Study of biological markers in skin quality treatment by subcutaneous injection of a stabilized composition of 26 mg/mL of high molecular weight HA
Abstract
Aim: Hyaluronic acid (HA) injectables have gained rapid acceptance for the treatment of skin rejuvenation. A novel HA/sorbitol composition intended for skin quality improvement containing 2.6% of high molecular weight HA stabilized by sorbitol was recently designed to be injected subcutaneously. The aim of this study was to assess the expression of biological markers of skin quality after administration of the composition.
Methods: The HA/sorbitol composition was evaluated after injection into the superficial adipose tissue with ex vivo cultured human skin explants versus a product comparator to study the general morphology of the skin tissues and the expression of HA, elastin, collagen type I, collagen type III, and fibrillin-1 in the dermal layer.
Results: The results demonstrate that the HA/sorbitol composition is able to boost the production of HA, elastin, collagen type I, collagen type III, and fibrillin-1 in the dermis while providing a proper quality of skin morphology.
Conclusion: The HA/sorbitol composition improved biological markers of skin quality in the dermis after product injection into the superficial adipose tissue. This novel composition can be considered as an attractive solution to treat skin aging by injecting a specific HA/sorbitol formulation to strategically target the subcutaneous tissue to improve the quality of the different layers of the skin.
Keywords
INTRODUCTION
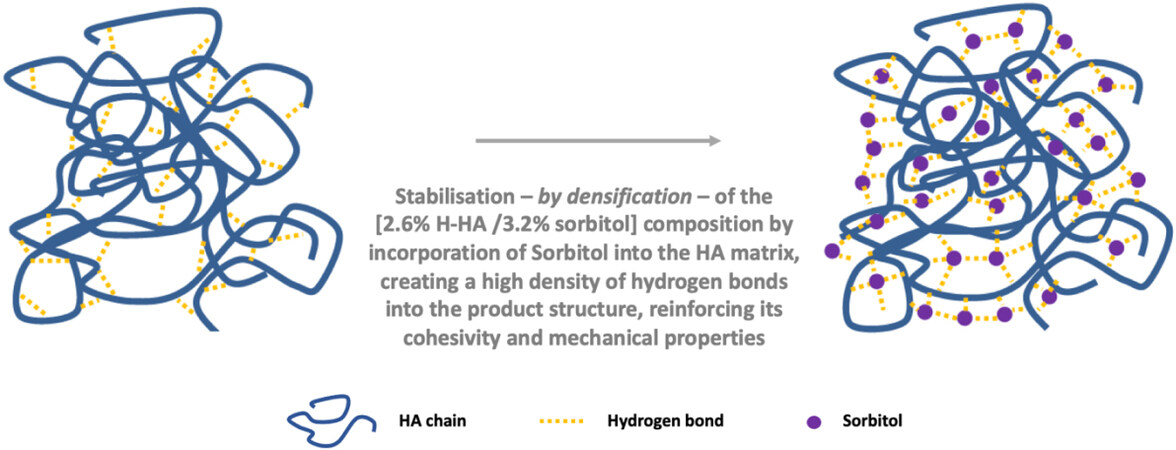
Skin quality represents an essential component of human attractiveness with a strong influence on the perception of age and health[1]. The skin aging process is determined by intrinsic and extrinsic factors such as genetics, hormones, sun exposure, dietary habits, and pollution[2]. Skin tissues are responsible for structural integrity and elasticity. Aging is the cause of major changes in the different skin layers, including the loss of skin elasticity, atrophy of the dermis, and alteration of the interface between the dermis and the epidermis[3]. The degradation of key skin components such as hyaluronic acid (HA), collagen, elastin, and fibrillin in skin tissue increases with age, which plays an essential role in the loss of skin quality over time[3]. Collagen is a protein that structures the skin, strengthening it and enabling it to resist mechanical constraints[4]. Collagen type I is the most abundant collagen in the body, and it is particularly important in skin, bone, and connective tissue. Collagen type III is the second most abundant collagen in skin tissues. Elastin is a protein that contributes to elasticity and flexibility in the skin[4]. Fibrillin-1 is a glycoprotein of the network of fibers that contributes to ensuring skin elasticity[4]. HA is a polysaccharide that plays a key role in the structural integrity and hydration of the skin[4]. Thus, HA is a molecule that is strongly involved in aging, and this fact drives the use of HA injectables as HA filler products to correct age-related facial contour abnormalities and rhytides due to its ability to function as a space-occupying device or as a skin booster product to notably improve skin quality by stimulation of the production of several extracellular matrix components[5-7]. With a large consensus on HA injectables globally, the demand for treatments with these products increases strongly and continuously in the world[1,7-10]. In this environment, a 2.6% H-HA/3.2% sorbitol composition (Kylane Laboratoires, Switzerland) [Figure 1] was recently developed for treatment by subcutaneous injection, especially by injection into the superficial adipose tissue, of the skin laxity and the loss of skin elasticity due to skin aging. This composition contains 52 mg of high molecular weight (H-HA) hyaluronic acid stabilized with 64 mg of sorbitol into phosphate buffer solution, and it benefits from unique and specific biophysical properties[11].
Figure 1. Schematic representation of the 2.6% H-HA/3.2% sorbitol composition[11].
The aim of this study was to assess the stimulation capacity of the 2.6% H-HA/3.2% sorbitol composition to produce the key components collagen type I, collagen type III, elastin, HA, and fibrillin-1 in the dermis matrix of the skin after product administration into the hypodermis, especially into the superficial adipose tissue, close to the dermis. This study of the stimulation capacity was performed versus another commercially available HA product by assessment of the expression of biological markers related to collagen type I, collagen type III, elastin, HA, and fibrillin-1 in living human skin explants after subcutaneous injection.
METHODS
Materials
The 2.6% H-HA/3.2% sorbitol composition (Kylane Laboratoires, Switzerland) indicated to be injected into the subcutaneous tissue for skin quality improvement was studied in comparison with another commercial injectable product intended for the same clinical indication: the 1.6% L-HA + 1.6% H-HA composition (Profhilo, IBSA, Italy) is a mixture of low molecular weight HA (0.08-0.10 MDa) at 16 mg/mL and of high molecular weight HA (1.10-1.40 MDa) at 16 mg/mL into a buffered sodium chloride solution[12]. The 1.6% L-HA + 1.6% H-HA composition is indicated to be injected into the dermal/subcutaneous levels.
Human skin explants collection and preparation
This study was performed on biopsies obtained from surgical residues after written informed consent from the donor, in full compliance with the Declaration of Helsinki and article L.1243-4 of the French Public Health Code. The latter does not require any prior authorization by an ethics committee for sampling and using surgical waste. Twenty-one skin explants of 12 ± 1 mm in diameter with the three skin layers epidermis, dermis, and hypodermis were collected from an abdominoplasty of a 39-year-old Caucasian woman with a phototype II according to the Fitzpatrick classification.
Skin explants treatment and culture in CO2 humid incubator
On Day 0 (D0), the skin explants were treated with the following methods:
- Three skin explants were not injected and were designated as untreated control for testing at D0.
- Six skin explants were not injected and were designated as the control of the study after D0.
- Six skin explants were injected via a needle into the superficial adipose tissue, close to the dermis, with
- Six skin explants were injected via a needle into the superficial adipose tissue, close to the dermis, with
After treatment, the skin explants were cultured in a survival culture medium [glucose (3.65 g/L),
Sampling and histologic preparation
On Day 0 (D0), three round samples from the untreated control group were collected and cut into two parts, with one part frozen, cryofixed, and cryosectioned (histological sections of 5 µm) and the other part formalin-fixed, paraffin-embedded, and sectioned.
On Days 3 (D3) and 8 (D8) after injection, the same process was performed on three round samples from each of the three other treatment groups. The histological sections were observed under a Leica DMLB microscope (Leica Microsystems GmbH, Germany) or BX43/BX63 Olympus microscopes (Olympus Corporation, Japan) and stained to assess the general morphology of the skin tissues and biological markers of hyaluronic acid, elastin, collagen type I, collagen type III, and fibrillin-1 into the dermal layer.
Assessment of general morphology
The general morphology of the epidermis, dermis, and hypodermis of the untreated and treated skin explants was examined by a histopathologist from the paraffinized sections stained according to Masson’s trichrome (Goldner variant). As part of this general morphology assessment, the histopathologist evaluated the general aspect of the tissues and the cell morphology (shape and size) for stratum corneum, epidermis, dermis, and hypodermis. The dermal-epidermal junction (DEJ) was also analyzed, especially by the determination of the DEJ index, i.e., the measurement of the levels of relief and undulations of the DEJ with the use of Cell^D (Olympus Corporation, Japan) software.
Assessment of the biological marker of hyaluronic acid
To evaluate expression of hyaluronic acid, paraffinized sections were stained with HABP (hyaluronic acid binding protein) biotinylated (amsbio, ref AMS.HKD.BC41) diluted 1:800 into PBS-BSA 0.3% for 1 h at room temperature before amplification by a biotin/streptavidin system and VIP substrate kit (Vector, ref SK 4600), a peroxidase substrate developing a purple color when oxidized. Microscopic evaluation of the hyaluronic acid expression was based on the qualitative microscopic observation of the stained sections (nine images per sample) with CellSens (Olympus Corporation, Japan) software and the quantitative analysis of the images (nine images per sample) with Cell^D (Olympus Corporation, Japan) software (calculating three percentage of interest area covered by the immunostaining).
Assessment of the biological marker of collagen type I
To evaluate collagen type I expression, frozen sections were stained with an anti-collagen type I polyclonal antibody (Monosan, ref PS047) diluted 1:50 into PBS-BSA 0.3%-tween 20 0.05% for 1 h at room temperature and stained with AlexaFluor 488 (Life Technologies, ref A11008). Nuclei were post-stained with propidium iodide. Microscopic evaluation of the collagen type I expression was based on the qualitative microscopic observation of the stained sections (nine images per sample) with CellSens (Olympus Corporation, Japan) software and the quantitative analysis of the images (nine images per sample) with Cell^D (Olympus Corporation, Japan) software (calculating the percentage of interest area covered by the immunostaining).
Assessment of the biological marker of elastin
To evaluate elastin expression, paraffinized sections were stained with an anti-elastin polyclonal antibody (Novotec, ref 25011) diluted 1:100 into PBS-BSA 0.3%-tween 20 0.05% for 1 h at room temperature and stained with AlexaFluor 488 (Life Technologies, ref A11008). Microscopic evaluation of the elastin expression was based on the qualitative microscopic observation of the stained sections (nine images per sample) with CellSens (Olympus Corporation, Japan) software and the quantitative analysis of the images (nine images per sample) with Cell^D (Olympus Corporation, Japan) software (calculating the percentage of interest area covered by the immunostaining).
Assessment of the biological marker of collagen type III
To evaluate collagen type III expression, frozen sections were stained with an anti-collagen type III polyclonal antibody (SBD, ref 1330-01) diluted 1:100 into PBS-BSA 0.3% for 1 h at room temperature before amplification by a biotin/streptavidin system and VIP substrate kit (Vector, ref SK 4600), a peroxidase substrate developing a purple color when oxidized. Microscopic evaluation of the collagen type III expression was based on the qualitative microscopic observation of the stained sections (nine images per sample) with CellSens (Olympus Corporation, Japan) software and the quantitative analysis of the images (nine images per sample) with Cell^D (Olympus Corporation, Japan) software (calculating the percentage of interest area covered by the immunostaining).
Assessment of the biological marker of fibrillin-1
To evaluate fibrillin-1 expression, frozen sections were stained with an anti-fibrillin-1 polyclonal antibody (Sigma Aldrich, ref HPA021057) diluted 1:200 into PBS-BSA 0.3% for 10 h at room temperature and stained with AlexaFluor 488 (Life Technologies, ref A11008), giving green fluorescence. Nuclei were post-stained with propidium iodide. Microscopic evaluation of the fibrillin-1 expression was based on the qualitative microscopic observation of the stained sections (nine images per sample) with CellSens (Olympus Corporation, Japan) software and the quantitative analysis of the images (nine images per sample) with Cell^D (Olympus Corporation, Japan) software (calculating the percentage of interest area covered by the immunostaining).
Statistics
Quantitative data were expressed as mean. Comparison between injected explants from the tested products and untreated control was assessed at each time point using the Student’s t-test, and significance was determined at P < 0.1, 0.05, or 0.01.
RESULTS
Skin explants morphology
Histological examination revealed that general integrity of the skin explants was maintained throughout the study for epidermis, dermis, and hypodermis. Minor and relatively consistent evolutions in skin morphology from baseline to Day 8 (D8) were observed in all groups.
On Day 3 (D3) after injection, in comparison with the control, the morphologic assessment revealed good quality for the three layers of the skin (epidermis, dermis, and hypodermis) for the skin explants treated with both the 2.6% H-HA/3.2% sorbitol composition and the 1.6% L-HA + 1.6% H-HA composition. In comparison with the control, the skin explants treated with the 2.6% H-HA/3.2% sorbitol composition showed on Day 3 (D3) a slight increase in the stratum corneum thickness and the laminated structure. No visible modification was observed in the case of the skin explants treated with the 1.6% L-HA + 1.6% H-HA composition compared with the control.
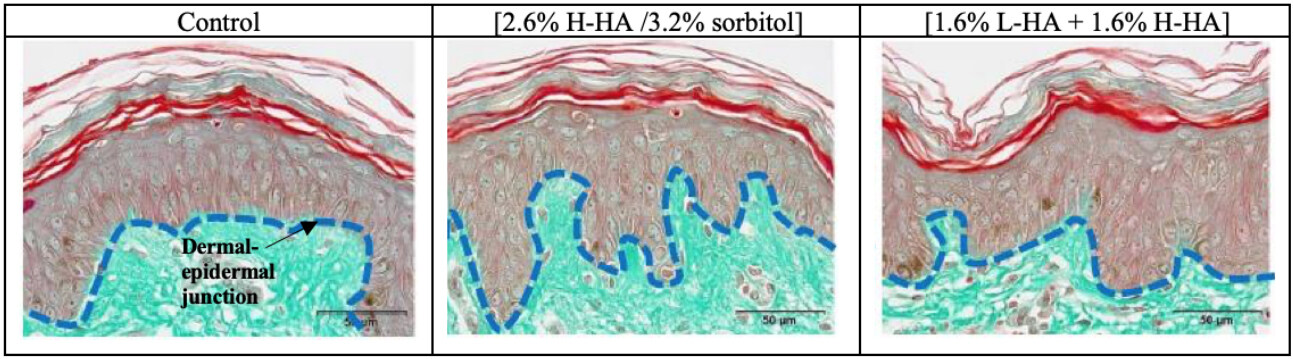
On Day 8 (D8) after injection, in comparison with the control, the morphologic assessment revealed good quality for the three layers of the skin (epidermis, dermis, and hypodermis) for the skin explants injected with both the 2.6% H-HA/3.2% sorbitol composition and the 1.6% L-HA + 1.6% H-HA composition. In comparison with the control, the skin explants treated with the 2.6% H-HA/3.2% sorbitol composition showed an improvement in the relief of the dermal-epidermal junction. No visible modification was observed in the case of the skin explants treated with the 1.6% L-HA + 1.6% H-HA composition.
Figure 2 presents images of the general skin morphology on D8 for the control and the skin explants treated with the 1.6% L-HA + 1.6% H-HA and 2.6% H-HA/3.2% sorbitol compositions.
Figure 2. Microscopic observation of the general skin morphology on Day 8 (D8) after injection. Improvement of the relief of the dermal-epidermal junction (DEJ) was observed with the 2.6% H-HA/3.2% sorbitol and 1.6% L-HA + 1.6% H-HA compositions, as compared with the control. The blue dotted line represents the DEJ.
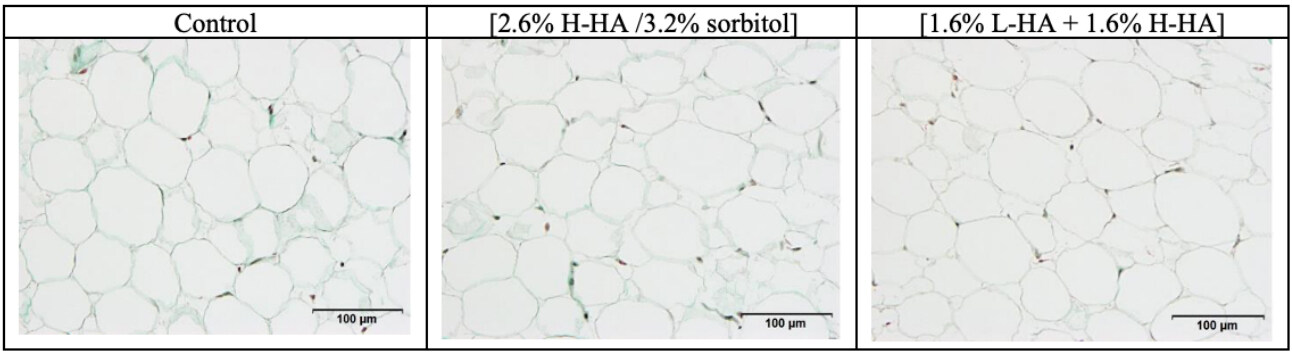
Figure 3 presents images of the adipose tissue morphology on D8 for the control and the skin explants treated with the 1.6% L-HA + 1.6% H-HA and 2.6% H-HA/3.2% sorbitol compositions. The morphology (size and shape) of the adipocytes was ranked as “good” by the histopathologist for the control, 2.6%
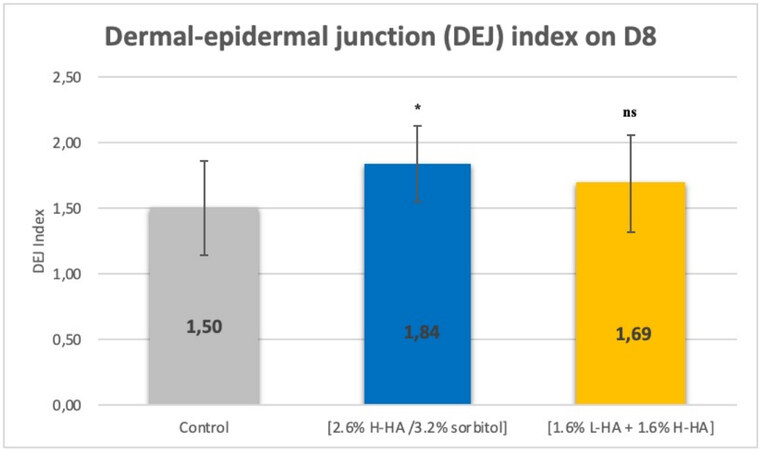
Figure 4 presents the dermal-epidermal junction index on D8 for the control and the skin explants treated with the 1.6% L-HA + 1.6% H-HA and 2.6% H-HA/3.2% sorbitol compositions. In comparison with the control, a significant increase of 23% (P < 0.05) in the dermal-epidermal junction index was observed for the skin explants treated with the 2.6% H-HA/3.2% sorbitol composition versus a non-significant increase of 13% for the skin explants treated with the 1.6% L-HA + 1.6% H-HA composition.
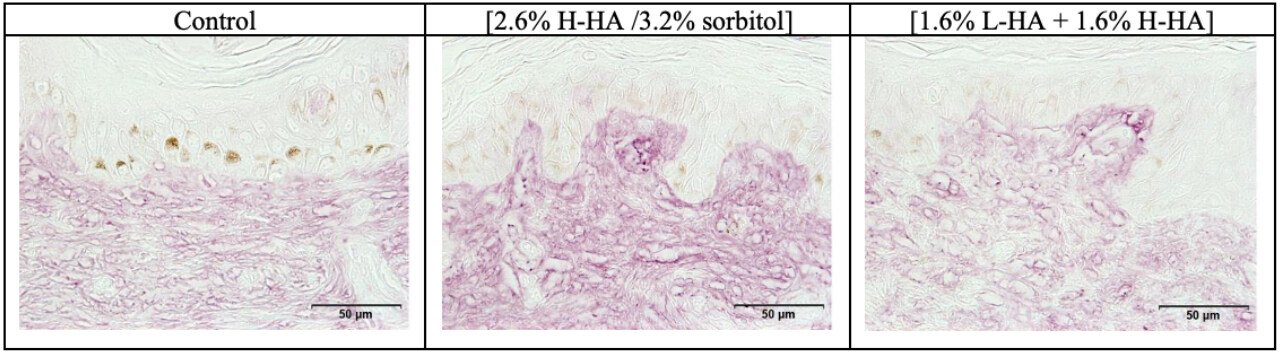
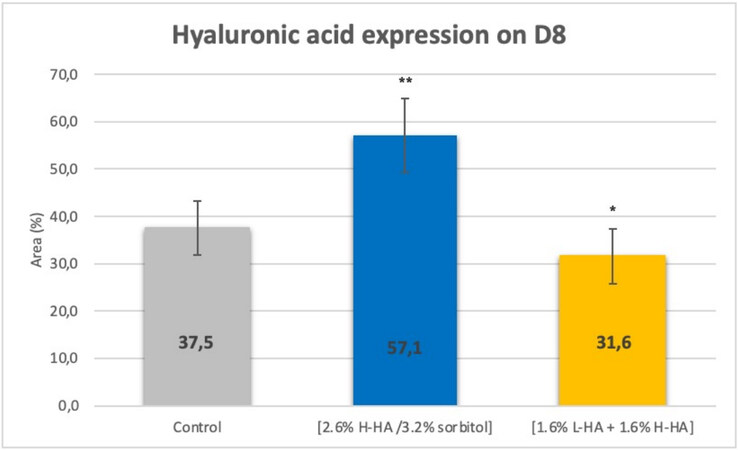
Evidence on hyaluronic acid expression in dermal layer
On Day 8 (D8) after injection, in comparison with the control, microscopic examination [Figure 5] and images analyses revealed that expression of the hyaluronic acid in the dermis was significantly increased by 52% (P < 0.01) for the 2.6% H-HA/3.2% sorbitol composition and significantly decreased by 16% (P < 0.05) for the 1.6% L-HA + 1.6% H-HA composition [Figure 6].
Figure 5. Microscopic observation of the hyaluronic acid expression in the dermal layer on Day 8 (D8) after injection. Increases in the density of the hyaluronic acid network were observed in the dermis following injection of the 2.6% H-HA/3.2% sorbitol composition, by the heightened intensity of purple staining, as compared with the control.
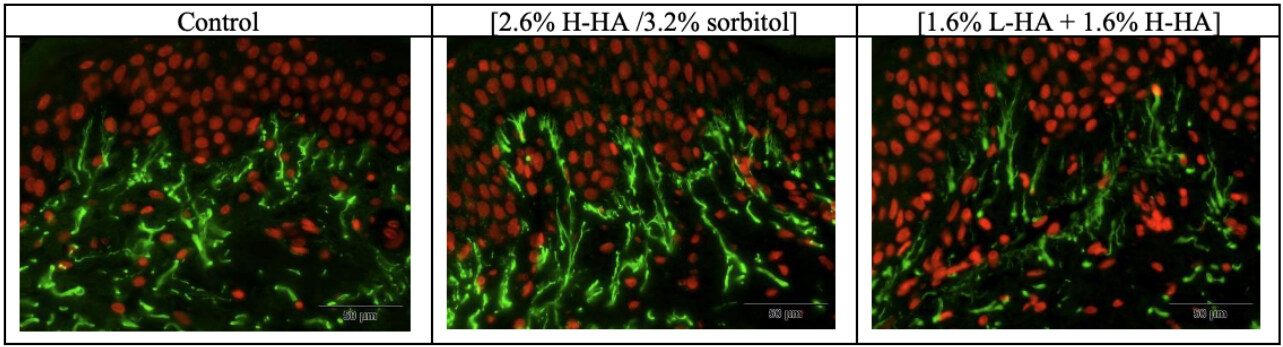
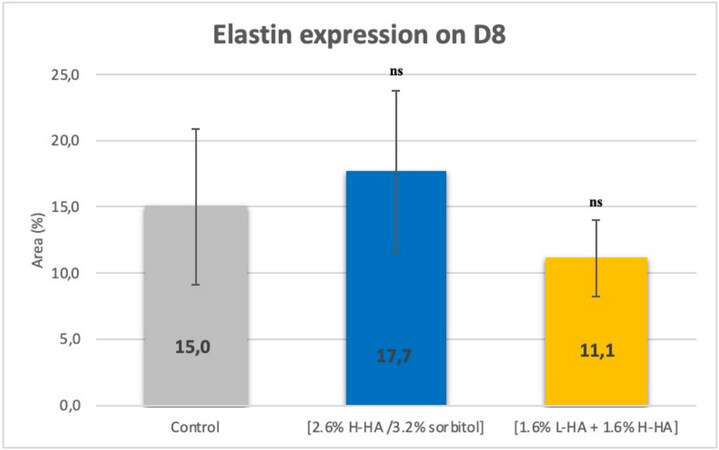
Evidence on elastin expression in dermal layer
On Day 8 (D8) after injection, in comparison with the control, microscopic examination [Figure 7] and images analyses revealed that expression of the elastin in the dermis was non-significantly increased by 18% for the 2.6% H-HA/3.2% sorbitol composition and non-significantly decreased by 26% for the 1.6% L-HA + 1.6% H-HA composition [Figure 8].
Figure 7. Microscopic observation of the elastin expression in the dermal layer on Day 8 (D8) after injection. Increases in the density of the elastin network were observed in the dermis following injection of the 2.6% H-HA/3.2% sorbitol composition, by the heightened intensity of green staining, as compared with the control.
Evidence on collagen type I expression in dermal layer
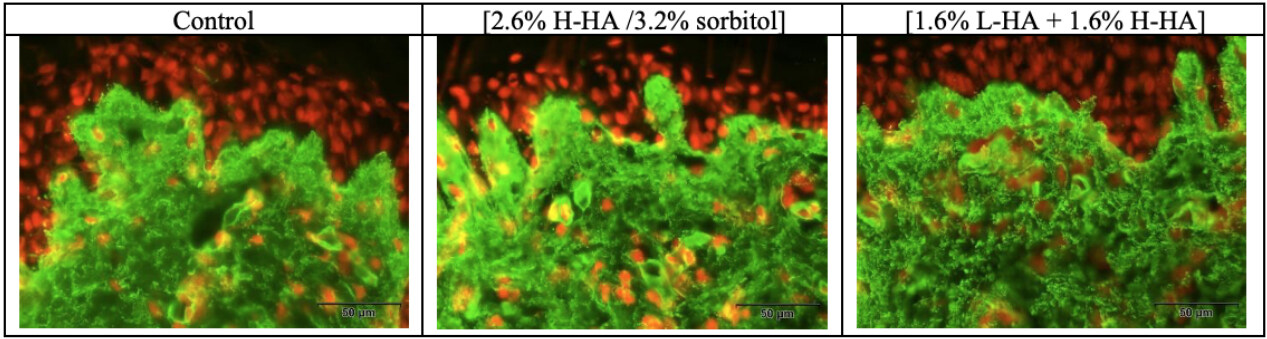
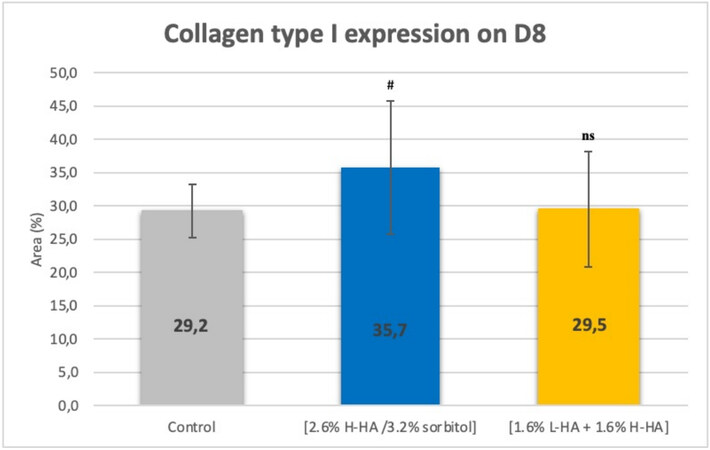
On Day 8 (D8) after injection, in comparison with the control, microscopic examination [Figure 9] and images analyses revealed that expression of the collagen type I in the dermis was significantly increased by 22% (P < 0.1) for the 2.6% H-HA/3.2% sorbitol composition and non-significantly increased by 1% for the 1.6% L-HA + 1.6% H-HA composition [Figure 10].
Figure 9. Microscopic observation of the collagen type I expression in the dermal layer on Day 8 (D8) after injection. Increases in the density of the collagen type I network were observed in the dermis following injection of the 2.6% H-HA/3.2% sorbitol composition, by the heightened intensity of green staining, as compared with the control.
Evidence on collagen type III expression in dermal layer
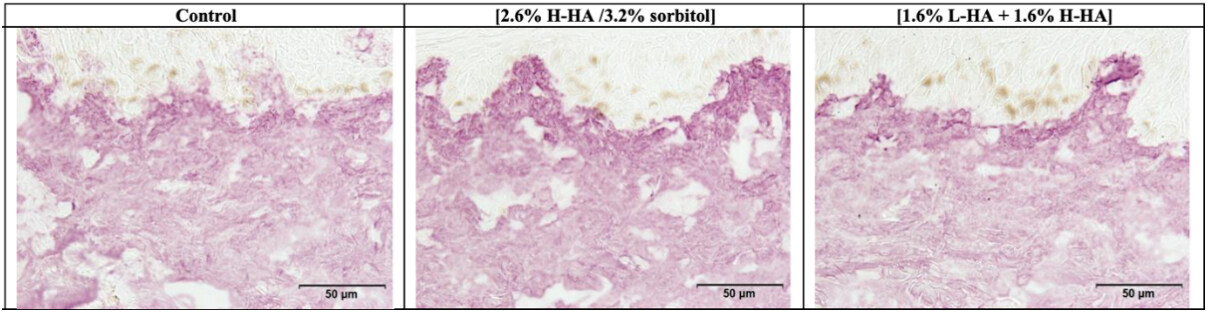
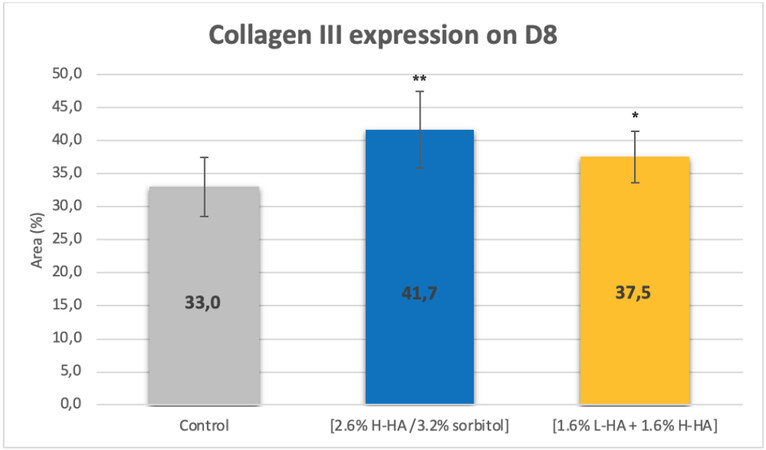
On Day 8 (D8) after injection, in comparison with the control, microscopic examination [Figure 11] and images analyses revealed that expression of the collagen type III in the dermis was significantly increased by 26% (P < 0.01) for the 2.6% H-HA/3.2% sorbitol composition and significantly increased by 14% (P < 0.05) for the 1.6% L-HA + 1.6% H-HA composition [Figure 12].
Figure 11. Microscopic observation of the collagen type III expression in the dermal layer on Day 8 (D8) after injection. Increases in the density of the collagen type III network were observed in the dermis following injection of the 2.6% H-HA/3.2% sorbitol composition, by the heightened intensity of purple staining, as compared with the control.
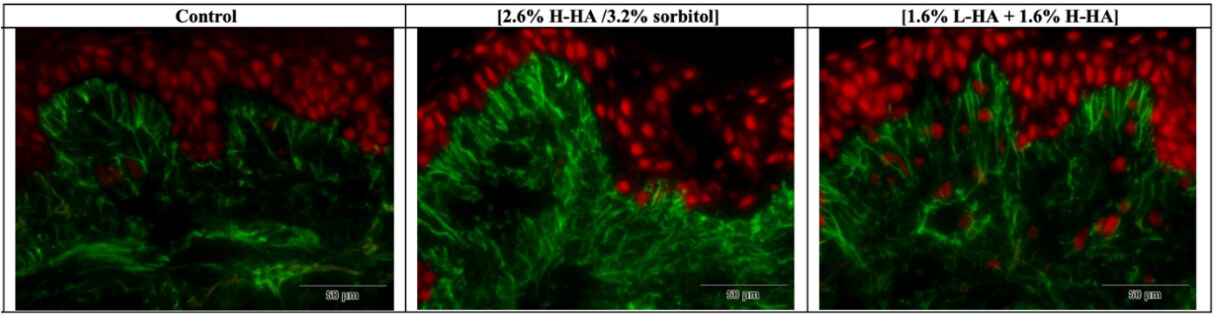
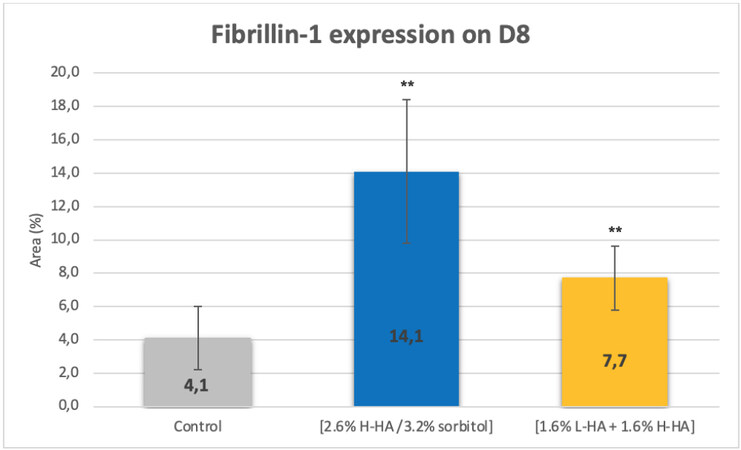
Evidence on fibrillin-1 expression in dermal layer
On Day 8 (D8) after injection, in comparison with the control, microscopic examination [Figure 13] and images analyses revealed that expression of the fibrillin-1 in the dermis was significantly increased by 241%
Figure 13. Microscopic observation of the fibrillin-1 expression in the dermal layer on Day 8 (D8) after injection. Increases in the density of the fibrillin-1 network were observed in the dermis following injection of the 2.6% H-HA/3.2% sorbitol composition, by the heightened intensity of green staining, as compared with the control.
DISCUSSION
The 2.6% H-HA/3.2% sorbitol product was developed for skin quality improvement with the aim to propose a safe hyaluronic acid composition with a high concentration of high molecular weight hyaluronic acid stabilized with sorbitol having differentiated biophysical properties for injection specifically into the subcutaneous tissue[11]. This HA/sorbitol composition was previously evaluated regarding its biophysical properties, its tolerance in an in vivo animal study, and its efficacy with skin firmness assessment on human skin explants[11]. This investigation demonstrated that this novel composition has differentiated biomechanical features, proper biocompatibility with skin tissues, and high performance to improve skin firmness[11]. The histological examinations performed in this study as part of the skin explants morphology evaluation confirmed the tolerance and safety of the 2.6% H-HA/3.2% sorbitol composition with the demonstration of the general integrity of the epidermis, dermis, and hypodermis for all the treated skin explants during the whole duration of the study. Furthermore, on Day 3 (D3) after injection, in comparison with the control, the skin explants treated with the 2.6% H-HA/3.2% sorbitol composition were characterized by a slight increase in the stratum corneum thickness and an epidermis structure which was more laminated, supporting the beneficial role of the composition on the quality of the skin layers structure, up to the surface of the skin, even though the 2.6% H-HA/3.2% sorbitol composition was implanted into the superficial adipose tissue. In addition to these observations, as illustrated in Figure 2, the dermal-epidermal junction, which is the interface between the lower part of epidermis and the top layer of dermis, was demonstrated to have a higher dermal-epidermal junction index than that of the control, significantly increased by 23% (P < 0.05) on Day 8 (D8) after injection of the 2.6% H-HA/3.2% sorbitol composition.
This increase supports the fact that the dermal-epidermal junction had more important relief and undulations, reflecting the structure of younger skin. This dermal-epidermal junction is very important for the skin because it serves the functions of mechanical support for the epidermis and the dermal-epidermal adherence[13]. One of the characteristics of skin aging is the disappearance of epidermal villi, significantly affecting the structure of the dermal-epidermal junction. While sinusoidal in young skin, the dermal-epidermal junction flattens with aging. The exchange surface between the epidermis and the dermis is therefore reduced. Cohesion between the dermis and the epidermis is less effective. Thus, the skin loses its firmness, contours relax, and wrinkles form. The healing and tissue regeneration processes are also altered. The relief of the dermal-epidermal junction is largely linked to the underlying elastic network. The reinforcement of this dermal-epidermal junction following the administration of the 2.6% H-HA/3.2% sorbitol composition is thus highly positive for the quality of the skin surface, especially to maintain the architectural integrity of the epidermis and provide protection against shearing forces[14].
On the other hand, the present ex vivo study with biological markers in living human skin explants demonstrated that the production of HA, elastin, collagen type I, collagen type III, and fibrillin-1 was stimulated in the dermal layer following the administration of the 2.6% H-HA/3.2% sorbitol composition into the superficial adipose tissue. This relevant model has been used in different studies to assess the early treatment effects in skin tissues because this model is able to maintain tissue viability in culture for up to
During the last two decades, more and more evidence has been brought to demonstrate that adipose tissue, especially subcutaneous white adipose tissue (sWAT), plays a key role in skin aging and should be considered as an important target in skin rejuvenation[18-22]. With aging, the structure, volume, and mechanical features of sWAT are strongly modified, and sWAT can influence the mechanical features of the skin layers[18,23], in line with the architectural continuum of skin from the deep fascia to the skin surface[24,25]. Additionally, it was demonstrated that adipocytes from sWAT have the potential to interact physiologically with fibroblasts to act on their synthetic properties, thereby affecting the mechanical properties of different skin layers[18,26-28].
The design of the novel 2.6% H-HA/3.2% sorbitol composition has its root in this, wherein the dermal tissue is not the only target of treatment for the HA skin booster injectables, but the subcutaneous tissue should become a very important target for the improvement of the skin quality. With this new treatment strategy, the aim is to provide available product solutions with specific composition and biophysical properties tailored for this targeted tissue, bringing adapted modifications at the level of the subcutaneous tissue and influencing the structure and mechanical properties of different skin layers to achieve positive effects on global skin quality and better resistance to structural alterations.
In accordance with the safety and performance outcomes obtained on the novel composition HA/sorbitol in[11], the results of the present study support this approach, which is increasingly reported in the literature. Injection of the 2.6% H-HA/3.2% sorbitol composition into the superficial adipose tissue, instead of in the dermis as is the case with most available HA skin booster products that are indicated and designed for intradermal administration, has the following benefits:
- There are some benefits in the skin morphology up to the stratum corneum.
- The production of hyaluronic acid, elastin, collagen, and fibrillin is stimulated in the dermis.
In conclusion, most skin quality injectable boosters containing hyaluronic acid available on the market are designed for intradermal injections. On the contrary, the novel 2.6% H-HA/3.2% sorbitol composition was specifically invented and developed to focus on adipose tissue treatment.
More and more publications in the literature provide evidence that modification of the subcutaneous tissue must be considered as an important target in facial aging, and factors such as the volume reduction of adipose tissue and the role of adipocytes to physiologically interact with skin fibroblasts can contribute to modifying the structure and elastic properties of the skin layers and, ultimately, improving the global quality of the skin.
With the present ex vivo study on biological markers in living human skin explants, this article shows that the treatment of adipose tissue with an HA injectable developed specifically to target this tissue is relevant to improve skin quality, in line with publications supporting the importance of the hypodermis as a key target to rejuvenate the skin. Indeed, although the 2.6% H-HA/3.2% sorbitol composition was injected into the superficial adipose tissue and not the dermis, benefits to the skin morphology were observed up to the stratum corneum, and evidence of expression of hyaluronic acid, elastin, collagen type I, collagen type III, and fibrillin-1 in the dermal layer was obtained.
In this respect, with a treatment strategy targeting adipose tissue to rejuvenate the skin, the 2.6% H-HA/3.2% sorbitol composition can be considered as an attractive treatment solution to structure the skin foundations and improve the skin quality to reach benefits not only in the subcutaneous tissue but more globally in the different skin layers.
DECLARATIONS
Authors’ contributionsObtain all the experimental data: Gavard Molliard S, Bon Bétemps J
All authors contributed to design of the concept and writing of the article.
Availability of data and materialsExperimental data were obtained by Kylane Laboratoires SA.
Financial support and sponsorshipKylane Laboratoires SA provided the logistical and financial support for the execution of this study.
Conflicts of interestGavard Molliard S, Bon Bétemps J, Hadjab B are employed by Kylane Laboratoires SA. Other authors declared that there are no conflicts of interest.
Ethical approval and consent to participateThere is no ethical approval number for the ex vivo study in accordance with the following element of the “METHODS/Human skin explants collection and preparation” section in this article: “This study was performed on biopsy, obtained from surgical residues after written informed consent from the donor, in full compliance with the Declaration of Helsinki and article L.1243-4 of the French Public Health Code. The latter does not require any prior authorization by an ethics committee for sampling and using surgical waste”. The human skin tissue (surgical residues) was obtained under the authorization DC-2008-542 issued by the Ministry of Research after validation by the national ethics committee.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Goldie K, Kerscher M, Fabi SG, et al. Skin quality - a holistic 360 ° view: consensus results. Clin Cosmet Investig Dermatol 2021;14:643-54.
2. Sparavigna A. Role of the extracellular matrix in skin aging and dedicated treatment - state of the art. Plast Aesthet Res 2020;7:14.
3. Stellavato A, Corsuto L, D’Agostino A, et al. Hyaluronan hybrid cooperative complexes as a novel frontier for cellular bioprocesses re-activation. PLoS One 2016;11:e0163510.
4. Nakab L, Hee CK, Guetta O. Improvements in skin quality biological markers in skin explants using hyaluronic acid filler VYC-12L. Plast Reconstr Surg Glob Open 2020;8:e2723.
5. Paliwal S, Fagien S, Sun X, et al. Skin extracellular matrix stimulation following injection of a hyaluronic acid-based dermal filler in a rat model. Plast Reconstr Surg 2014;134:1224-33.
6. Safa M, Natalizio A, Hee CK. A prospective, open-label study to evaluate the impact of VYC-12L injection on skin quality attributes in healthy volunteers. Clin Cosmet Investig Dermatol 2022;15:411-26.
7. Kleine-Börger L, Meyer R, Kalies A, Kerscher M. Approach to differentiate between hyaluronic acid skin quality boosters and fillers based on their physicochemical properties. J Cosmet Dermatol 2022;21:149-57.
8. Cassuto D, Bellia G, Schiraldi C. An overview of soft tissue fillers for cosmetic dermatology: from filling to regenerative medicine. Clin Cosmet Investig Dermatol 2021;14:1857-66.
9. Gavard Molliard S, Bon Bétemps J, Hadjab B , Topchian D, Micheels P, Salomon D. Key rheological properties of hyaluronic acid fillers: from tissue integration to product degradation. Plast Aesthet Res 2018;5:17.
10. Bon Betemps J, Marchetti F, Lim T, et al. Projection capacity assessment of hyaluronic acid fillers. Plast Aesthet Res 2018;5:19.
11. Gavard Molliard S, Bon Bétemps J, Hadjab B, Ghazal A, Badi M, Cerrano M. Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality. Plast Aesthet Res 2022;9:52.
12. Cassuto D, Delledonne M, Zaccaria G, Illiano I, Giori AM, Bellia G. Safety assessment of high- and low-molecular-weight hyaluronans (Profhilo®) as derived from worldwide postmarketing data. Biomed Res Int 2020;2020:8159047.
14. Goletz S, Zillikens D, Schmidt E. Structural proteins of the dermal-epidermal junction targeted by autoantibodies in pemphigoid diseases. Exp Dermatol 2017;26:1154-62.
15. Sundaram H, Mackiewicz N, Burton E, et al. Pilot comparative study of the topical action of a novel, crosslinked resilient hyaluronic acid on skin hydration and barrier function in a dynamic, three-dimensional human explant model. J Drugs Dermatol 2016;15:434-441.
16. Lebonvallet N, Jeanmaire C, Danoux L, Sibille P, Pauly G, Misery L. The evolution and use of skin explants: potential and limitations for dermatological research. Eur J Dermatol 2010;20:671-84.
17. Park GH, Chang SE, Bang S, et al. Usefulness of skin explants for histologic analysis after fractional photothermolysis. Ann Dermatol 2015;27:283-90.
18. Wollina U, Wetzker R, Abdel-Naser MB, Kruglikov IL. Role of adipose tissue in facial aging. Clin Interv Aging 2017;12:2069-76.
19. Sadick NS, Dorizas AS, Krueger N, Nassar AH. The facial adipose system: its role in facial aging and approaches to volume restoration. Dermatol Surg 2015;41 Suppl 1:S333-9.
21. Kruglikov IL, Scherer PE. Skin aging: are adipocytes the next target? Aging (Albany NY) 2016;8:1457-69.
22. Trivisonno A, Rossi A, Monti M, et al. Facial skin rejuvenation by autologous dermal microfat transfer in photoaged patients: clinical evaluation and skin surface digital profilometry analysis. J Plast Reconstr Aesthet Surg 2017;70:1118-28.
24. Wong R, Geyer S, Weninger W, Guimberteau JC, Wong JK. The dynamic anatomy and patterning of skin. Exp Dermatol 2016;25:92-8.
25. Guimberteau JC, Delage JP, Wong J. New views about the skin. Ann Chir Plast Esthet 2010;55:255-66.
26. Ezure T, Amano S. Influence of subcutaneous adipose tissue mass on dermal elasticity and sagging severity in lower cheek. Skin Res Technol 2010;16:332-8.
27. Ezure T, Amano S. Negative regulation of dermal fibroblasts by enlarged adipocytes through release of free fatty acids. J Invest Dermatol 2011;131:2004-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Bon Bétemps J, Gavard Molliard S, Hadjab B, Badi M, Ghazal A, Cerrano M. Study of biological markers in skin quality treatment by subcutaneous injection of a stabilized composition of 26 mg/mL of high molecular weight HA. Plast Aesthet Res 2022;9:59. http://dx.doi.org/10.20517/2347-9264.2022.61
AMA Style
Bon Bétemps J, Gavard Molliard S, Hadjab B, Badi M, Ghazal A, Cerrano M. Study of biological markers in skin quality treatment by subcutaneous injection of a stabilized composition of 26 mg/mL of high molecular weight HA. Plastic and Aesthetic Research. 2022; 9: 59. http://dx.doi.org/10.20517/2347-9264.2022.61
Chicago/Turabian Style
Bon Bétemps, Jérémie, Samuel Gavard Molliard, Basste Hadjab, Mhd Badi, Anas Ghazal, Marco Cerrano. 2022. "Study of biological markers in skin quality treatment by subcutaneous injection of a stabilized composition of 26 mg/mL of high molecular weight HA" Plastic and Aesthetic Research. 9: 59. http://dx.doi.org/10.20517/2347-9264.2022.61
ACS Style
Bon Bétemps, J.; Gavard Molliard S.; Hadjab B.; Badi M.; Ghazal A.; Cerrano M. Study of biological markers in skin quality treatment by subcutaneous injection of a stabilized composition of 26 mg/mL of high molecular weight HA. Plast. Aesthet. Res. 2022, 9, 59. http://dx.doi.org/10.20517/2347-9264.2022.61
About This Article
Copyright
Data & Comments
Data
 Cite This Article 7 clicks
Cite This Article 7 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.