Distal median and radial nerve branch transfer techniques for upper extremity reanimation
Abstract
Nerve transfers for peripheral nerve injuries have become increasingly popular over the past two decades. While techniques for ulnar nerve repair have been well-documented, more recent techniques for median and radial nerve branch reinnervation are still being explored. This review describes the outcomes of common and emerging techniques for reinnervation of the distal branches of the median and radial nerves.
Keywords
INTRODUCTION
Nerve transfers provide additional options for restoring function through neurotizing recipient nerves with expendable donor nerves after severe injuries[1]. Extensive research into upper extremity shoulder, elbow, and distal ulnar nerve reanimation has been conducted, and new techniques for median and radial nerve branch reinnervation have been described[2]. This review discusses common and emerging hand and wrist reanimation themes, specifically looking at more recent techniques for the neurotization of muscles innervated by the anterior interosseous nerve (AIN) and distal radial nerve branches.
OVERVIEW OF NERVE INJURIES AND REPAIR
A basic understanding of the physiology of nerve repair is required to understand the factors that contribute to the success of nerve transfers. After traumatic transection, the nerve fibers distal to the injury lose contact with the neuronal cell body. Axonal regeneration is the primary means of recovery for these injuries and involves Wallerian degeneration, axonal regeneration, and end-organ reinnervation. Any disruption of these 3 processes can affect functional outcomes[3]. Wallerian degeneration, or the clearing process of the distal stump, serves to create a microenvironment in which axonal regrowth and reinnervation can occur. This process generally occurs within the first week after the injury, after which a peripheral nerve will start to regenerate at a rate of approximately 1 to 3 millimeters (mm) per day toward a distal target. However, muscle fibrosis and atrophy begin as early as 3 weeks following denervation[3]. Given the distance needed to travel in a distal nerve injury, irreversible functional damage can occur within a few months[3]. Although the window for repair and functional recovery is generally accepted as within 12 to 15 months, it is ideal for motor nerve regeneration and target reinnervation to actively occur within 3 to 4 months. Some evidence suggests this is a critical time point, after which regeneration outcomes start to become poor[4]. Of note, timing is different with sensory nerves. Target muscles with pure sensory receptors are less time-sensitive to regeneration, but mixed motor nerves degrade even more rapidly, with repairs delayed more than one month demonstrating significant functional decline[5].
Not all nerve injuries require repair, as management depends on injury severity and resulting functional deficits. Nerve transfers and other surgical options such as nerve grafting and tendon transfers are generally reserved for Sunderland grades IV and V injuries, which involve complete loss of axonal, endoneurial, and perineural continuity; and spontaneous recovery is not expected[6,7]. This contrasts with Sunderland grades I-III, which involves damage of local myelin to axons and endoneurium with intact perineurium. Full recovery is expected in these cases and management is generally conservative[8]. Although nerve grafting was predominantly favored in the past for severe injuries, recent advances in nerve transfer techniques have led to faster, superior outcomes and created a paradigm shift in the treatment strategy for all peripheral nerve injuries[2]. This is especially true for nerve injuries in the upper extremity, with the most common indications for nerve transfers including restoration of shoulder abduction, elbow flexion, radial nerve function, and hand function[6].
ANATOMY, NERVE FUNCTION, AND INJURY CONSEQUENCE
To discuss the outcomes of high AIN and radial nerve transfer techniques, this review provides a general overview of the anatomy and function of particular nerves of interest: the AIN and radial nerve branches in the forearm, including the posterior interosseous nerve (PIN) and the nerve to the extensor carpi radialis brevis (ECRB).
Anterior interosseous nerve
The AIN is a motor branch of the median nerve (C8-T1) with some joint branches that provide proprioceptive and deep pain feedback. It innervates the deep muscles in the forearm that control finger flexion, specifically the flexor pollicis longus (FPL), the lateral portion of the flexor digitorum profundus (FDP), and the pronator quadratus (PQ)[9,10]. It can be found branching from the median nerve at the cubital fossa, usually on the distal border of the pronator teres muscle. However, the origin of the AIN and its relation to the pronator teres muscle can be variable[11].
AIN injuries can be traumatic or spontaneous, caused by penetrating stab wounds, supracondylar fractures, orthopedic surgery complications, compartment syndrome, neuritis, or entrapment under the pronator teres muscle[12]. Patients will often be unable to make an "OK" sign and will have a positive Pinch Grip Test, where a patient will be unable to pinch an object with normal strength[13].
Radial nerve branches
The radial nerve originates from the posterior cord (C5-T1) of the brachial plexus[14]. In the proximal forearm, it gives off branches to the brachioradialis, extensor carpi radialis longus (ECRL) and ECRB before dividing into a superficial branch and the PIN[15]. In most cases, these branches are found proximal to the supinator canal, although the location can be variable. Notably, the nerve to the ECRB is found in the proximal forearm and can have anatomical variation: originating from the radial nerve before it divides, the PIN before it pierces through the supinator, or the superficial branch of the radial nerve[16]. The ECRL and ECRB are responsible for wrist extension, while the PIN is responsible for finger extension, innervating the extensor digitorum communis (EDC), extensor digitorum minimi (EDM), extensor carpi ulnaris (ECU), abductor pollicis (AP), extensor pollicis brevis (EPB), extensor pollicis longus (EPL), and extensor indicis proprius (EIP).
Injury to the radial nerve causes weakness in extension, with an isolated PIN injury resulting in finger extension weakness. As stated previously, the branches to the ECRL and ECRB typically come off the radial nerve before it passes through the supinator muscle and branches to form the PIN, so wrist extension is spared in cases of PIN injury. Additionally, radial deviation is usually present due to the lack of motor input from the ECU with the preserved function of the ECRL and ECRB[17].
REINNERVATION TECHNIQUES
Median nerve: anterior interosseous nerve transfers
Brachialis to AIN
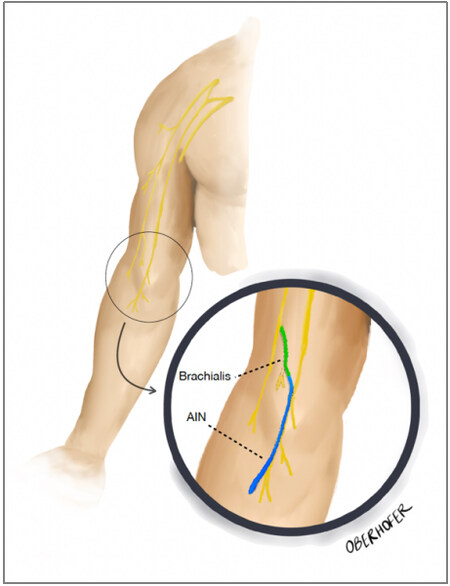
Technique: In this technique, the distal portion of the nerve to the brachialis, coming from the musculocutaneous nerve, is transferred to the AIN [Figure 1]. The patient is placed supine, and a sharp incision is made in the medial arm. Subcutaneous tissue is divided with sharp dissection and cautery, paying particular attention to the medial antebrachial cutaneous nerve. Once the median nerve and musculocutaneous nerve are exposed, the nerve to the brachialis can be identified, branching off the musculocutaneous nerve approximately 17 centimeters (cm) from the acromion. The brachialis branch is more distal than the biceps brachii nerve (13 cm from the acromion). The nerve branches are marked and protected, while the median nerve is inspected. The AIN fascicle is found within the median nerve proper, and intraoperative mapping is aided by a nerve stimulator to identify these fascicles. Once identified, the AIN is dissected proximally, and the remainder of the median nerve fascicles are identified. Following confirmation of the brachialis nerve with stimulation demonstrating brachialis muscle contraction, the brachialis nerve is dissected distally to decrease tension during the repair. The brachialis and AIN can then be coapted end-to-end using microsurgical techniques[18].
Figure 1. Illustration of relevant anatomy for nerve to brachialis to AIN transfer technique. The donor nerve to the brachialis is transferred to the recipient AIN. AIN: Anterior interosseous nerve.
Outcomes: The nerve to brachialis to AIN transfer is widely reported in the literature, and many reports have demonstrated favorable results. When assessing outcomes and clinical function, the Medical Research Council (MRC) scale for muscle strength can be utilized, with a grade 3 sufficient for object release and hand opening following AIN reinnervation[19]. Mackinnon et al. reported the first case of thumb and finger reinnervation after a spinal cord injury with brachialis to AIN transfer 23 months after injury[20]. Fifteen months postoperatively, the patient regained MRC grade 3 strength of the FPL and FDP[20]. In a case series of 4 patients written by Ray et al., all patients with brachial plexus injuries who received brachialis to AIN transfers regained grip strength, and 3/4 regained at least partial pinch strength[21]. All patients met at least MRC grade 3, with 3/4 patients recovering MRC grade 4 function of the FPL and FDP[21]. Hawasli et al. reported a detailed case of a patient who received a brachialis to AIN transfer after a complete C7 spinal cord injury[18]. At 3 months postoperatively, the patient demonstrated early reinnervation, regaining MRC grade 3 strength in the FDP and FPL of the left hand[18]. One of the most recent case series for this technique was described by Souza et al. in 2020, where 11 patients had lower brachial plexus injuries and received brachialis to AIN transfers in addition to supinator to PIN transfers[22]. Regarding the brachialis to AIN transfers, 8/11 recovered MRC grade 3 or higher on finger flexion[22].
Summary: Although this is an older reported technique, brachialis to AIN has shown relatively good outcomes, with 14/17 cases regaining at least MRC grade 3 flexion strength within two years of follow-up.
ECRB to AIN
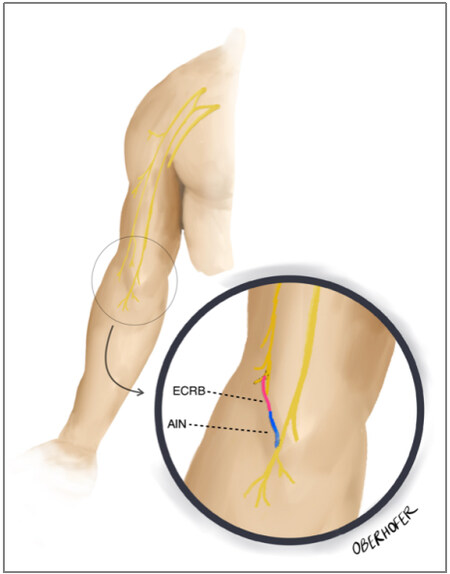
Technique: This technique transfers the nerve to the ECRB to the AIN [Figures 2 and 3]. An oblique incision in the proximal forearm is made, following the line of the pronator teres muscle. The median nerve is then exposed through medial retraction of the pronator teres. On the anterior or lateral aspect of the median nerve, the AIN can be identified as it passes under the pronator teres muscle, while the superficial branch of the radial nerve can be identified adjacent to the radial artery. The ECRB motor nerve branch can be identified most commonly as a trifurcation with the superficial radial nerve and PIN. It is confirmed with nerve stimulation demonstrating wrist extension. The ECRB is then dissected distally, and the AIN is cut proximally and the transfer is performed with tension-free coaptation[23].
Figure 2. Illustration of relevant anatomy for nerve to the ECRB to AIN nerve transfer technique. The donor nerve to the ECRB is transferred to the recipient AIN. AIN: Anterior interosseous nerve; ECRB: extensor carpi radialis brevis.
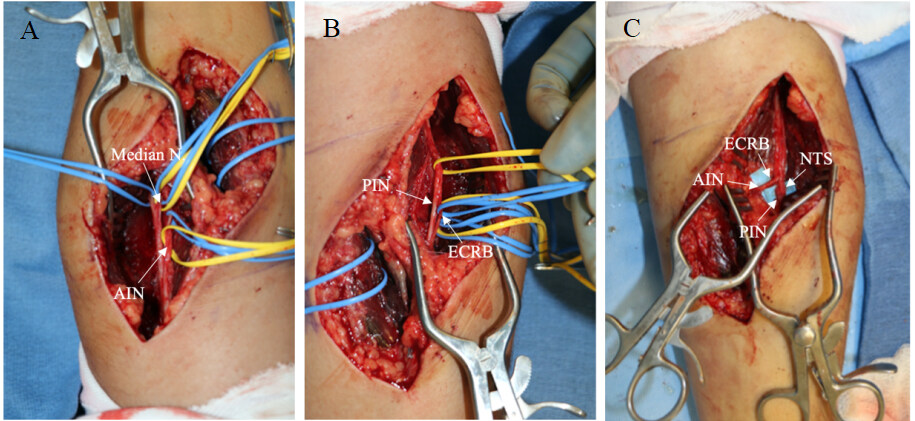
Figure 3. (A) Identification of the AIN branching from the median nerve. (B) Identification of the PIN and nerve to ECRB. (C) The donor nerve to the ECRB is transferred to the recipient AIN. The nerve to the supinator (NTS) is also transferred to the PIN. AIN: Anterior interosseous nerve; ECRB: extensor carpi radialis brevis; PIN: posterior interosseous nerve.
Outcomes: This technique has shown very favorable outcomes. Bertelli et al. reported 4 patients with high median or ulnar palsy or C7-T1 brachial plexus root avulsions who received nerve to ECRB to AIN transfers within 8 months of injury[23]. At 13 months postoperatively, all patients regained full finger and thumb flexion with grade MRC grade 4 strength[23]. Another study by Bertelli et al. compared surgical outcomes of 9 patients and 17 limbs after cervical spinal cord injury[24]. Nerve to the brachialis to AIN transfer was performed in 3 limbs, brachialis to other median nerve motor fascicles in 5 limbs, brachioradialis to AIN in 4 limbs, and nerve to the ECRB to AIN in 5 limbs. Finger flexion restoration was only observed in 4/8 limbs with brachialis transfer, with 3 limbs achieving MRC grade 3 flexion and one limb achieving MRC grade 4 flexion. Similarly, brachioradialis to AIN transfer showed incomplete flexion with MRC grade 4 strength. Meanwhile, ECRB to AIN had the best reported outcomes, with MRC grade 4 strength and full finger flexion in all 5 limbs and no downgrading of wrist extension or elbow flexion[24]. Salomão et al. most recently reported a single case report of a 29-year-old male who sustained a gunshot wound and received ECRB to AIN transfer 16 months after injury[25]. At a 2-year follow-up, the patient regained full flexion with MRC grade 4 strength and no donor site morbidity[25].
Summary: This technique shows great results with 10/10 cases of nerve to ECRB to AIN regaining MRC grade 4 finger flexion strength. This contrasts with brachialis to AIN, in which the majority of patients only regained MRC grade 3 strength. In addition to good reported outcomes, there were no cases of donor site morbidity.
Supinator to AIN
Technique: This technique utilizes an expendable branch of the radial nerve, the nerve to the supinator, to reinnervate the AIN. First, an incision is made below the antecubital fossa. To visualize the median nerve and branches, the superficial head of the pronator teres is retracted medially. Next, the AIN is identified and dissected from the median nerve. The radial nerve can be identified by locating the superficial radial nerve and following it proximally. There are typically 1 to 3 small nerve branches to the supinator, which can be confirmed with nerve stimulation. The nerve(s) to the supinator is then divided distally, and the AIN is divided proximally to allow for tension-free end-to-end coaptation.
This technique is advantageous because it does not preclude future tendon transfer to muscles innervated by the AIN if the resulting motor function is not adequate after the transfer. Furthermore, as forearm supination is primarily powered by the biceps, there is minimal donor site deficiency after transfer[26].
Outcomes: There are fewer reports on this technique in the literature than on transferring the nerve to the brachialis or nerve to the supinator to the AIN. Notably, Hsiao et al. described a case report of a patient with median nerve palsy following a proximal humerus fracture who received nerve transfers of supinator to AIN and ECRB to the pronator teres branch of the median nerve[26]. At 1-year follow-up, grip strength and pinch strength were regained at MRC grade 4+ for FPL and 4- for FDP. Although strength was adequate, the patient underwent tenodesis at 18 months to improve index finger flexion strength[26].
Summary: Although there are not many cases reported in the literature, this technique is promising, with 2/2 cases of supinator to AIN regaining MRC grade 4 FPL and FDP flexion strength. Although this has shown equal MRC grade 4 strength compared to ECRB to AIN, the latter technique has been more widely reported.
Distal radial nerve transfers
FDS to ECRB
Technique: This technique transfers the branch of the median nerve innervating the flexor digitorum superficialis (FDS) muscle to the branch of the radial nerve innervating the ECRB muscle. An incision is made below the antecubital crease in the proximal forearm. An intraoperative nerve stimulator is then used to identify the median nerve and its branches. The FDS branch can be identified by visualizing finger flexion at the proximal interphalangeal joints after stimulation. Of note, there can be significant anatomical variation in the location of the FDS branch. Once the median nerve and its branches are protected, the radial sensory nerve is identified and followed proximally to find the PIN and the branch to the ECRB. Following the identification of all nerves, the ECRB is divided proximally, and the FDS is divided distally to allow minimal tension and is repaired end-to-end with microsurgical techniques[28].
Outcomes: Although outcomes for this seem promising, there are few reports in the literature. Good outcomes for radial nerve branch transfers are defined by achieving at least grade MRC grade 3 on extension[29]. In 2007, Mackinnon et al. described a case report of a 32-year-old woman with radial nerve palsy after intramedullary humerus rod placement who received a transfer of FDS and FCR to ECRB and PIN, respectively[28]. At 18 months postoperatively, she regained MRC grade 4 finger and wrist extension strength[28]. Similarly, Ukrit et al. described two case reports of patients with C5, C6, and C7 avulsion injuries who received FDS to ECRB[30]. Both patients recovered MRC grade 4 wrist extension strength at the 2-year follow-up visits[30].
Summary: There are not many reports of this technique in the literature, but this technique is very promising, with 3/3 cases of FDS to ECRB regaining MRC grade 4 extension strength.
Distal AIN (PQ) to ECRB
Technique: The nerve to the pronator quadratus is transferred to the nerve to the ECRB in this technique. An oblique incision is made in the proximal forearm, a few centimeters below the antecubital fossa. The nerve to the ECRB can be identified by tracing the superficial branch of the radial nerve proximally. Following identification of the ECRB branch, the incision is extended distally and the distal AIN can be identified through a trans-FCR approach to expose the proximal aspect of the pronator quadratus. The AIN can then be seen entering the pronator quadratus. Contraction of the pronator quadratus with nerve stimulation confirms the correct identification of the AIN. The AIN is traced proximally, and care is taken to preserve the branch to the FPL. The ECRB is cut proximally at its origin, and the AIN is cut distally and, if necessary, further dissected within the substance of the pronator quadratus muscle for additional length. Following division, the AIN is turned proximally and passed radially to allow for coaptation to the ECRB motor branch.
Outcomes: In 2012, Bertelli et al. described the technique for transferring the distal AIN branch to pronator quadratus to the ECRB motor branch and reported 4 patients with brachial plexus injuries who underwent surgery within 10 months of injury[31]. At 12 months postoperatively, all patients gained MRC grade 4 wrist extension without loss or downgrading of pronation or strength in FPL or FDP flexion[31]. In another case series by Bertelli et al., 28 patients with C5-8 root injuries had this operation within 7 months after injury[32]. At approximately 22 months postoperatively, 25/28 patients scored MRC grade 4 extension, 2/28 scored MRC grade 3, and one scored MRC grade 2. Furthermore, there was no loss of function or downgrading of the FPL or FDP flexion strength[32]. Similarly, Bhatia et al. reported results of 20 patients with C5-8 root injuries who underwent operations within 9 months of injury[33]. In this series, 17/20 patients gained MRC grade 4 wrist extension, with the remaining 3 gaining MRC grade 3 extension. However, the authors reported that the 3 patients with lower scores had MRC grade 3 recordings of the DIP and thumb flexion before the transfer, indicating weakness of the donor nerve. Additionally, there was no loss in pronation in 14/20 patients, while 4/20 were downgraded to MRC grade 3 and one patient had complete loss of pronation. There were no cases of thumb and DIP flexion strength loss or downgrading[33]. Bertelli recently reported a larger case series of 14 patients with radial nerve lesions who received AIN to ECRB and FCR to PIN in 2020. 13/14 recovered M4 and 1/14 recovered M3 wrist extension strength[34].
Summary: Distal AIN (PQ) to ECRB is a reliable technique with very good reported outcomes, as 59/66 cases of PQ to ECRB regained MRC grade 4 wrist extension strength. This technique is much more widely reported compared to FDS to ECRB, although outcomes with FDS to ECRB are similar.
FCR to PIN
Technique: This technique utilizes a branch of the median nerve, the nerve to Flexor Carpi Radialis (FCR), for reinnervation of the PIN. A proximal, volar forearm incision is made below the antecubital fossa. The FCR branch of the median nerve is identified with nerve stimulation, with stimulation causing wrist flexion. The PIN can then be found by following the radial sensory nerve proximally. Following identification, the PIN is divided proximally, and the FCR is divided distally to allow tension-free coaptation[28].
Outcomes: Previous reports demonstrated good wrist and finger extension results. As described earlier, Mackinnon et al. reported a transfer of FDS to ECRB and FCR to PIN[28]. At 18 months postoperatively, the patient gained MRC grade 4 finger and wrist extension strength[28]. Additionally, García-López et al. reported 6 cases of nerve to the pronator teres (PT) to nerve to ECRL and nerve to FCR to PIN in patients with radial nerve palsy or posterior cord injuries[35]. After 20 months, all patients recovered MRC grade 4 ECRL strength with PT to ECRL transfer. With FCR to PIN transfer, 2/6 recovered MRC grade 3 and 4/6 recovered MRC grade 4 metacarpophalangeal extension and ECU strength. All patients recovered MRC grade 4 thumb extension strength[35]. As previously described, in Bertelli’s case series of 14 patients who received AIN to ECRB and FCR to PIN, 8/14 recovered M4 and 4/14 recovered M3 finger extension, and 13/14 recovered M4 wrist extension. 11/14 recovered full thumb extension[34].
Summary: FCR to PIN has shown good results with 18/21 cases of FCR to PIN recovering MRC grade 4 and 3/21 recovering MRC grade 3 wrist extension strength. In addition to wrist extension, 13/21 recovered MRC grade 4, and 6/21 recovered MRC grade 3 finger extension strength. Finally, 18/21 recovered full thumb extension.
Supinator to PIN
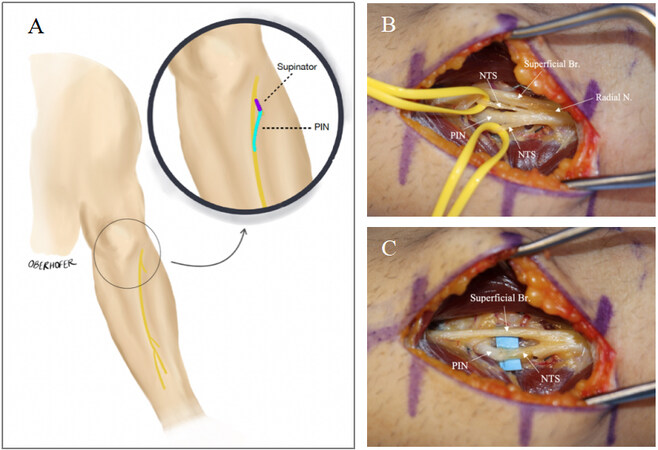
Technique: This technique transfers the nerve to the supinator to the PIN [Figure 4]. On the dorsal side of the arm, an incision is made at the level of the lateral epicondyle between the ECRL and brachioradialis. Careful dissection is essential to preserve branches of the posterior antebrachial cutaneous nerve, which provides sensation to the posterior portion of the forearm[19]. Once the brachioradialis and ECRL are identified, dissection in this interval allows exposure of the superficial branch of the radial nerve, PIN and supinator branches. Alternatively, the supinator can also be exposed by dissecting in the interval between the ECRB and EDC[36]. Finally, a volar approach can also be used, dissecting radially deep to the brachioradialis muscle to expose the branches of the radial nerve. These nerves can be identified by stimulation, with contraction of the supinator confirming its branches and PIN stimulation not causing contraction of the EDC, ECU, EPL and EIP in the setting of injury. The supinator branch is then divided distally, and the PIN is divided proximally to allow for tension-free coaptation[19].
Figure 4. (A) Illustration of relevant anatomy for nerve to supinator to PIN nerve transfer technique. (B) Identification of the nerves prior to transfer: Radial Nerve (Radial N.), Superficial branch of the radial nerve (Superficial Br.), nerve to supinator (NTS), and posterior interosseous nerve (PIN). (C) The donor nerve to the supinator (NTS) is transferred to the recipient PIN.
Outcomes: The supinator to PIN technique is widely documented in the literature and described as the most reliable technique for achieving good outcomes for restoring finger extension[19]. In 2015, Bertelli et al. described a case series of 7 patients and thirteen limbs that received supinator to PIN transfer[37]. After 19 months, 12/13 achieved at least MRC grade 3 thumb and finger extension, with 8 achieving MRC grade 4 thumb extension. The last limb regained MRC grade 2 function[37]. In another series by Bertelli et al., 7 patients with tetraplegia received nerve to the supinator to PIN or gracilis muscle transfer to the extensor compartment of the forearm[38]. After 26 months, 3/3 upper limbs receiving nerve transfer recovered MRC grade 3 thumb and finger extension, compared to none of the patients with gracilis transfer scoring above MRC grade 2[38]. In 2018, Emamhadi et al. described a case report of a patient with tetraplegia after a C6 burst fracture who received brachialis to AIN and supinator to PIN nerve transfer[36]. From the supinator to PIN transfer, the patient achieved MRC grade 3 on thumb extension and MRC grade 4 on finger extension. The patient also achieved MRC grade 4 on thumb and finger flexion with brachialis to AIN transfer[36]. In another investigation, van Zyl et al. reported a case series of 16 participants with spinal cord injury and 59 total nerve transfers[39]. Of the nerve transfers, supinator to PIN had the highest-rated satisfaction. In these cases, 19/21 limbs receiving supinator to PIN nerve transfers achieved MRC grade 3 or higher finger extension, and 17/21 achieved at least MRC grade 3 thumb extension at 24 months follow-up[39]. Khalifeh et al. reported worse outcomes in a case series of 17 participants and 42 nerve transfers after spinal cord injury[40]. Thirteen out of forty-two nerve transfers were supinator to PIN, and only 7/13 achieved MRC grade 3 or higher finger extension[40], although this could be attributed to the longer delay from the time of injury to surgery[19]. Finally, Souza et al. reported a case series of 11 patients with lower brachial plexus injuries who received brachialis to AIN and supinator to PIN within 13 months of injury[22]. After 12 to 24 months postoperatively, 8/11 patients achieved MRC grade 3 or better finger extension with supinator transfer and finger flexion with brachialis transfer. There was no significant loss in donor site function[22].
Summary: Supinator to PIN is one of the most widely documented upper extremity nerve transfer techniques and has shown good outcomes, with 50/62 cases achieving at least MRC grade 3 finger extension and 33/38 achieving at least MRC grade 3 thumb extension. Although results are similar to FCR to PIN, this technique is more widely documented and has been reported to have very high patient satisfaction scores.
CONCLUSION
Distal nerve transfer techniques provide new options to restore function after median and radial nerve injuries. The ECRB to AIN nerve transfer has shown the most promising results for restoration of finger flexion, with all cases examined recovering MRC grade 4 finger flexion.
Regarding transfer techniques for radial nerve injuries, supinator to PIN is a well-documented method for reinnervation of finger and thumb extension. Moreover, it is often used in conjunction with other nerve transfer techniques for finger flexion restoration in the cases of spinal cord or brachial plexus injuries. Although FCR to PIN has shown promising outcomes, there have been few descriptions of this technique published in the past few years, partially due to the popularity of the supinator to PIN. The FCR to PIN nerve transfer does have a role in proximal radial nerve injuries where the supinator branches are not available as an expendable donor nerve. For wrist extension, both FDS to ECRB and distal AIN (PQ) to ECRB can be used to regain MRC grade 4 extension. The choice of donor nerve will depend on the mechanism of injury.
Although specific nerve transfer methods may be reported more than others, it is essential to remember that the optimal donor nerve may vary in different patients. Available donor nerves would vary in patients with tetraplegia, brachial plexus injury or proximal median or ulnar nerve injury. Nerve transfers should be tailored to the requirements of the patient to potentially achieve the best possible outcome. While physical examination remains the most important method for determining the candidacy of donor nerves, imaging techniques such as magnetic resonance imaging may have a role in decision making[18].
CLINICAL RECOMMENDATIONS
Median and radial nerve transfers are reliable methods for upper extremity reanimation after nerve injury. The authors recommend that decisions on the technique used for nerve transfer should be made on a case-by-case basis depending on injury patterns and available donor nerves. Based on the comprehensive review, if there are multiple nerve transfer options for reinnervation of the AIN, we recommend ECRB to AIN as it has better overall reported outcomes compared to brachialis to AIN. Supinator to AIN is another option, but only a few cases have been published.
For wrist and finger extension restoration, we recommend supinator to PIN as a well-documented, reliable method with good results and high overall reported patient satisfaction. FCR to PIN also has good results. We believe it can still be considered when supinator to PIN is not possible with proximal radial nerve injuries or when imaging or physical exam suggests FCR to be a better donor nerve. When only wrist extension restoration is indicated, we recommend distal AIN (PQ) to ECRB as it has good outcomes and is more widely reported than FDS to ECRB, although the latter has good outcomes as well.
Lastly, we reiterate that our clinical recommendations are based on the current documented outcomes in the literature and may evolve as more cases are reported. We recommend that the final decision for choosing a nerve transfer technique should be based on the clinician’s best judgment by utilizing physical exam and imaging to choose a donor nerve that allows a technically feasible dissection and coaptation with the highest return to function and least donor site morbidity.
DECLARATIONS
Authors’ contributionsMade substantial contributions to the article including data curation, background research, interpretation, writing, editing, and revising: Bryan J
Made substantial contributions to writing, editing, and revising: Nichols DS
Contributed to writing, editing, and background research: Polansky C, Cox E
Contributed to writing, editing, and figure illustration: Oberhofer Barker H
Contributed to writing, editing, and project idea: Sullivan B, Chim H
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Midha R, Grochmal J. Surgery for nerve injury: current and future perspectives. J Neurosurg 2019;130:675-85.
2. Domeshek LF, Novak CB, Patterson JMM, et al. Nerve transfers - a paradigm shift in the reconstructive ladder. Plast Reconstr Surg Glob Open 2019;7:e2290.
3. Menorca RM, Fussell TS, Elfar JC. Nerve physiology: mechanisms of injury and recovery. Hand Clin 2013;29:317-30.
4. Jonsson S, Wiberg R, McGrath AM, et al. Effect of delayed peripheral nerve repair on nerve regeneration, Schwann cell function and target muscle recovery. PLoS One 2013;8:e56484.
5. MacKay BJ, Cox CT, Valerio IL, et al. Evidence-based approach to timing of nerve surgery: a review. Ann Plast Surg 2021;87:e1-e21.
6. Grinsell D, Keating CP. Peripheral nerve reconstruction after injury: a review of clinical and experimental therapies. Biomed Res Int 2014;2014:698256.
7. Sullivan R, Dailey T, Duncan K, Abel N, Borlongan CV. Peripheral nerve injury: stem cell therapy and peripheral nerve transfer. Int J Mol Sci 2016;17:2101.
8. Chhabra A, Ahlawat S, Belzberg A, Andreseik G. Peripheral nerve injury grading simplified on MR neurography: as referenced to Seddon and Sunderland classifications. Indian J Radiol Imaging 2014;24:217-24.
9. Aljawder A, Faqi MK, Mohamed A, Alkhalifa F. Anterior interosseous nerve syndrome diagnosis and intraoperative findings: a case report. Int J Surg Case Rep 2016;21:44-7.
10. Caetano EB, Vieira LA, Sabongi Neto JJ, Caetano MBF, Sabongi RG. Anterior interosseous nerve: anatomical study and clinical implications. Rev Bras Ortop 2018;53:575-81.
11. Rao YL, Pai MM, Krishnaprasad PR, Murlimanju BV, Mamatha T, Prabhu LV. Exploring the morphology of anterior interosseous nerve and relating it to its clinical conditions. Turk Neurosurg 2021;31:107-11.
12. Kesserwani H. Pseudo-anterior interosseus nerve syndrome: a case report and a review of clinical signs, pathology and functional anatomy of the precision grip. Cureus 2021;13:e15180.
13. Wertheimer A, Kiel J. Anatomy, shoulder and upper limb, forearm anterior interosseous nerve. StatPearls: StatPearls Publishing LLC.; 2022.
14. Latef TJ, Bilal M, Vetter M, Iwanaga J, Oskouian RJ, Tubbs RS. Injury of the radial nerve in the arm: a review. Cureus 2018;10:e2199.
15. Prasartritha T, Liupolvanish P, Rojanakit A. A study of the posterior interosseous nerve (PIN) and the radial tunnel in 30 Thai cadavers. J Hand Surg 1993;18:107-12.
16. Nayak SR, Ramanathan L, Krishnamurthy A, et al. Extensor carpi radialis brevis origin, nerve supply and its role in lateral epicondylitis. Surg Radiol Anat 2010;32:207-11.
17. Wheeler R, DeCastro A. Posterior interosseous nerve syndrome. StatPearls: StatPearls Publishing LLC.; 2022.
18. Hawasli AH, Chang J, Reynolds MR, Ray WZ. Transfer of the brachialis to the anterior interosseous nerve as a treatment strategy for cervical spinal cord injury: technical note. Global Spine J 2015;5:110-7.
19. Bazarek S, Sten M, Nin D, Brown JM. Supinator to posterior interosseous nerve transfer for restoration of finger extension. Oper Neurosurg (Hagerstown) 2021;21:E408-13.
20. Mackinnon SE, Yee A, Ray WZ. Nerve transfers for the restoration of hand function after spinal cord injury. J Neurosurg 2012;117:176-85.
21. Ray WZ, Yarbrough CK, Yee A, Mackinnon SE. Clinical outcomes following brachialis to anterior interosseous nerve transfers. J Neurosurg 2012;117:604-9.
22. Souza FH, Bernardino SN, Junior ABC, et al. Nerves transfers for functional hand recovery in traumatic lower brachial plexopathy. Surg Neurol Int 2020;11:358.
23. Bertelli JA. Transfer of the radial nerve branch to the extensor carpi radialis brevis to the anterior interosseous nerve to reconstruct thumb and finger flexion. J Hand Surg Am 2015;40:323-328.e2.
24. Bertelli JA, Ghizoni MF. Nerve transfers for restoration of finger flexion in patients with tetraplegia. J Neurosurg Spine 2017;26:55-61.
25. Salomão R, de Oliveira JP, Junger CF, Soares Ricardo LC, de Lima CR, Acioly MA. Delayed transfer of the extensor carpi radialis brevis branch of the radial nerve to the anterior interosseous nerve for restoration of thumb and index finger flexion: case report. J Neurol Surg A Cent Eur Neurosurg 2020;81:571-4.
26. Hsiao EC, Fox IK, Tung TH, Mackinnon SE. Motor nerve transfers to restore extrinsic median nerve function: case report. Hand (N Y) 2009;4:92-7.
27. Murphy RK, Ray WZ, Mackinnon SE. Repair of a median nerve transection injury using multiple nerve transfers, with long-term functional recovery. J Neurosurg 2012;117:886-9.
28. Mackinnon SE, Roque B, Tung TH. Median to radial nerve transfer for treatment of radial nerve palsy. Case report. J Neurosurg 2007;107:666-71.
29. Bertelli JA, Ghizoni MF. Results of nerve grafting in radial nerve injuries occurring proximal to the humerus, including those within the posterior cord. J Neurosurg 2016;2016:179-85.
30. Ukrit A, Leechavengvongs S, Malungpaishrope K, Uerpairojkit C, Chongthammakun S, Witoonchart K. Nerve transfer for wrist extension using nerve to flexor digitorum superficialis in cervical 5, 6, and 7 root avulsions: anatomic study and report of two cases. J Hand Surg Am 2009;34:1659-66.
31. Bertelli JA, Tacca CP, Winkelmann Duarte EC, Ghizoni MF, Duarte H. Transfer of the pronator quadratus motor branch for wrist extension reconstruction in brachial plexus palsy. Plast Reconstr Surg 2012;130:1269-78.
32. Bertelli JA, Ghizoni MF, Tacca CP. Results of wrist extension reconstruction in C5-8 brachial plexus palsy by transferring the pronator quadratus motor branch to the extensor carpi radialis brevis muscle. J Neurosurg 2016;124:1442-9.
33. Bhatia A, Salama M. Pronator quadratus to extensor carpi radialis brevis nerve transfer in C5-C7 or C5-C8 brachial plexus injuries for independent wrist extension. Indian J Plast Surg 2020;53:36-41.
34. Bertelli JA. Nerve versus tendon transfer for radial nerve paralysis reconstruction. J Hand Surg Am 2020;45:418-26.
35. García-López A, Navarro R, Martinez F, Rojas A. Nerve transfers from branches to the flexor carpi radialis and pronator teres to reconstruct the radial nerve. J Hand Surg Am 2014;39:50-6.
36. Emamhadi M, Andalib S. Double nerve transfer for restoration of hand grasp and release in C7 tetraplegia following complete cervical spinal cord injury. Acta Neurochir (Wien) 2018;160:2219-24.
37. Bertelli JA, Ghizoni MF. Nerve transfers for elbow and finger extension reconstruction in midcervical spinal cord injuries. J Neurosurg 2015;122:121-7.
38. Bertelli JA, Ghizoni MF. Nerve and free gracilis muscle transfers for thumb and finger extension reconstruction in long-standing tetraplegia. J Hand Surg Am 2016;41:e411-6.
39. van Zyl N, Hill B, Cooper C, Hahn J, Galea MP. Expanding traditional tendon-based techniques with nerve transfers for the restoration of upper limb function in tetraplegia: a prospective case series. Lancet 2019;394:565-75.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Bryan J, Nichols DS, Polansky C, Oberhofer Barker H, Cox E, Sullivan B, Chim H. Distal median and radial nerve branch transfer techniques for upper extremity reanimation. Plast Aesthet Res 2022;9:53. http://dx.doi.org/10.20517/2347-9264.2022.39
AMA Style
Bryan J, Nichols DS, Polansky C, Oberhofer Barker H, Cox E, Sullivan B, Chim H. Distal median and radial nerve branch transfer techniques for upper extremity reanimation. Plastic and Aesthetic Research. 2022; 9: 53. http://dx.doi.org/10.20517/2347-9264.2022.39
Chicago/Turabian Style
Bryan, Jaimie, D. Spencer Nichols, Caroline Polansky, Haley Oberhofer Barker, Elizabeth Cox, Brianne Sullivan, Harvey Chim. 2022. "Distal median and radial nerve branch transfer techniques for upper extremity reanimation" Plastic and Aesthetic Research. 9: 53. http://dx.doi.org/10.20517/2347-9264.2022.39
ACS Style
Bryan, J.; Nichols DS.; Polansky C.; Oberhofer Barker H.; Cox E.; Sullivan B.; Chim H. Distal median and radial nerve branch transfer techniques for upper extremity reanimation. Plast. Aesthet. Res. 2022, 9, 53. http://dx.doi.org/10.20517/2347-9264.2022.39
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 8 clicks
Cite This Article 8 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.