Advances in upper limb loss rehabilitation: the role of targeted muscle reinnervation and regenerative peripheral nerve interfaces
Abstract
Upper limb loss results in significant physical and psychological impairment and is a major financial burden for both patients and healthcare services. Current myoelectric prostheses rely on electromyographic (EMG) signals captured using surface electrodes placed directly over antagonistic muscles in the residual stump to drive a single degree of freedom in the prosthetic limb (e.g., hand open and close). In the absence of the appropriate muscle groups, patients rely on activation of biceps/triceps muscles alone (together with a mode switch) to control all degrees of freedom of the prosthesis. This is a non-physiological method of control since it is non-intuitive and contributes poorly to daily function. This leads to the high rate of prosthetic abandonment. Targeted muscle reinnervation (TMR) reroutes the ends of nerves in the amputation stump to nerves innervating “spare” muscles in the amputation stump or chest wall. These then become proxies for the missing muscles in the amputated limb. TMR has revolutionised prosthetic control, especially for high-level amputees (e.g., after shoulder disarticulation), resulting in more intuitive, fluid control of the prosthesis. TMR can also reduce the intensity of symptoms such as neuroma and phantom limb pain. Regenerative peripheral nerve interface (RPNI) is another technique for increasing the number of control signals without the limitations of finding suitable target muscles imposed by TMR. This involves wrapping a block of muscle around the free nerve ending, providing the regenerating axons with a target organ for reinnervation. These RPNIs act as signal amplifiers of the previously severed nerves and their EMG signals can be used to control prosthetic limbs. RPNI can also reduce neuroma and phantom limb pain. In this review article, we discuss the surgical technique of TMR and RPNI and present outcomes from our experience with TMR.
Keywords
INTRODUCTION
Upper limb amputations result in significant functional impairment and psychological morbidity. They are a major financial burden for both patients and health services. There are an estimated 94,000 (major) upper limb amputees in the general population in Europe[1] and 41,000 in the United States[2], with an estimated 1 million upper limb amputees globally.
Allograft reconstruction can restore the loss of an entire upper limb. However, allograft upper limb transplantation surgery is complex, expensive, and requires a high degree of surgical expertise, and finding suitable donors is very difficult. Moreover, not every amputee is a candidate for an allograft. The need for life-long immunosuppression and the poor functional recovery of transplanted upper limbs (especially for amputations above the elbow) are additional major obstacles that are likely to prevent the widespread adoption of this solution for the foreseeable future[3]. Prostheses are therefore likely to be the mainstay of upper limb loss reconstruction after amputation for the foreseeable future.
PROSTHETIC CONTROL
Conventional, body-powered prosthetics are controlled using straps and cables attached to the shoulder girdle. They rely on “trick” movements (such as shrugging of the shoulders) to operate a hook on the end of the prosthesis. Although the setup is simple, reliable, and low cost, the movements are non-intuitive, and the cables and straps are unsightly and difficult to don and doff - especially for women. Myoelectric prosthetics address some of these issues. They are controlled by detecting the millivolt electromyographic (EMG) signals generated by contracting muscles in the residual limb. These signals are captured using skin-surface electrodes placed directly over the muscles. The EMG signals are then used to drive a single degree of freedom (DoF) in the prosthetic limb (e.g., hand open and close)[4]. While myoelectric control systems can improve the fidelity of control compared to body-powered systems, there are a significant number of problems. Most myoelectric prosthetics must rely on only two myoelectric activation points to control the different DoF of the prosthesis. For example, for a transhumeral amputee, EMG signals from biceps/triceps can be used to control elbow movement. For all other movements such as opening/closing the hand and wrist rotation, the patient must rely on the same muscles pairs, and has to access these functions via a mode switch, which the user selects using a trick movement (e.g., co-contraction of the biceps and triceps or double contraction) or by pressing a button on the prosthesis with their normal hand[5]. This then changes the prosthesis mode from elbow to hand movement. The disadvantage of this setup is clear: it is non-intuitive, and actions can only be performed in series, not simultaneously. As a result, the cognitive burden of training to use these devices is significant. Many patients are simply unable to manage the complex learning needed to use these devices, contributing to the high rate of prosthesis abandonment[6-10].
The normal human hand has 22 DoF controlled by 38 muscles[11]. Using an upper limb prosthesis to completely replace these lost functions would require 38 individual actuators, each with a separate activating signal, to fully mirror hand motion. Therefore, to restore control to normal, we need to greatly increase the number of control signals under full voluntary control compared with what is achievable with current technology. Furthermore, the prosthesis needs to restore the sensory feedback of a normal hand.
TARGETED MUSCLE REINNERVATION
Kuiken and Dumanian developed the technique of targeted muscle reinnervation (TMR) more than 15 years ago, with the specific goal of increasing the number of myoelectric control channels available for patients with amputations proximal to the elbow, to strive for more intuitive control of a prosthesis[12,13]. In this technique, the stumps of the peripheral nerves that are no longer supplying their intended upper limb muscles are rerouted to nerves innervating redundant muscles in the upper limb or pectoral girdle. For example, the pectoralis muscles in the chest wall are no longer useful for a patient who has suffered a through-shoulder amputation. Following the successful transfer of the nerve stumps to their new targets, the newly innervated muscles are able to act as proxies for the missing parts of the amputated limb. EMG signals captured using skin-surface electrodes can then be used to control a prosthesis in a fully intuitive manner which was not previously possible.
There are two typical scenarios for TMR in the upper limb:
● The first is a patient with a transhumeral amputation.
● The second is a patient who has sustained a more proximal, glenohumeral amputation or high transhumeral amputation.
PATIENTS WITH A SHOULDER DISARTICULATION
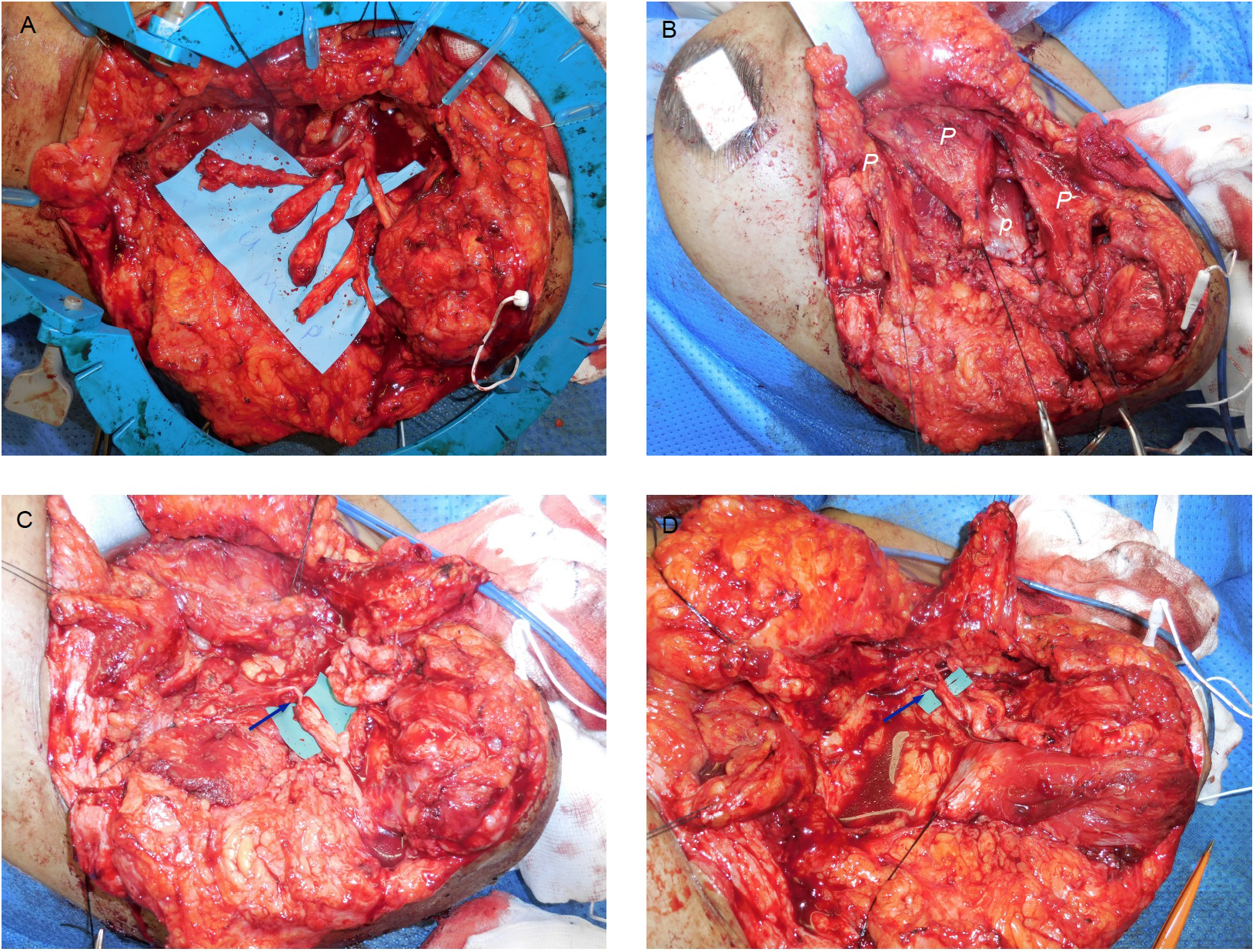
TMR surgery at the glenohumeral/high transhumeral level can be technically demanding because of the presence of heavy scarring around the nerves. Clear identification and careful separation of the nerve stumps from any encasing scar tissue is a necessary pre-requisite for a successful outcome. Once this has been done, our preferred approach is to divide the pectoralis major muscle into three distinct neurovascular territories. This is done by physically splitting the muscle into three separate neuromuscular units: an upper clavicular part and two lower sternal parts [Figure 1], creating three separate muscle targets, each with their own nerve and vascular supply pectoralis minor is often used as another muscle target, but it then needs to be detached from its insertion into the coracoid process and mobilised laterally into a subcutaneous (axillary) position for easier identification with skin-surface electrodes (at a later stage) and to reduce potential crosstalk with the pectoralis major targets. Other potential muscle targets for nerve transfers on the chest wall include the serratus anterior and latissimus dorsi muscles. However, the latter can sometimes take a long time to reinnervate due to the length of its motor branch.
Figure 1. Typical nerve transfers performed in a patient with a shoulder[42] disarticulation/high transhumeral amputation. A: Pre-operative identification of the stumps of the median, ulnar, radial, and musculocutaneous nerves based on the patients’ reports of paraesthesiae in the expected nerve territories - on palpation of their neuromas. B: Nerve transfers performed during the TMR procedure. Division of the pectoralis major muscle into three neuromuscular units which are used as targets for the musculocutaneous, ulnar, and median nerves.
TRANSHUMERAL AMPUTATION
TMR surgery for transhumeral amputees is often easier since the proximal parts of the nerve stumps are (often) not so badly injured. These can then be followed from proximal to distal, making identification of the individual nerve stumps much easier. Moreover, the EMG signals needed to activate elbow movement in a myoelectric prosthesis are preserved by simply leaving the normal motor innervation intact to both the long head of biceps (via the musculocutaneous nerve) and the short or medial head of triceps (via the radial nerve). EMG signals that allow for ‘hand close or open’ are then created by transferring the distal stump of the radial nerve to the motor branch of the lateral head of the triceps and by transferring the stump of the median nerve to the motor branch of the medial head of the biceps[12-14] [Figures 2 and 3]. Once reinnervation is complete, when the patient wants to close the hand of their prosthesis, nerve signals transmitted down the median nerve result in contraction of the medial head of biceps. The EMG signals generated by the reinnervated medial head of biceps are easily distinguished from those generated by the lateral head and result in activation of the appropriate actuator in the prosthesis. The same is true on the extensor side of the residual limb in relation to the reinnervated lateral head of biceps. This creates the opportunity for more intuitive control than before the TMR surgery. If the brachialis muscle is still present, then it can also be pressed into service as a target for the ulnar nerve stump, providing more myoelectric activation points in the residual limb. Interestingly, brachialis itself can often be separated into two distinct muscle territories, each with its own nerve supply (in a similar way to pectoralis major). This creates the opportunity for even more targets, if suitable donor nerves become available during the initial exploration.
Figure 2. Typical nerve transfers performed in patients with a transhumeral[42] amputation. A: Pre-operative identification of the stumps of the radial, median and ulnar nerves based on the patients’ reports of paraesthesiae in the expected nerve territories - on palpation of their neuromas. B: Nerve transfers performed during the TMR procedure. From a neuromuscular perspective, when present, the brachialis can be separated into two parts (medial and lateral), providing two additional targets for a nerve transfer.
Figure 3. Example of TMR to a left transhumeral amputee. A: Dissection of the short [bic (s)] and long [bic (l)] heads of biceps, brach: brachialis. Vessel loop around motor branch to medial head of brachialis. B: TMR transfers as follows: Radial nerve to lateral head of brachialis, ulnar nerve to medial head of brachialis (ulnar nerve hidden behind fat), median nerve reflected back and co-apted proximally to motor branch to medial head of biceps. Triceps left intact.
SURGICAL PEARLS AND PITFALLS
TMR should be considered in any patient who has suffered from a major upper limb amputation and wishes to improve their ability to achieve better control over a myoelectric prosthesis. However, another important primary indication for TMR surgery is to control symptoms related to neuroma (NP) and/or phantom limb pain (PLP) (see relevant section below). Pre-operatively, patients must be carefully assessed. There are several important parts to the assessment:
(1) The level of the amputation. This will determine the (likely) muscle targets in the residual limb and the number of nerve stumps that will act as donors for those targets [Figures 1-4].
Figure 4. Example of TMR to a left glenohumeral amputee. A: Dissection of the terminal branches of brachial plexus with identification of ulnar, median, radial and musculocutaneous nerves. B: Pectoralis major muscle (P) is split into three neurovascular territories. Pectoralis minor (P) can be seen. C and D: TMR transfers indicated with arrows; radial nerve to pectoralis minor and musculocutaneous nerve to upper third of pectoralis major. The median and ulnar nerves were subsequently transferred to the lower and middle pectoralis major muscles, respectively.
(2) The nature of the original injury. Brachial plexus traction/avulsion injuries often result in a dense, flail upper limb for which the standard treatment is an amputation at either the transhumeral level or (occasionally) at a more proximal level. Unfortunately, these patients often have few or no suitable donor nerves and few or no suitable targets for a TMR procedure. In contrast, guillotine type injuries leave all the proximal structures intact, making a TMR procedure a more realistic possibility, although this is only likely when the amputation is carried out as a truly elective procedure (e.g., for cancer). More often, the injury is mixed with some degree of traction (at the time of the initial injury), which is then revised by a trauma surgeon on admission to create a guillotine amputation. In such cases, there may be few suitable target muscles in the residual limb, even if the donor nerves may be present more proximally. These nerves may also exhibit a Sunderland-6 type of injury with intervening segments of scar mixed together with healthy axons. These donor nerves regenerate poorly but can still work usefully in the context of TMR surgery. In such cases, with few suitable muscle targets, a free vascularized muscle transfer can be employed to import fresh muscle targets into the residual limb. For example, the serratus anterior muscle has a segmental motor innervation, and each muscle slip can be reinnervated with a separate donor nerve. Similarly, the gracilis and rectus abdominis muscles may also be used as these muscles also have a segmental motor nerve supply which allows multiple donor nerve stumps to be used to reinnervate different parts of each muscle[15-17].
(3) The aims and goals of the patient. Most patients who undergo TMR surgery will not continue with a complex multiaxial prosthesis with myoelectric control. There are many reasons for this, including problems with funding for such devices, lack of suitable training and the heavy mechanical and engineering support needed to make these devices a useful addition to daily life. For the majority of upper limb amputees, being pain-free is a major goal. In such a situation, concerns about crosstalk from adjacent neuromuscular territories become irrelevant and the need to carry out complicated maneuvers to move muscle units into areas where the prosthetists can detect the EMG signals also becomes irrelevant. This makes some aspects of the surgery much simpler.
As with all nerve transfer surgery, the donor nerves should be co-apted as close as possible to the hilum of the target motor nerve so that the reinnervation distances are as short as possible. We make extensive use of intraoperative nerve stimulation and try to avoid the use of local anesthetics in the area around the muscle targets. We also try to avoid using tourniquets since hypoxia can quickly lead to muscle fatigue, making interpretation of nerve stimulator results difficult. When there is an intention to use a multiaxial prosthesis after surgery, special attention should be paid to placing the muscle units in such a way (under the skin) to reduce electrical crosstalk (signal contamination from adjacent muscles). This can be done by interposing adipofascial flaps between the muscle units to help isolate the different EMG signals[18]. The overlying skin should also be thinned to reduce conduction distance between the skin surface and the underlying muscles in order to better detect surface EMG signals and increase the signal-to-noise ratio.
One intriguing observation made in TMR patients relates to the recovery of sensation from the missing hand in the skin overlying the reinnervated muscles[12]. The mechanism for this phenomenon is unclear since it seems unlikely that the sensory parts of the (mixed) donor nerves can physically grow through the target muscle and up into the overlying skin to make connections with the sensory end-organs. Nevertheless, this observation has led to the development of a technique called targeted sensory reinnervation (TSR)[19]. For TSR, a cutaneous branch supplying sensation to the skin of the chest wall is co-apted in an end-to-side fashion with either the median or ulnar nerve. As with TMR, the recovery of sensation is not completely normal, with touch often perceived more as paraesthesiae arising in parts of the absent limb. However, the degree of sensory recovery is more intense than in patients who undergo TMR alone. Despite this, functional MRI studies have shown that sensory cortical remapping does occur following TSR surgery[20]. Therefore, this observation creates the very real possibility that true sensory feedback from the prosthetic limb may become possible in the future - with the development of the necessary hardware.
The main limitations of TMR include:
(a) The (often) limited availability of suitable target muscles or donor nerves in the residual limb (especially after brachial plexus injuries). As a result, it is sometimes difficult to create more than two or three additional myoelectric EMG sites for control of a prosthetic limb. This is a long way from the 38 muscles and 22 DoF present in a normal hand.
(b) The nature of the soft tissues overlying the intended target muscles. Heavily scarred tissue makes any dissection more difficult, but this is not an absolute contraindication to TMR surgery.
POSTOPERATIVE REHABILITATION
Once TMR surgery has been performed, return of voluntary control over the reinnervated muscles may be evident as quickly as 8 weeks later but can take up to 6 months or more to be complete, depending on the reinnervation distances[21]. Although training of the reinnervated muscles is helpful from the perspective of strengthening the muscles to generate EMG signals for prosthetic control, one of the benefits of TMR is that the reinnervation process occurs regardless of any specific training. Therefore, patients who undergo TMR can often learn to use a multiaxial prosthetic limb (especially a limb equipped with a pattern-recognition control system) within minutes, even without any specific training[22].
To make full use of the new sources of EMG signals, patients who have undergone TMR surgery are fitted with an array of surface electrodes which are then used by their prosthetists to “map out” the muscle areas which are responsible for specific upper limb functions. Equipped with such a map, a prosthetist is then able to adjust the end-device to respond in a way that allows the patient to regain intuitive and simultaneous elbow and hand control[12,17,23] (see Supplementary Video of TMR patient with prosthetist). Amputees who do not have TMR surgery are limited to a prosthesis with perhaps 6 degrees of freedom using just 1 or 2 myoelectric activation sites. Such control cannot be intuitive and individual movements must occur sequentially. TMR surgery provides upwards of 6-9 inputs, all under intuitive and simultaneous control.
PHANTOM LIMB AND NEUROMA PAIN
Symptomatic neuromas and phantom limb pain (PLP) affect up to 80% and 85% of extremity amputations, respectively. Although these symptoms may only become disabling in about 30% of amputees, they can have a major negative impact on quality of life and prosthetic use[24]. The traditional surgical treatment of neuroma pain relies on excision of the neuroma followed by burying of the resulting nerve stump within muscle or bone. Alternatively, the two ends of the nerve are co-apted together [Figure 5]. Although this surgery usually provides immediate relief from the symptoms of NP, recurrence rates can be as high as 60%[25].
Figure 5. Three different (conventional) techniques for dealing with a neuroma[43].
Fundamentally, these techniques do not prevent the recurrence of a neuroma, and they simply provide additional cushioning for the nerve stump and the new neuroma that forms at the end of the nerve stump, thereby reducing fewer symptoms. A more physiological approach is to supply nerves with a target organ - ideally by repairing the nerve and joining it to its original intended target. This provides neurotrophic feedback to the free axons, which helps to decrease the sensitivity of the nerve ends and down-regulates regrowth[26]. This is what is believed to happen with TMR surgery, where the nerves are given “somewhere to go and something to do” rather than simply allowing them to re-form a new and symptomatic neuroma. Although the exact mechanism for the reduction in symptoms is not fully understood, the reduction in PLP after TMR surgery is particularly thought to arise because the target muscles provide feedback from an “end organ” to parts of the cortex which still perceive the amputated part[27].
We agree strongly with the observations made by Dumanian et al. who have noted significant improvements in nerve-related pain amongst the upper limb amputees they treated with TMR surgery. Most patients report an improvement in their NP and PLP after TMR surgery[27,28]. In our experience of 10 consecutive patients undergoing TMR primarily for NP and PLP after upper limb amputation, all patients experienced complete relief from their NP at 12 months after surgery. With regards to PLP, all patients also experienced a slight increase in PLP at 3 months but then noted significant reductions by 12 months after their surgery[29]. This contrasts with our experience of TMR surgery in lower limb patients, who experience a gradual reduction in PLP over time but without the same dramatic reductions in symptoms of both NP and PLP. We have speculated on the reasons for this difference which may be due to the more complete loss of feedback to the central nervous system after upper limb amputation and the more extensive nature of the nerve transfers we performed during upper limb TMR surgery. Interestingly, the temporary worsening of PLP after TMR surgery in the upper limb is consistent with the experiences described by Dumanian et al.[14]. Importantly, our results confirm the outcomes of previous studies suggesting that TMR surgery is an effective technique for providing durable, reproducible and predictable relief from NP and PLP after upper limb amputation[27,30-32].
Regenerative peripheral nerve interface
One of the limitations of TMR surgery is the (sometimes complete) absence of suitable muscle targets. Therefore, Cederna has taken a different approach to Dumanian. Instead of relying on the use of innervated and fully vascularised muscles as a target for any transected nerves, he places the nerve stumps into the centre of a devascularised block of muscle. The muscle grafts are carefully wrapped around the end of the nerve after the neuroma has been excised [Figure 6]. Since these muscle grafts are small, they survive through the same process by which any graft survives in the body - initially by serum imbibition, then by revascularisation from the surrounding tissue bed and the vasa nervorum of the nerve itself[33,34]. These muscle grafts then provide the regenerating axons with a target organ for reinnervation in exactly the same way that TMR does. Therefore, both TMR and RPNI share a common principle of using muscle as a target, and both muscles need to be completely “disconnected” (i.e., denervated) from their native nerve supply for the donor axons to regenerate, as they are unable to “outcompete” muscles with an existing innervation[35].
Figure 6. Schematic of RPNI: a nerve end with a neuroma is treated by excision of the neuroma and wrapping the free end with skeletal muscle[43].
As with Dumanian, Cederna initially developed RPNI with the primary purpose of increasing the number of control signals available for improved upper limb prosthetic control. The long-term intention of his research in this area is to place a biocompatible myoelectrode into the muscle-nerve units that have been fashioned to allow later detection of EMG signals. The composite of donor nerve, muscle graft and electrode then provides the EMG signals that can be used to control an artificial limb.
Used in this way, Regenerative peripheral nerve interface (RPNI) technology has the potential to massively increase the number of EMG signals available to control a prosthetic limb. Instead of using a single (named) donor nerve attached to just one (named) target muscle (e.g., median nerve stump co-apted to motor nerve of medial head of biceps), the donor nerve can be split into multiple fascicles. Each fascicle can then be buried into a muscle graft to create a single RPNI unit, each generating its own unique EMG signal. The data generated by each RPNI unit can be extracted from the residual limb using the implanted muscle electrodes and these signals can then be transmitted directly to a prosthesis using a wireless or hard-wired system. Because the number of potential signals is limited only by the number of fascicles that can be dissected free from the donor nerve, this approach has the potential to provide a (potentially) infinite number of EMG signals. It is definitely possible for RPNI to create the large number of signals required to reproduce and control fine hand and wrist movements - something that is currently very difficult to achieve after TMR surgery - even using pattern recognition systems. However, even if this approach becomes technically possible, current forms of prosthetic engineering do not have the means for processing so many EMG signals and transforming them into the fine movements we attribute to a normal upper limb.
Currently, in vivo studies in animals have demonstrated long-term EMG signal reliability and selectivity of RPNI units and their associated muscle electrodes[34,36]. Cederna’s group has successfully implanted RPNI-electrode systems in four human subjects with upper limb loss for intuitive prosthetic control. The signals have been shown to be stable over a three-year period, despite the signals being transmitted via transcutaneous wires, and subjects were able to use these signals to reliably and intuitively control their prosthetic limbs[37]. The main challenge remains how to transmit these EMG signals across the skin barrier in a stable and durable manner using wireless telemetry, and to date, no fully implantable system exists with wireless signal transmission.
The RPNI concept has also been extended to provide a solution for sensory feedback. Termed sensory RPNI, a sensory nerve can be used to neurotise a muscle unit in the same manner as for motor RPNI. An electrode is placed onto the surface of the muscle, and this is used to stimulate contraction of the muscle. This in turn causes depolarization of the afferent nerve to provide sensory feedback. Electrophysiological testing at 3 months in a rat model has confirmed that sensory RPNI produced reliable signals with minimal cellular inflammatory responses on histological analysis[38].
RPNI has also been successfully used to treat NP in patients with digital neuromas[39] and has been used prophylactically to prevent postamputation pain[40]. Data has also supported its use for PLP, although patient numbers remain small[41] and it is not as well studied as TMR. In our own unit, we use RPNI routinely for the treatment of NP in patients who do not havesuitable TMR target muscles [Figure 7].
Figure 7. Examples of the use of RPNI both to prevent and treat neuroma-related pain. Upper images of patient undergoing elective amputation of his right index finger. A: Dorsal and volar digital nerve stumps. B: Muscle grafts from remnants of intrinsic muscles used to create RPNI units, which are buried deep in the adjacent intrinsic muscles. At 3 years postoperatively, the patient had no painful neuromas. Lower images of patient with painful neuroma of the middle finger. C: neuroma (indicated by arrow) prior to excision. D: Excised neuroma prior to RPNI of the free end of the nerve. The patient had no further neuroma-related symptoms at 12 months follow-up.
RPNI is less invasive, easier to perform and easier to revise. If the data on the control of NP and PLP become available in the future, it may well become more widely applicable than TMR.
CONCLUSION
Both TMR and RPNI are new surgical techniques that (although they differ significantly in their surgical execution) share significant common ground in terms of their fundamental mode of action and their intended function. Both have been shown to significantly reduce the incidence of nerve-related pain associated with amputations. Both have shown great promise in terms of the way in which they will allow amputees to control and/or sense their prosthetic limbs in the future. We anticipate that both procedures will become a routine part of amputee reconstruction in the future.
DECLARATIONS
Authors’ contributionsMade substantial contributions to conception and design of the study and performed data analysis and interpretation: Al-Ajam Y, Kang N, Woollard A
Performed data acquisition and provided administrative, technical, and material support: Al-Ajam Y, Kang N, Woollard A
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationWritten informed consent for publication of patient images was obtained. All images used are the authors’ original pictures and images.
Copyright© The Author(s) 2022.
Supplementary MaterialsREFERENCES
1. Micera S, Carpaneto J, Raspopovic S. Control of hand prostheses using peripheral information. IEEE Rev Biomed Eng 2010;3:48-68.
2. Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil 2008;89:422-9.
3. Salminger S, Roche AD, Sturma A, Mayer JA, Aszmann OC. Hand transplantation versus hand prosthetics: pros and cons. Curr Surg Rep 2016;4:8.
4. Dalley SA, Varol HA, Goldfarb M. Multigrasp myoelectric control for a transradial prosthesis. IEEE Int Conf Rehabil Robot 2011;2011:5975479.
6. Biddiss EA, Chau TT. Upper limb prosthesis use and abandonment: a survey of the last 25 years. Prosthet Orthot Int 2007;31:236-57.
7. Peerdeman B, Boere D, Witteveen H, et al. Myoelectric forearm prostheses: state of the art from a user-centered perspective. J Rehabil Res Dev 2011;48:719-37.
8. Atkins DJ, Heard DCY, Donovan WH. Epidemiologic overview of individuals with upper-limb loss and their reported research priorities. JPO J Prosthet Orthot. 1996 Winter;8:2. Available from: https://journals.lww.com/jpojournal/abstract/1996/00810/epidemiologic_overview_of_individuals_with.3.aspx [Last accessed on 26 Dec 2022].
9. Nazarpour K, Cipriani C, Farina D, Kuiken T. Guest editorial: advances in control of multi-functional powered upper-limb prostheses. IEEE Trans Neural Syst Rehabil Eng 2014;22:711-5.
10. Farina D, Aszmann O. Bionic limbs: clinical reality and academic promises. Sci Transl Med 2014;6:257ps12.
11. Zecca M, Micera S, Carrozza MC, Dario P. Control of multifunctional prosthetic hands by processing the electromyographic signal. Crit Rev Biomed Eng 2002;30:459-85.
12. Kuiken TA, Miller LA, Lipschutz RD, et al. Targeted reinnervation for enhanced prosthetic arm function in a woman with a proximal amputation: a case study. Lancet 2007;369:371-80.
13. Kuiken TA, Li G, Lock BA, et al. Targeted muscle reinnervation for real-time myoelectric control of multifunction artificial arms. JAMA 2009;301:619-28.
14. Kuiken, Todd, Schultz Feuser, Aimee, Barlow, Ann K. Targeted muscle reinnervation a neural interface for artificial limbs. 1st ed. CRC Press; 2013. Available from: https://www.taylorfrancis.com/books/edit/10.1201/b15079/targeted-muscle-reinnervation-aimee-schultz-feuser-todd-kuiken-ann-barlow [Last accessed on 26 Dec 2022].
15. Gart MS, Souza JM, Dumanian GA. Targeted muscle reinnervation in the upper extremity amputee: a technical roadmap. J Hand Surg Am 2015;40:1877-88.
16. Bueno RA Jr, French B, Cooney D, Neumeister MW. Targeted muscle reinnervation of a muscle-free flap for improved prosthetic control in a shoulder amputee: case report. J Hand Surg Am 2011;36:890-3.
17. Cheesborough JE, Smith LH, Kuiken TA, Dumanian GA. Targeted muscle reinnervation and advanced prosthetic arms. Semin Plast Surg 2015;29:62-72.
18. Kuiken TA, Lowery MM, Stoykov NS. The effect of subcutaneous fat on myoelectric signal amplitude and cross-talk. Prosthet Orthot Int 2003;27:48-54.
19. Kuiken TA, Marasco PD, Lock BA, Harden RN, Dewald JP. Redirection of cutaneous sensation from the hand to the chest skin of human amputees with targeted reinnervation. Proc Natl Acad Sci USA 2007;104:20061-6.
20. Yao J, Chen A, Kuiken T, Carmona C, Dewald J. Sensory cortical re-mapping following upper-limb amputation and subsequent targeted reinnervation: a case report. Neuroimage Clin 2015;8:329-36.
21. Lancashire HT, Al Ajam Y, Dowling RP, Pendegrass CJ, Blunn GW. Hard-wired epimysial recordings from normal and reinnervated muscle using a bone-anchored device. Plast Reconstr Surg Glob Open 2019;7:e2391.
22. Hargrove LJ, Miller LA, Turner K, Kuiken TA. Myoelectric pattern recognition outperforms direct control for transhumeral amputees with targeted muscle reinnervation: a randomized clinical trial. Sci Rep 2017;7:13840.
23. O'Shaughnessy KD, Dumanian GA, Lipschutz RD, Miller LA, Stubblefield K, Kuiken TA. Targeted reinnervation to improve prosthesis control in transhumeral amputees. A report of three cases. J Bone Joint Surg Am 2008;90:393-400.
24. Hsu E, Cohen SP. Postamputation pain: epidemiology, mechanisms, and treatment. J Pain Res 2013;6:121-36.
25. Pet MA, Ko JH, Friedly JL, Smith DG. Traction neurectomy for treatment of painful residual limb neuroma in lower extremity amputees. J Orthop Trauma 2015;29:e321-5.
26. Kang N, Woollard A, Konczalik W. New surgical options to improve the quality of life of amputees. Surg Oxf 2019;37:276-87.
27. Souza JM, Cheesborough JE, Ko JH, Cho MS, Kuiken TA, Dumanian GA. Targeted muscle reinnervation: a novel approach to postamputation neuroma pain. Clin Orthop Relat Res 2014;472:2984-90.
28. Kim PS, Ko J, O’Shaughnessy KK, Kuiken TA, Dumanian GA. Novel model for end-neuroma formation in the amputated rabbit forelimb. J Brachial Plex Peripher Nerve Inj 2010;5:6.
29. Kang NV, Woollard A, Michno DA, Al-Ajam Y, Tan J, Hansen E. A consecutive series of targeted muscle reinnervation (TMR) cases for relief of neuroma and phantom limb pain: UK perspective. J Plast Reconstr Aesthet Surg 2022;75:960-9.
30. Dumanian GA, Potter BK, Mioton LM, et al. Targeted muscle reinnervation treats neuroma and phantom pain in major limb amputees: a randomized clinical trial. Ann Surg 2019;270:238-46.
31. Bowen JB, Wee CE, Kalik J, Valerio IL. Targeted muscle reinnervation to improve pain, prosthetic tolerance, and bioprosthetic outcomes in the amputee. Adv Wound Care (New Rochelle) 2017;6:261-7.
32. Alexander JH, Jordan SW, West JM, et al. Targeted muscle reinnervation in oncologic amputees: early experience of a novel institutional protocol. J Surg Oncol 2019;120:348-58.
33. Urbanchek MG, Wei B, Baghmanli Z, Sugg K, Cederna PS. Long-term stability of regenerative peripheral nerve interfaces (RPNI). Plast Reconstr Surg 2011;128:88-9.
34. Kung TA, Langhals NB, Martin DC, Johnson PJ, Cederna PS, Urbanchek MG. Regenerative peripheral nerve interface viability and signal transduction with an implanted electrode. Plast Reconstr Surg 2014;133:1380-94.
35. Thompson W, Jansen J. The extent of sprouting of remaining motor units in partly denervated immature and adult rat soleus muscle. Neuroscience 1977;2:523-35.
36. Irwin ZT, Schroeder KE, Vu PP, et al. Chronic recording of hand prosthesis control signals via a regenerative peripheral nerve interface in a rhesus macaque. J Neural Eng 2016;13:046007.
37. Vu PP, Vaskov AK, Irwin ZT, et al. A regenerative peripheral nerve interface allows real-time control of an artificial hand in upper limb amputees. Sci Transl Med 2020:12.
38. Larson JV, Urbanchek MG, Moon JD, et al. Abstract 17: prototype sensory regenerative peripheral nerve interface for artificial limb somatosensory feedback. Plast Reconstr Surg 2014;133:26-7.
39. Hooper RC, Cederna PS, Brown DL, et al. Regenerative peripheral nerve interfaces for the management of symptomatic hand and digital neuromas. Plast Reconstr Surg Glob Open 2020;8:e2792.
40. Kubiak CA, Adidharma W, Kung TA, Kemp SWP, Cederna PS, Vemuri C. “Decreasing postamputation pain with the regenerative peripheral nerve interface (RPNI)”. Ann Vasc Surg 2022;79:421-6.
41. Kubiak CA, Kemp SWP, Cederna PS, Kung TA. Prophylactic regenerative peripheral nerve interfaces to prevent postamputation pain. Plast Reconstr Surg 2019;144:421e-30e.
42. Kang NV, Woollard A, Michno DA, Al-Ajam Y, Tan J, Hansen E. A consecutive series of targeted muscle reinnervation (TMR) cases for relief of neuroma and phantom limb pain: UK perspective. J Plast Reconstr Aesthet Surg 2022;75:960-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Al-Ajam Y, Woollard A, Kang N. Advances in upper limb loss rehabilitation: the role of targeted muscle reinnervation and regenerative peripheral nerve interfaces. Plast Aesthet Res 2022;9:63. http://dx.doi.org/10.20517/2347-9264.2022.24
AMA Style
Al-Ajam Y, Woollard A, Kang N. Advances in upper limb loss rehabilitation: the role of targeted muscle reinnervation and regenerative peripheral nerve interfaces. Plastic and Aesthetic Research. 2022; 9: 63. http://dx.doi.org/10.20517/2347-9264.2022.24
Chicago/Turabian Style
Al-Ajam, Yazan, Alexander Woollard, Norbert Kang. 2022. "Advances in upper limb loss rehabilitation: the role of targeted muscle reinnervation and regenerative peripheral nerve interfaces" Plastic and Aesthetic Research. 9: 63. http://dx.doi.org/10.20517/2347-9264.2022.24
ACS Style
Al-Ajam, Y.; Woollard A.; Kang N. Advances in upper limb loss rehabilitation: the role of targeted muscle reinnervation and regenerative peripheral nerve interfaces. Plast. Aesthet. Res. 2022, 9, 63. http://dx.doi.org/10.20517/2347-9264.2022.24
About This Article
Copyright
Data & Comments
Data
 Cite This Article 14 clicks
Cite This Article 14 clicks

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.