled to allow for confident localization |
| Measurement error (extended elbow during UNE study) | Demyelinating injury to a nerve that is intermittent or proximal | ||
| Anatomic variants (e.g., Martin-Gruber anastomosis, accessory nerve branches, variability in nerve root myotomes) | |||
| Poor activation/effort, precluding analysis of motor unit architecture or recruitment | |||
Another source of underdiagnosis occurs if only sensory fibers are involved in the root injury. The nerve root’s ventral (motor) branch is adjacent to the protruding intervertebral disk and, therefore, at greater risk than the dorsal (sensory) branch in the setting of a disk herniation. However, if a root injury affects only the sensory branch, sparing motor axons, needle EMG will be normal even in the setting of radicular pain and sensation change. This is similar to the situation if the root injury is purely demyelinating due to nerve root compression. If there is insufficient conduction block to cause reduced recruitment, the electrodiagnostic studies will also be normal. If there is only intermittent nerve root compression, as can be seen with spinal stenosis where the symptoms are purely positional, fixed EMG changes may not occur[8].
Most errors in electrodiagnostic testing for radiculopathy affect the sensitivity rather than the specificity of testing [Table 2]. However, specificity can be affected if the diagnosis relies solely on MUAP morphology changes (49%-50% specificity) rather than fibrillation potentials and positive sharp waves (71%-89% specificity). Similarly, if too few or inappropriate muscles are sampled during the needle EMG, a false negative study may result. Therefore, the diagnosis should be based on a minimum of two muscles within the same myotome with different peripheral nerve innervation. Needle EMG of paraspinal muscles revealing active denervation potentials confirms the lesion is at or proximal to the nerve root level[24,25]. A caveat to the interpretation of paraspinal muscle abnormalities is overlapping innervation, making it difficult to localize a root lesion based on paraspinal denervation alone[8]. Localization in the extremity can also be limited by sampling error, myotomal overlap or variability in the innervation of muscles. A range like L4-L5 might be the most precise localization electrical studies can offer. Finally, fibrillation potentials and positive sharp waves are nonspecific and may be seen in various conditions, including peripheral neuropathy. This can limit electrodiagnostic specificity in the setting of co-morbid conditions.
Intraoperative electrodiagnostic monitoring during spine surgery
EMG monitoring can be quite helpful for improving the postoperative outcome of spine procedures, particularly in avoiding injury to nerve roots from compression, trauma, stretching, and ischemia. In comparison to peripheral nerves, nerve roots differ in structure, i.e., they lack epineurium, and the endoneurium contains less collagen, which may make them more prone to mechanical injury. Neurotonic EMG discharges correlate closely to intraoperative manipulation of neural structures, particularly spinal nerve roots. Many centers utilize multimodal IONM, including spontaneous and triggered EMG, as well as somatosensory evoked potentials (SSEPs) and transcranial electrical motor evoked potentials, the latter of which are beyond the scope of this review. Although somatosensory evoked potentials are commonly used during spine surgeries, they are less sensitive than EMG for monitoring nerve root function and irritation[18,20].
Intraoperative EMG monitoring nerve roots is greatly useful during spine surgery for several reasons. Free-running, continuous EMG provides real-time feedback regarding the integrity and irritation of nerve roots, which are at significant risk of injury during surgery. The surgical procedure can be altered, limited, or discontinued if excessive bursts or trains of neurotonic discharges in the corresponding monitored muscles occur[26]. Gunnarsson et al. reported that the sensitivity of intraoperative EMG activation to identify patients with new neurologic deficits after thoracolumbar spine surgery was 100%, although the specificity was only 24%[26]. Relatively few patients in their series (6.6%) had postoperative deficits despite the large percentage (78%) with significant neurotonic discharges: therefore, it is likely that the surgeons could avoid permanent neural injury by modifying the surgical strategy. Other IONM modalities can be employed to provide complementary information regarding intraoperative neurophysiologic integrity. Triggered EMG, e.g., pedicle screw stimulation, may also inform the surgeon regarding the proximity of instrumentation to nerve roots[21-23,26,27].
Pedicle screw testing
Intraoperative pedicle screw stimulation is a highly sensitive and specific method for detecting pedicle wall breach into the spinal canal during screw placement. A malpositioned pedicle screw poses a significant risk to the nerve root. Although the secure placement of pedicle screws can be determined by fluoroscopy, the local neuroanatomy cannot be visualized, so triggered EMG provides an essential complementary tool for this purpose. The holes and screws correctly positioned within the pedicle wall are separated from the adjacent nerve roots by a high-impedance cortical bony layer. When a pedicle screw is placed correctly, the surrounding bone impedes electrical conduction to adjacent nerve roots. Therefore, a higher electrical current will be needed to stimulate the local nerve root. During testing, a monopolar electrical stimulator is used to apply a series of increasing currents to the pedicle screws. Electrodes placed into muscles innervated by the respective spinal nerve root levels will record CMAPs synchronized with the stimulation. If a pedicle wall breach occurs, the stimulation threshold is significantly reduced. A stimulation threshold greater than 15-20 mA reliably indicates adequate screw position. A stimulation threshold of less than 10 mA is often associated with significant pedicle cortical perforation by the screw. When less bone separates the screw from the nerve root, the stimulation threshold will be reduced due to lower electrical impedance. This should prompt the surgeon to remove or redirect the screw at that site, particularly if there is concurrent neurotonic EMG activity in a corresponding muscle. Stimulation thresholds between these two current levels usually indicate adequate screw position, although surgical exploration to determine the integrity of the pedicle wall should be considered. False negatives can occur due to various factors, such as the use of muscle relaxants or pre-existing nerve damage, which can result in a higher stimulation threshold or no CMAP response at all. In addition, the region around the pedicle screws should be sufficiently dried to avoid current shunting, which can also result in an erroneously high stimulation threshold. In such cases, if possible, one should first determine the stimulation threshold to evoke a CMAP by directly stimulating the exposed chronically compressed spinal nerve root and then performing pedicle screw testing[18,20,27,28].
Outpatient electrodiagnostic studies in brachial plexopathy
As with the other entities, the electrodiagnosis of plexopathies is based on inference from clinical and electrophysiologic data. Nerve conduction data are an essential aspect in evaluating plexopathies, particularly in distinguishing plexopathy from radiculopathy or polyradiculopathy. This is partly because of the informative nature of sensory nerve studies in plexopathy. As noted above, sensory nerve studies are normal in radiculopathy because the lesion is usually proximal to the dorsal root ganglion, leaving the sensory axon intact. In plexopathies, however, the lesion is distal to the dorsal root ganglion, and the sensory axon degenerates. This allows the electromyographer to test several different sensory nerves, allowing for the localization of a plexus lesion. For instance, alteration of sensation over the lateral aspect of the hand and forearm prompts evaluation of the median sensory response to the thumb, radial sensory response, and lateral antebrachial cutaneous nerves, which localizes to the upper trunk of the plexus. The differential of this distribution of sensory alteration would include mononeuropathies involving any of these single nerves; if more than one is affected, however, a plexopathy is supported. Side-to-side comparison can help detect more subtle axonal injury and support plexopathy versus mononeuropathy. For example, sensory abnormalities over the medial hand and forearm with reduced amplitudes in the ulnar sensory response to the fifth digit, the dorsal ulnar cutaneous response, and the medial antebrachial cutaneous response localize to the lower trunk and medial cord. These are conduction studies that are generally not performed but can be of value in the assessment of plexopathy.
To establish the correct pattern, the subsequent needle EMG study in plexopathy is more extensive than it is for radiculopathy or mononeuropathy. Muscles tested must represent trunks, cords, and individual nerves of the upper extremity. Muscles innervated proximal to the plexus, including cervical paraspinal muscles, serratus anterior (long thoracic nerve), and rhomboids (dorsal scapular nerve), are essential to rule out radiculopathy. Of particular importance in possible surgical management is loss of axonal continuity; this is evidenced by the absence of recruitment in weak muscles (that is, no motor units produced on a voluntary contraction of a muscle). Even in the acute period before the appearance of fibrillation potentials and positive sharp waves, the absence of MUAP recruitment in the setting of good effort should prompt surgical exploration or nerve grafts, considering tendon transfer in the chronic setting.
Errors in the electrodiagnosis of plexopathy
An advantage of the brachial plexus study over radiculopathy is the availability of sensory responses as part of the diagnostic arsenal. This not only helps to distinguish a lesion as localized distal to the dorsal root ganglion (DRG) but also increases the number of responses that can be obtained to detect an abnormality. Similar considerations regarding limitations in plexus studies apply, as have been discussed for radiculopathy studies. As in radiculopathy studies, there exist similar timing considerations, with poor sensitivity during the hyperacute period (< 3 weeks). The injury must either result in axonal loss or conduction block to be detected. An adequate sample of muscles representing trunks, cords, and nerves must be tested to be confident of localization. Brachial plexus lesions can often be patchy to complicate these challenges further, leading to incomplete lesions of specific anatomic branches. There can also be variation in the spinal levels supplying the plexus, leading to an incomplete denervation picture or denervation in unexpected muscles. As discussed above, differentiation of plexus injury from radicular or polyradicular involvement depends on the sensory NCS, the pattern of denervation on needle EMG, and, essentially, denervation in paraspinal muscles. However, errors can occur in these metrics: (i) sensory nerve action potentials can be affected if the DRG is intraspinal or very proximal; (ii) needle study can be insensitive in the absence of active denervation; (iii) fascicular involvement can mislead localization or result in incomplete data; and (iv) paraspinal muscle study may be contraindicated due to anticoagulation or limited by incomplete relaxation. Therefore, plexus studies are often more extensive and difficult to localize.
Intraoperative monitoring of plexopathy
Electrodiagnostic assessment of the brachial plexus can be quite challenging due to the number of neural elements and the intricacies and variability of the neuroanatomy involved, i.e., roots, trunks, divisions, and cords. Given the inherent complexities, pre-operative assessment of the nerves that comprise the brachial plexus can be limited. A thorough understanding of the functional anatomy and neuropathologic changes is crucial, so IONM could be of greater importance for surgery involving this portion of the peripheral neuroaxis, e.g., precisely determining the location(s) and severity of nerve injury, as well as the potential for recovery.
The anatomic focus of the surgery will determine the selection of nerves and muscles to monitor. However, one should be prepared for the possibility that this could change during the procedure, e.g., given the intraoperative findings, the location of dissection can shift from the upper trunk to the lower trunk, necessitating monitoring the median and ulnar nerves, as well as the respective muscles they each innervate[21-23,29].
As with radiculopathy and mononeuropathy, intraoperative neurophysiologic monitoring enables the surgical team to identify components and determine the baseline neurophysiologic status of the brachial plexus, e.g., the severity of the injury and the changes that may occur during surgery. Neurophysiologic monitoring, i.e., EMG and NCS, also help the surgeon to avoid accidental injury to the brachial plexus neural structures, including the spinal nerve roots, trunks, cords, and nerves, during dissection, neurolysis, and tumor resection. Multimodal intraoperative testing, including NAPs and somatosensory and motor-evoked potentials, can be quite helpful for determining whether ventral or dorsal root avulsions and preganglionic injuries have occurred. However, the latter two neurophysiologic modalities are beyond the scope of this review. IONM can also provide an assessment of the potential for spontaneous nerve regeneration. If a NAP or CMAP can be recorded across scarring or a neuroma involving the brachial plexus, local neurolysis might be a more successful surgical intervention rather than resection and nerve grafting. However, if such responses are absent, resection would be necessary. Intraoperative electrical stimulation could identify which nerve fascicles can be utilized for grafts and transfers; the resultant NAP and CMAP responses enable the surgeon to determine which portion of the nerves and branches may be expendable for this purpose. The specific noncritical muscles and potential overlapping innervation can be confirmed using intraoperative stimulation testing with NAP and CMAP recordings.
Given the complex anatomy, IONM for brachial plexus procedures can be tedious and time-consuming. However, it is worth the investment since this technology can provide essential information for achieving favorable postoperative outcomes following tumor resection and post-traumatic nerve repairs and reconstruction.
Outpatient electrodiagnostic studies of mononeuropathy
Electrodiagnostic studies are most useful for localizing the site of injury along the nerve in a mononeuropathy and excluding alternative localizations, such as plexus or nerve root. Ulnar neuropathy at the elbow (UNE) is an illustrative example of the second most common entrapment neuropathy in the upper extremity, after median neuropathy at the wrist. Potential mimics of UNE include ulnar entrapment at the wrist or alternative site, lower brachial plexopathy, or C8-T1 radiculopathy. Electrodiagnostic testing can distinguish between these possibilities. EMG demonstrates denervation and reinnervation in the ulnar-innervated muscles of the hand and forearm without the involvement of median or radial-innervated C8 muscles. The most proximal ulnar-innervated muscle is the flexor carpi ulnaris, often spared due to differential fascicular sparing in compression at the elbow. On NCS, slowed conduction velocity or conduction block at the elbow can be seen in a demyelinating UNE providing localization within the ulnar nerve.
In contrast, for an axonal UNE, it may be more difficult to localize the lesion to the elbow site on NCS. Additional studies could be considered in that setting, such as dorsal ulnar cutaneous sensory NCS, short segment incremental inching studies, mixed and sensory NCS across the elbow, or motor NCS recording from the first dorsal interosseous (FDI) muscle[8]. Some of these electrodiagnostic tests can be time-consuming or technically challenging to perform. Even if these challenges are overcome, it may remain impossible to precisely localize the ulnar neuropathy with electrodiagnostic testing. Neuromuscular ultrasound is emerging as an adjunctive technique in this setting but is outside the scope of this review[30].
In addition to localization value for the surgeon, there is prognostic value provided by outpatient electrodiagnostic studies. For example, the existence of a conduction block across the elbow to the FDI with a normal CMAP amplitude from the abductor digiti minimi is strongly associated with significant or complete recovery[31]. In another series, a reduced FDI CMAP amplitude predicted pre-operative weakness, while isolated slowing across the elbow did not, leading the authors to conclude that CMAP amplitude is a sensitive indicator of axonal loss[32]. In addition to confirming or at least supporting a diagnosis of UNE, the degree of severity can be graded based on electrodiagnostic studies[33]. This can be invaluable in counseling patients about outcomes and consideration of surgical intervention.
Errors in the electrodiagnosis of mononeuropathies
Several potential pitfalls exist in the routine electrodiagnostic evaluation of errors in the electrodiagnosis of mononeuropathies (UNE). In particular, the position of the upper extremity when performing the studies can have a significant effect on measurements used to calculate conduction velocities[34]. The elbow should be kept in a moderate flexion position to provide the greatest correlation between the surface skin measurement and true nerve length[35]. The distance between below and above elbow nerve conduction testing sites should be 10 cm, as shorter segments may amplify errors and longer segments may dilute abnormal findings[17]. The below-elbow stimulation site should be
Intraoperative monitoring of mononeuropathy
IONM can be quite beneficial to patients undergoing a variety of types of peripheral nerve surgeries, including exploration and repair of entrapments, nerve transposition, resection of tumors and cysts, neurolysis and nerve grafts post trauma. For all of these procedures, utilization of free-running and triggered EMG, as well as NCS, can be helpful for locating and identifying peripheral nerves, thereby protecting them from the risks posed by surgical manipulation, and also assessing their neurophysiologic function intraoperatively. The latter can support pre-operative NCS where particularly proximal sites are inaccessible. Whenever it is difficult to differentiate neural from non-neural tissue, free-running and triggered EMG can be beneficial. Stimulation through normal tissue, scar and tumor can also help guide the surgeon during dissection. An expanding mass can often result in significant alterations of the normal anatomical relationships, such as the displacement of nerve fibers over the tumor capsule that needs to be identified by the surgeon. In addition, blunt trauma and compression do not usually result in transection of the peripheral nerve. The extent of recovery, i.e., regeneration through remyelination and regrowth of axons, is related to the degree and type of injury (neuropraxic vs. axonal) to the internal nerve bundle, which is not necessarily evident from gross visual inspection[18]. Such a determination is essential when deciding whether a nerve graft repair or decompression is indicated. The existence and location of functioning nerve can be determined by means of nerve conduction when it is obscured by scarring, being in the closest proximity to where the amplitude of the NAP response is the largest. Peripheral nerves and their branches can also be identified by means of CMAPs generated in specific muscles with stimulation. For example, stimulation of the median and ulnar nerves can elicit motor responses over the thenar eminence, but only stimulation of the ulnar nerve would also activate the hypothenar muscles. Assessment of the integrity of nerve function can also be achieved by means of stimulation within the surgical field. The surgeon can stimulate and record the NAPs or CMAPs along the course of a nerve. If there exists a location where the responses are no longer detected, this could indicate a nonfunctioning portion of the nerve. However, if the NAPs or CMAPs can be elicited throughout the course, including across scar tissue at least several months after injury, this suggests that ongoing reinnervation is occurring. Therefore, it may be best to leave this area alone or to perform neurolysis, rather than a nerve graft. It should be noted that false negative CMAPs and NAPs can occur for reasons that have been stated previously, e.g., current shunting, technical issues, muscle relaxants or ischemia. If tourniquets are utilized during surgery, they should be deflated for up to 30 minutes before performing electrodiagnostic testing distally in the limb[18,29].
CONCLUSION
There is sufficient evidence that EMG and NCS can provide the surgeon with useful information regarding the neurophysiologic status of spinal nerve roots, the brachial and lumbosacral plexuses, and peripheral nerves pre-operatively, as well as during surgery. IONM can help the surgeon to accurately identify neural structures and assess their functionality, as well as guide a safe and successful dissection or resection, nerve repair or placement of spinal instrumentation. Intraoperative EMG and NCS provide an effective method for minimizing intraoperative risks to the nervous system for procedures where nervous tissue is manipulated, thereby improving outcomes in terms of postoperative neurologic function. The advantages offered by pre-operative electrodiagnostic testing are significant and include improved sensitivity/specificity, potentially precise localization, duration of injury, severity and prognostic data. The loss of recruitment of motor unit potentials in the setting of full activation/effort during contraction of a muscle is a compelling indication for nerve grafting, while the demonstration of recruitment allows for a more conservative approach. Inconsistencies in clinical or radiologic data in the pre-operative setting, particularly when considering a high-risk surgical approach, should prompt a re-examination of pre-operative data with the outpatient electromyographer; errors and limitations in the interpretation of data must always be considered. The sources of type 1 error in diagnostic studies include over-reliance on subtle or equivocal electrodiagnostic changes, anatomic variation, patchy or fascicular injury, poor effort during muscle contraction and measurement errors. An insufficient number of sampled muscles, limitations of study due to poor tolerance or concomitant anticoagulation and overlapping innervation can result in misdiagnosis. Similarly, during IONM, errors can result from localized ischemia, excess fluid/blood in the recording field and electrical interference from surgical equipment. That said, the pre-operative assessment and continuous monitoring of the nervous system can allow for more aggressive surgical interventions to be achieved.
It should be emphasized that in the end, the value of pre-operative and intraoperative electrodiagnostic studies depends on a collaborative interaction and effective communication between the neurophysiologist and surgeon such that the neurophysiologic information is incorporated during pre-operative planning and crucial stages of the surgical procedure. Close cooperation among all members of the surgical team is of utmost importance for preventing intraoperative neural compromise and optimizing postoperative outcomes. As surgical techniques progress in advancing the field of surgical management of nerve, plexus and root injuries, studies aimed at correlating pre-operative electrodiagnostic localization and intra-operative electrodiagnostic function with outcomes will facilitate a better understanding of the relevance of some of the errors discussed in this review. This has particular relevance in an appreciation of the redundancy of myotomal innervation and the extent of anomalous innervation. This not only has potential implications for surgical fields but also for nerve and muscle disorders.
DECLARATIONS
Authors’ contributionsMade substantial contributions to conception and writing of the review article: Kobylarz EJ, Randhawa J, Mason S, Lawson VH
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Authors 2023.
REFERENCES
1. Mashour GA, Woodrum DT, Avidan MS. Neurological complications of surgery and anaesthesia. Br J Anaesth 2015;114:194-203.
2. Oh SJ. Clinical electromyography: nerve conduction studies. Available from: https://books.google.com.hk/books/about/Clinical_Electromyography.html?id=KtwFzdPQebUC&redir_esc=y [Last accessed on 9 Jan 2023].
3. Liu EA, Salazar T, Chiu E, et al. Focal peripheral neuropathies observed in patients diagnosed with COVID-19: a case series. Am J Phys Med Rehabil 2022;101:164-9.
4. Robinson LR, Micklesen PJ, Wang L. Optimizing the number of tests for carpal tunnel syndrome. Muscle Nerve 2000;23:1880-2.
5. Bland JD. Do nerve conduction studies predict the outcome of carpal tunnel decompression? Muscle Nerve 2001;24:935-40.
6. Shi Q, MacDermid J, Grewal R, King GJ, Faber K, Miller TA. Predictors of functional outcome change 18 months after anterior ulnar nerve transposition. Arch Phys Med Rehabil 2012;93:307-12.
7. Ney JP, van der Goes DN, Nuwer MR. Does intraoperative neurophysiologic monitoring matter in noncomplex spine surgeries? Neurology 2015;85:2151-8.
8. Preston DC, Shapiro BE. Electromyography and neuromuscular disorders. Available from: https://www.elsevier.com/books/electromyography-and-neuromuscular-disorders/978-0-323-66180-5 [Last accessed on 16 Jan 2023].
9. Ochoa J, Fowler TJ, Gilliatt RW. Changes produced by a pneumatic tourniquet. In: Desmedt J, editor. Pathological conduction in nerve fibers, electromyography of sphincter muscles, automatic analysis of electromyogram with computers. S. Karger AG; 1973. p. 174-80.
10. Vanhoestenberghe A. Chronic nerve root entrapment: compression and degeneration. J Neural Eng 2013;10:011001.
11. Robinson LR, Binhammer P. Role of electrodiagnosis in nerve transfers for focal neuropathies and brachial plexopathies. Muscle Nerve 2022;65:137-46.
13. Daube JR. AAEM minimonograph #11: needle examination in clinical electromyography. Muscle Nerve 1991;14:685-700.
14. Kimura J. Intraoperative monitoring of neural function, handbook of clinical neurophysiology. Available from: https://journals.lww.com/ajpmr/Citation/2003/04000/Electrodiagnosis_in_Diseases_of_Nerve_and_Muscle_.10.aspx [Last accessed on 16 Jan 2023].
15. Brown WF. 7-Electromyography-normal. The physiological and technical basis of electromyography. Elsevier; 1984. p. 287-316.
16. Levin KH, Maggiano HJ, Wilbourn AJ. Cervical radiculopathies: comparison of surgical and EMG localization of single-root lesions. Neurology 1996;46:1022-5.
17. Levin KH. Electrodiagnostic approach to the patient with suspected radiculopathy. Neurol Clin 2002;20:397-421.
20. Holland NR. Intraoperative electromyography during thoracolumbar spinal surgery. Spine (Phila Pa 1976) 1998;23:1915-22.
21. Louis H. A practical approach to neurophysiologic intraoperative monitoring. Neurologist 2008;14:339.
22. Møller AR. Evaluating the benefits of intraoperative neurophysiological monitoring. in: intraoperative neurophysiological monitoring. Available from: https://doi.org/10.1007/978-1-59745-018-8_19 [Last accessed on 16 Jan 2023].
23. Nuwer MR. Intraoperative monitoring of neural function, handbook of clinical neurophysiology. Available from: https://www.amazon.com/Intraoperative-Monitoring-Neural-Function-Neurophysiology/dp/044451824X [Last accessed on 16 Jan 2023].
24. Dillingham TR, Lauder TD, Andary M, et al. Identifying lumbosacral radiculopathies: an optimal electromyographic screen. Am J Phys Med Rehabil 2000;79:496-503.
25. Dillingham TR, Lauder TD, Andary M, et al. Identification of cervical radiculopathies: optimizing the electromyographic screen. Am J Phys Med Rehabil 2001;80:84-91.
26. Gunnarsson T, Krassioukov AV, Sarjeant R, Fehlings MG. Real-time continuous intraoperative electromyographic and somatosensory evoked potential recordings in spinal surgery: correlation of clinical and electrophysiologic findings in a prospective, consecutive series of 213 cases. Spine 2004;29:677-84.
27. Nichols GS, Manafov E. Utility of electromyography for nerve root monitoring during spinal surgery. J Clin Neurophysiol 2012;29:140-8.
28. Kaliya-Perumal AK, Charng JR, Niu CC, et al. Intraoperative electromyographic monitoring to optimize safe lumbar pedicle screw placement - a retrospective analysis. BMC Musculoskelet Disord 2017;18:229.
30. Alrajeh M, Preston DC. Neuromuscular ultrasound in electrically non-localizable ulnar neuropathy. Muscle Nerve 2018;58:655-9.
31. Friedrich JM, Robinson LR. Prognostic indicators from electrodiagnostic studies for ulnar neuropathy at the elbow. Muscle Nerve 2011;43:596-600.
32. Power HA, Sharma K, El-Haj M, Moore AM, Patterson MM, Mackinnon SE. Compound muscle action potential amplitude predicts the severity of cubital tunnel syndrome. J Bone Joint Surg Am 2019;101:730-8.
33. Odusote K, Eisen A. An electrophysiological quantitation of the cubital tunnel syndrome. Can J Neurol Sci 1979;6:403-10.
34. Hsu K, Robinson LR. Effect of shoulder and elbow position on ulnar nerve conduction. Muscle Nerve 2019;60:88-90.
35. Association of electrodiagnostic medicine, american academy of neurology, american academy of physical medicine and rehabilitation. practice parameter for electrodiagnostic studies in ulnar neuropathy at the elbow: summary statement. Muscle Nerve 1999;22:408-11.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Kobylarz EJ, Randhawa J, Mason S, Lawson VH. Pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring: power and pitfalls. Plast Aesthet Res 2023;10:2. http://dx.doi.org/10.20517/2347-9264.2022.38
AMA Style
Kobylarz EJ, Randhawa J, Mason S, Lawson VH. Pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring: power and pitfalls. Plastic and Aesthetic Research. 2023; 10(1): 2. http://dx.doi.org/10.20517/2347-9264.2022.38
Chicago/Turabian Style
Kobylarz, Erik J., Jason Randhawa, Stephen Mason, Victoria H. Lawson. 2023. "Pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring: power and pitfalls" Plastic and Aesthetic Research. 10, no.1: 2. http://dx.doi.org/10.20517/2347-9264.2022.38
ACS Style
Kobylarz, EJ.; Randhawa J.; Mason S.; Lawson VH. Pre-operative electrodiagnostic studies and intraoperative neurophysiologic monitoring: power and pitfalls. Plast. Aesthet. Res. 2023, 10, 2. http://dx.doi.org/10.20517/2347-9264.2022.38
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 9 clicks
Cite This Article 9 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.