A unique late complication with the use of calcium hydroxylapatite filler in facial lipoatrophy rehabilitation
Sir,
Radiesse (Merz Aesthetics, Franksville, WI, USA) is an injectable filler material composed of synthetic calcium hydroxylapatite (CaHA) microspheres suspended in an aqueous carrier gel. Cosmetic use of Radiesse in facial rejuvenation is well-known. Treatment sites amenable to calcium hydroxylapatite (CH) injection include the naso-labial folds, marionette lines, perioral lines, prejowl sulcus, zygoma and malar eminence, tear trough depressions, nose, chin, acne scars, and it is also Food and Drug Administration-approved for facial augmentation in HIV patients with facial lipo-atrophy.[1]
Calcium hydroxylapatite filler enjoy an excellent safety record. The adverse events reported are similar to those observed with other short-acting fillers such as hyaluronic acid. There is no evidence of granuloma formation occurring with CaHA.[1] Although, presence of visible skin nodules has been reported, they are related to techniques, especially due to superficial injection of CH or its inappropriate use.
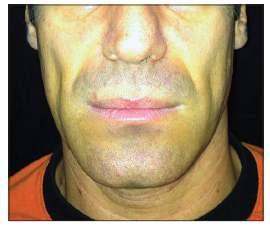
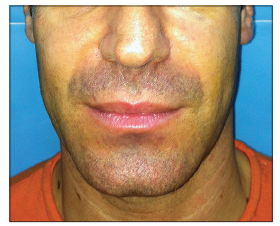
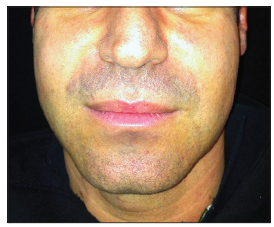
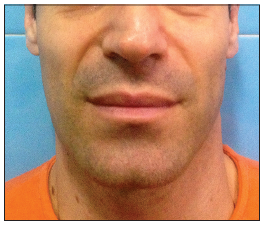
We describe a unique unreported complication, related to CH filler injection in an HIV positive patient. A 37-year-old, HIV positive, Caucasian male patient was referred to our University Hospital for facial lipo-atrophy rehabilitation [Figure 1]. The patient involved in this article agreed to have his pictures published and signed the consent form. We performed, as per our protocol,[2] the rehabilitation in one session injecting 7.5 mL of CH filler. After 10 days postinjection swelling and hematoma resolved [Figure 2]. Three weeks later, the patient came back to us with a complaint of a soft tissue swelling, involving the areas where the filler was injected [Figure 3]. The swelling was soft, nontender with sudden onset in the morning after waking up from sleep. A bromelina based therapy was prescribed for a week (Ananase, Rottapharm S.p.a., Milano) by us. After 2 weeks the swelling resolved completely with no recurrence in 8 months of follow-up [Figure 4]. To the best of our knowledge and after extensive literature search we could not determine the reason for this complication nor has such adverse effect ever reported in the literature.
Financial support and sponsorship
Nil.
Conficts of interest
None declared.
REFERENCES
1. Graivier MH, Bass LS, Busso M, Jasin ME, Narins RS, Tzikas TL. Calcium hydroxylapatite (Radiesse) for correction of the mid- and lower face: consensus recommendations. Plast Reconstr Surg 2007;120:55S-66S.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Rauso R. A unique late complication with the use of calcium hydroxylapatite filler in facial lipoatrophy rehabilitation. Plast Aesthet Res 2014;1:122-3. http://dx.doi.org/10.4103/2347-9264.143563
AMA Style
Rauso R. A unique late complication with the use of calcium hydroxylapatite filler in facial lipoatrophy rehabilitation. Plastic and Aesthetic Research. 2014; 1: 122-3. http://dx.doi.org/10.4103/2347-9264.143563
Chicago/Turabian Style
Rauso, Raffaele. 2014. "A unique late complication with the use of calcium hydroxylapatite filler in facial lipoatrophy rehabilitation" Plastic and Aesthetic Research. 1: 122-3. http://dx.doi.org/10.4103/2347-9264.143563
ACS Style
Rauso, R. A unique late complication with the use of calcium hydroxylapatite filler in facial lipoatrophy rehabilitation. Plast. Aesthet. Res. 2014, 1, 122-3. http://dx.doi.org/10.4103/2347-9264.143563
About This Article
Copyright
Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.