Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality
Abstract
Aim: Skin firmness is one of the key parameters to define skin quality and facial aging. Among the minimally invasive anti-aging strategies, hyaluronic acid (HA) injection is widely accepted to improve skin quality. While most of the available HA injectables are designed and intended for intradermal injection, a novel HA/sorbitol composition containing 2.6% high molecular weight hyaluronic acid stabilized by sorbitol was recently developed to be specifically injected into the adipose tissue to improve the quality of all skin layers, especially the skin firmness.
Methods: The HA/sorbitol composition was investigated versus product comparators in terms of biophysical properties, tolerance in subcutaneous tissue with in vivo implantation study, and skin firmness assessment on human skin explants.
Results: The HA/sorbitol composition was characterized by unique and differentiated biophysical properties, proper distribution and high tolerance of the gel composition into the adipose tissue, and the ability to efficiently improve skin firmness.
Conclusion: The HA/sorbitol composition represents a new attractive solution to treat facial skin aging with an injection strategy specifically targeting the adipose tissue instead of the dermis, to improve the quality of the skin.
Keywords
INTRODUCTION
Skin aging is a complex and unavoidable biological phenomenon that starts in the third decade of life and is determined by intrinsic (chronological, hormonal, and genetic) and extrinsic factors (sun exposure, cigarette smoking, dietary habits, alcohol intake, pollution, and weather environment)[1]. It is a dynamic process that results in structural alterations of soft and bony tissues with modifications of various constituent layers from skin surface to subcutaneous adipose tissue[1-3]. Skin quality is important to human attractiveness and significantly influences the perception of age, health, and youth across ethnicities[4]. Minimally invasive procedures have revolutionized the treatment paradigm for both facial and body skin rejuvenation and the recent history of cosmetic surgery[5]. In these procedures, the demand for treatments with hyaluronic acid (HA) injectables is constantly on the rise, and a large consensus has been gained worldwide for these products in aesthetic medicine and dermatology[4-6]. This market demand has driven the research and development of new advanced HA formulations for different clinical indications and applications while offering improved safety, effectiveness, and patient satisfaction. In this context, a novel 2.6% H-HA/3.2% sorbitol composition [Figure 1] containing 52 mg of stabilized high molecular weight
The purpose of this article is to investigate this novel 2.6% H-HA/3.2% sorbitol composition based on the following:
- We studied its key biophysical properties of cohesivity, elasticity, and viscosity.
- We studied its tissue tolerance and safety by implantation in the subcutaneous adipose tissue with an in vivo animal model.
- We studied its performance by assessment of the firmness of human skin explants cultured with CO2 humid incubator after implantation into deep skin tissues.
METHODS
Materials
The 2.6% H-HA/3.2% sorbitol composition (Kylane Laboratoires, Switzerland) was compared with 2 commercial injectable products intended for skin quality improvement:
- 1.6% L-HA + 1.6% H-HA (Profhilo, IBSA, Italy) is a mixture of low molecular weight HA (0.08-0.10 million Daltons) at 16 mg/mL and high molecular weight HA (1.10-1.40 million Daltons) at 16 mg/mL into buffered sodium chloride solution[12], indicated to be injected in the dermal/subcutaneous levels. This product was used as a comparator for the cohesivity, oscillatory shear stress, and flow tests and the ex vivo study.
- VYC-12L (Juvéderm Volite, Allergan, France) is a composition of 12 mg/mL of crosslinked hyaluronic acid with 3 mg/mL of lidocaine hydrochloride in phosphate-buffered solution indicated for intradermal injections only[13]. This product was used as a comparator for the cohesivity, oscillatory shear stress, and flow tests.
Cohesivity test
The cohesivity test was performed according to the method described in the article by Sundaram et al.[14]. In agreement with this article, the assessment of the cohesivity for each tested product was ranked according to the Gavard-Sundaram Cohesivity Scale, with cohesivity scores from 1 (fully dispersed) to 5 (fully cohesive).
Oscillatory shear stress test
The oscillatory shear stress test measures the elastic modulus G’. It was performed using a TA Instrument cone-plate rheometer at a temperature of 25 °C in shear-stress oscillation mode at 1.0% of strain, within the linear viscoelastic region, with a cone/plate aluminum geometry of 40 mm, 2°, and a 50 μm gap between the cone and the plate of the rheometer. The measurements were carried out in a frequency range of 0.1-5 Hz. The value of the elastic modulus G’ was measured at the physiological oscillation frequency of 1 Hz.
Flow test
The flow test measures the viscosity η of the gel. It was performed using a TA Instrument cone-plate rheometer at a temperature of 25 °C at 0.001 to 1000 s-1 shear rate with a cone/plate aluminum geometry of 40 mm, 2°, and a 50 μm gap between the cone and the plate of the rheometer. The value of the viscosity η was measured at the shear rate of 0.02 s-1.
In vivo implantation study with animal model
The in vivo study evaluated the local tissue effects of the 2.6% H-HA/3.2% sorbitol composition after subcutaneous administration. The study was conducted under international standards [International Standards Organization (ISO) 10993] as mandated by regulatory agencies in an AAALAC international accredited facility registered with the French Department of Agriculture for animal housing, care, and investigations. In this study, the gel composition was injected into the subcutaneous tissue of 15 rats with a follow-up period of 28 days. The 15 rats each received four subcutaneous injections of the gel composition and 2 subcutaneous implantations of negative controls. The negative control was high-density polyethylene (HDPE) (CAS number: 9002-88-4), as recommended in the ISO 10993 Standard, Part 12. The injections were performed by a qualified and trained operator using standard aseptic techniques. Six sites on the back of each rat were injected subcutaneously. The gel composition was injected with a needle with a volume of 200 µL. The negative control was implanted with a trocar inserted through the skin, perpendicular to the vertebral column, deep enough into the subcutaneous tissue to accommodate the negative control article and ensure that the implant was far from the implantation entry point. The sites were appropriately spaced to avoid merging them as much as possible. Immediately after the injections, the sites were peripherally marked with a surgical skin marker. The animals were observed daily for general health and to detect mortality and morbidity. In addition, a detailed clinical examination of the animals was conducted at least twice a month. This examination included but was not limited to general conditions, behavior and activity, locomotion, and posture. Marks were checked regularly. When necessary, the back of the rats was shaved and the marks were renewed. The injection sites were macroscopically examined at 6 h, 2 days, and 28 days. Any gross changes in tissues around each injection were recorded using the following parameters: size, shape, color, consistency, distribution, and any other observations, as appropriate. When visible, the properties of the injections (location and coloration) were described. Tissues at the injection sites and the surrounding tissues were harvested and fixed in 10% NBF (Carlo Erba) for histopathologic analysis. In addition, the draining lymph nodes (axillary and inguinal, four per animal) were macroscopically examined, sampled, and fixed in 10% NBF (Carlo Erba) for histopathologic analysis. As lymph nodes are organized in clusters, one to several lymph nodes could be collected at each sampling. After complete fixation in 10% NBF (Carlo Erba), the injected sites were dehydrated in alcohol solutions of increasing concentration, cleared in xylene, and embedded in paraffin. For all injected sites, one central transversal slide was cut with a microtome (4 um thickness). All created slides were stained with saffron-hematoxylin-eosin (SHE). The histopathologic qualitative and semi-quantitative analyses were conducted in accordance with the OECD Good Laboratory Practice regulations, ENV/MC/CHEM (98) 17 and the United States Food and Drug Administration Good Laboratory Practice regulations, 21 CFR 58. Qualitative and semi-quantitative histopathologic evaluations of the local tissue effects at the injection sites were conducted according to the standard ISO 10993, Part 6, by a pathologist (reactivity ranking calculations based on the scoring related to the presence of macrophages, lymphocytes, plasma cells, polymorphonuclear cells, giant cells, necrosis, fibrosis, neovascularization, fatty infiltrate, fibrin, hemorrhage, cell or tissue degeneration, and fibroplasia). The gel composition was evaluated and compared to the negative control articles.
Ex vivo study of the firmness of human skin explants cultured with CO2 humid incubator
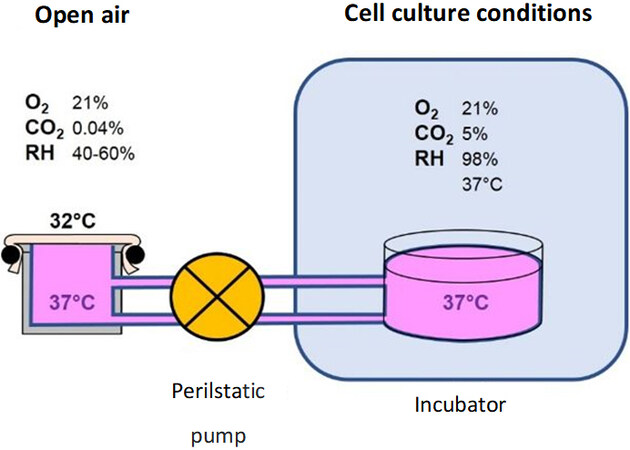
This study was performed on biopsy, obtained from surgical residues after written informed consent from the donor, in full compliance with the Declaration of Helsinki and article L.1243-4 of the French Public Health Code. The latter does not require any prior authorization by an ethics committee for sampling and using surgical waste. Six human skin explants with a round shape and a diameter of 38 mm were placed on a support, on which the skin was stretched [Figure 2]. These skin explants were obtained from an abdominoplasty (discarded abdominoplasty tissue) of a 44-year-old Caucasian woman with phototype II according to the Fitzpatrick classification. The supports were put in a tank containing a culture medium [composition: glucose (3.65 g/L), L-glutamine (0.66 g/L), HEPES (26 mM), Phenol Red (0.300 mg/L), fetal bovine serum (5%), fungizone (2.5 μg/mL), and gentamicin (50 μg/mL)] that was linked to a CO2 humid incubator at 37 °C, 21% O2, 5% CO2, and 98% relative humidity by a peristaltic pump [Figure 2].
Figure 2. Schematic representation of the ex vivo study method with skin explants cultured with CO2 humid incubator.
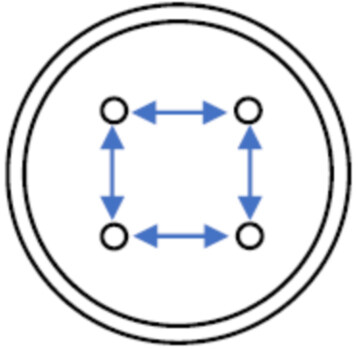
On Day 0 (D0), 2 skin explants were injected with the 2.6% H-HA/3.2% sorbitol composition, 2 skin explants were injected with the 1.6% L-HA + 1.6% H-HA composition, and 2 skin explants were not treated (reference). All injections were performed in the deep skin tissues of each skin explant with four injection sites of 25 µL per explant, forming a square with injection sites spaced 8 mm [Figure 3]. A cutometer study with measurements of the skin firmness R0 in the middle of the square formed by the four injection sites was then carried out with a cutometer (MPA580, Courage & Khazaka) at different survival times for all the skin explants: Day 0 (before and after injection, D0), Day 1 (D1), Day 2 (D2), and Day 5 (D5). The measuring principle of the cutometer is based on the suction method, where negative pressure deforms the skin explant mechanically. The pressure is created in the device and draws the skin into the aperture of the probe, and, after a defined time, releases it again. The resistance of the skin explants to the negative pressure is defined as the skin firmness R0, and the lower the R0, the firmer the skin.
RESULTS
Measurement of cohesivity
The assessment of the cohesivity for the 3 tested products ranked according to the Gavard-Sundaram Cohesivity Scale, is described in Table 1, and pictures of cohesivity for the 2.6% H-HA/3.2% sorbitol composition are provided in Figure 4.
Scores of cohesivity for the three tested products
| Tested HA composition | Gavard Sundaram Cohesivity Scale (1-5) | ||
| 15 Sec | 70 Sec | 95 Sec | |
| 2.6% H-HA/3.2% sorbitol | 5 | 5 | 5 |
| 1.6% L-HA + 1.6% H-HA | 5 | 4 | 4 |
| VYC-12L | 3 | 1 | 1 |
Measurement of elastic modulus G’ and viscosity η
The measured values of elastic modulus G’ and viscosity η are summarized for the 3 tested products in Table 2.
Elastic modulus G’ and viscosity η for the three tested products
| Tested HA composition | Elastic modulus G’ in shear-stress at 1 Hz (Pa) | Viscosity η at 0,02 s-1 (Pa.s) |
| 2.6% H-HA/3.2% sorbitol | 529 | 856 |
| 1.6% L-HA + 1.6% H-HA | 47 | 31 |
| VYC-12L | 142 | 281 |
Investigation of the tolerance by implantation with in vivo animal model
All animals appeared clinically normal at the beginning and throughout this study. All rats gained weight as expected during the study.
Macroscopically, no major findings were observed in the surrounding tissues, and no major abnormality was observed in the draining lymph nodes during the study. A slight redness was observed at 6 h for the gel composition. Slight to moderate redness was observed at 6 h and 2 days for the negative control articles. At 6 h, the gel composition was visible and palpable, with a round shape. At 2 days, the gel composition was visible and palpable but with a softer consistency than at 6 h. The bleb was very dispersed and slightly bulging for all gel composition sites. At 28 days, the gel composition was not visible and not palpable. Therefore, it is hypothesized that the gel composition was spread out around the injection point throughout the study and became non-palpable in the injected area. The negative control was visible and palpable for the whole duration of the study.
Microscopically, at 28 days, the sites injected with the gel composition showed slight local tissue effects that were slightly lower than those observed for the implanted negative control. The inflammatory reaction was slight for the gel composition and negative for the control. Under the conditions of the study, the average score of the changes seen in the gel composition injected sites resulted in a reaction described as “null to minimal” when compared to the reactivity ranking scores of the negative control according to the reactivity ranking calculations of the pathologist.
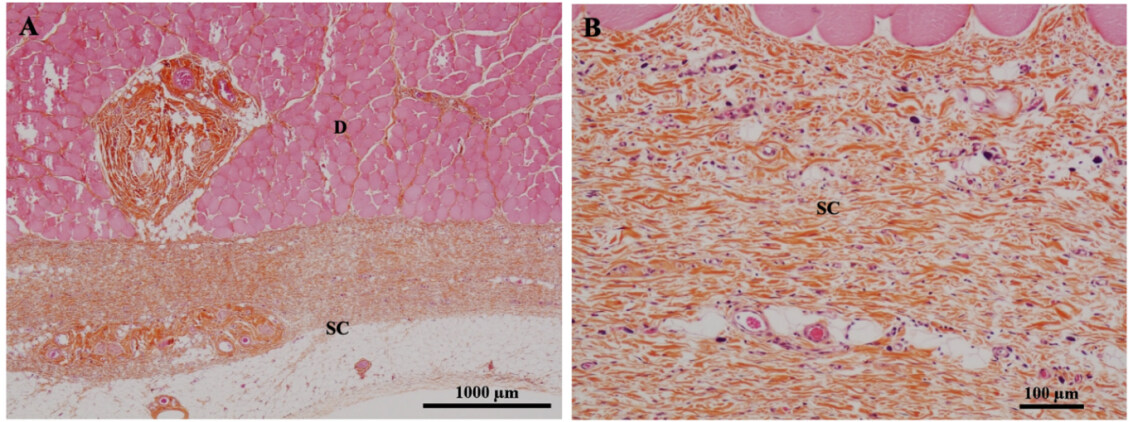
Figure 5 provides histopathologic photomicrographs of the injected area at 28 days after subcutaneous injection of the 2.6% H-HA/3.2% sorbitol composition, highlighting a proper and homogeneous anatomy of the adipose tissue in the injected area following the injection of the product.
Investigation of the performance of the 2.6% H-HA/3.2% sorbitol composition on the skin firmness by ex vivo human skin explant model
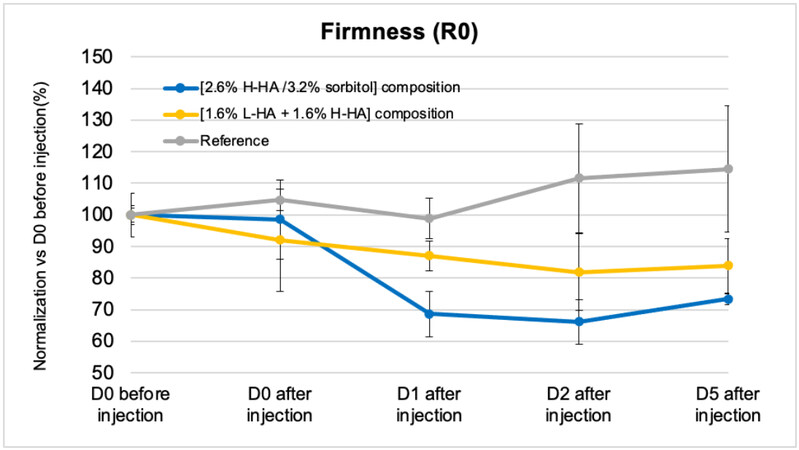
Cutometer study results of the skin firmness [Figure 6] demonstrate that the firmness (R0) of the reference skin explants (without any treatment) was quite stable over the five days of the study. On the contrary, the skin firmness increased significantly (i.e., R0 decreases) for the skin explants injected with both the 2.6%
DISCUSSION
Human skin is composed of 3 distinct layers, namely epidermis, dermis, and hypodermis, each with varying degrees of specialization. The epidermis and dermis are well characterized, but very little attention has been given to the hypodermis/subcutaneous tissue[15] despite its key role in facial aging[3].
While most skin quality boosters based on hyaluronic acid available on the market are designed for intradermal applications, the novel 2.6% H-HA/3.2% sorbitol injectable composition was specifically designed with tailored composition and biophysical features to be implanted into the subcutaneous tissue, with the aim to develop new treatment strategies specifically focused on the adipose tissue, especially the superficial adipose tissue, to significantly influence and improve the mechanical properties of the skin and treat skin aging.
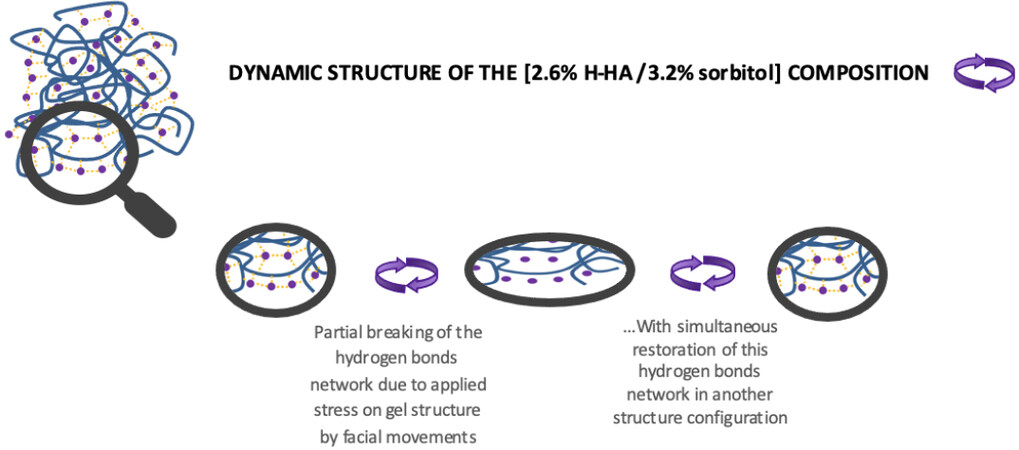
The 2.6% H-HA/3.2% sorbitol product is characterized by a composition of a high content of pure hyaluronic acid with high molecular weight stabilized with sorbitol, which creates a very dense network of hydrogen bonds within the matrix of the sorbitol, hyaluronic acid, and water molecules [Figure 1]. This capacity of sorbitol to form strong and extensive hydrogen bonds capable of forming water and hyaluronic acid is evidenced in the literature[16,17], notably through measurements of thermodynamic properties. This fact is consistent with the high stability observed with injectable hyaluronic acid and sorbitol products; for example, ophthalmic viscoelastic devices (OVD) containing hyaluronic acid and sorbitol are reported to benefit from better maintained viscoelastic properties versus injectable products without sorbitol, in the case of thermal treatment, light irradiation, or storage of the device at 25 °C[8]. On the other hand, cohesivity results of the 2.6% H-HA/3.2% sorbitol composition versus the non-crosslinked 1.6% L-HA + 1.6% H-HA composition and the crosslinked VYC-12L product demonstrate the high affinity which exists among the molecules of sorbitol, HA, and water within the gel. This offers a significantly higher cohesivity than the 2 tested products used as comparators in this article, independently of their HA concentrations, i.e.,
Figure 7. Schematic representation of the dynamic structure of the 2.6% H-HA/3.2% sorbitol composition.
The measurements of the elastic modulus G’ and the viscosity η of the 2.6% H-HA/3.2% sorbitol composition highlight the fact that the HA/sorbitol stabilization provides very high elasticity and viscosity, significantly higher than the non-crosslinked 1.6% L-HA + 1.6% H-HA composition and the crosslinked VYC-12L product. These unique rheological features, combined with the high cohesivity, are essential biophysical properties for the composition to provide elasticity to the skin tissues that need to be treated for their laxity and loss of elasticity, and combined with a mild filler to fill the soft tissues. Even though the 2.6% H-HA/3.2% sorbitol composition is not crosslinked, its elastic modulus G’ is higher than most other HA fillers, for example, the leading VYC-20L volumizer (Juvéderm Voluma, Allergan, France) with its G’ of
Figure 8. Photographs of 0.25 mL of 2.6% H-HA/3.2% sorbitol gel composition and VYC-20L HA filler after deposit on a glass slide
The in vivo animal study with 15 rats demonstrated a high tolerance for the proper distribution of the viscoelastic 2.6% H-HA/3.2% sorbitol gel into the adipose tissue. It confirmed that the unique and differentiated composition and biophysical properties of the HA/sorbitol formulation specifically designed to be injected into the subcutaneous tissue, especially the superficial adipose tissue, ensure a high tolerance for the treatment to improve the skin quality, in line with the positive background of HA in dermatology and medical aesthetics over the last two decades. The outcome of this study is also consistent with the safety demonstration of the use of injectable HA/sorbitol viscoelastic formulations in other medical fields, such as in ophthalmology with OVD or rheumatology with intra-articular products for the treatment of osteoarthrosis[8-11].
The ex vivo study by cutometer follow-up of the treatment with 2.6% H-HA/3.2% sorbitol composition of human skin explants that were cultured with CO2 humid incubator for five days demonstrated that the hydrogel complex of high molecular weight hyaluronic acid and sorbitol efficiently improves the skin firmness over the entire duration of the investigation after implantation. This outcome is consistent with the biophysical properties of high cohesivity, elasticity, and viscosity of the product, showing that the 2.6%
Therefore, as part of the currently available solutions in the market to improve the skin quality in facial aging, the injectable 2.6% H-HA/3.2% sorbitol composition is demonstrated to be a new relevant treatment alternative with respect to the following:
- Current injectable skin quality booster products based on hyaluronic acid are designed for intradermal applications and not specifically for the treatment of the subcutaneous adipose tissue.
- Currently available solutions usually target the treatment of adipose tissue firmness, such as fat grafting or thread lifts. These solutions are usually significantly more invasive and complex in their administration protocol and require a longer recovery time; thus, they do not meet the wishes of all patients and physicians.
In conclusion, the 2.6% H-HA/3.2% sorbitol composition represents a novel, attractive solution to form the skin matrix and improve the skin quality in facial aging by focusing the treatment on the subcutaneous adipose tissue, with benefits not only at the level of the subcutaneous tissue but also more comprehensively in all skin layers, notably in the dermal layer. Additional evidence by investigation of the improvement in skin quality biological markers will be obtained to offer further insight into the treatment. The objective of this new investigation will be to study the biological effects on the dermal layer skin quality after the injection of the 2.6% H-HA/3.2% sorbitol composition in the adipose tissue. In addition, a clinical study of the 2.6% H-HA/3.2% sorbitol composition in facial treatment will be completed to support and evidence the high product tolerance and performance to improve skin quality and ensure patient satisfaction.
DECLARATIONS
Authors’ contributionsObtained all the experimental data: Gavard Molliard S, Bon Bétemps J
All authors contributed to design of the concept and writing of the article.
Availability of data and materialsExperimental data were obtained by Kylane Laboratoires SA.
Financial support and sponsorshipKylane Laboratoires SA provided the logistical and financial support for the execution of this study.
Conflicts of interestGavard Molliard S, Bon Bétemps J, Hadjab B are employed by Kylane Laboratoires SA. Other authors declared that there are no conflicts of interest.
Ethical approval and consent to participateThe project authorization number from the Ethical Committee (France) associated with the in vivo animal study is: APAFIS#23401-2017050517312149 v17.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Sparavigna A. Role of the extracellular matrix in skin aging and dedicated treatment - state of the art. Plast Aesthet Res 2020;7:14.
2. Wan D, Amirlak B, Rohrich R, Davis K. The clinical importance of the fat compartments in midfacial aging. Plast Reconstr Surg Glob Open 2013;1:e92.
3. Wollina U, Wetzker R, Abdel-Naser MB, Kruglikov IL. Role of adipose tissue in facial aging. Clin Interv Aging 2017;12:2069-76.
4. Goldie K, Kerscher M, Fabi SG, et al. Skin quality - a holistic 360° view: consensus results. Clin Cosmet Investig Dermatol 2021;14:643-54.
5. Cassuto D, Bellia G, Schiraldi C. An overview of soft tissue fillers for cosmetic dermatology: from filling to regenerative medicine. Clin Cosmet Investig Dermatol 2021;14:1857-66.
6. Kleine-Börger L, Meyer R, Kalies A, Kerscher M. Approach to differentiate between hyaluronic acid skin quality boosters and fillers based on their physicochemical properties. J Cosmet Dermatol 2022;21:149-57.
7. Rowe RC, Sheskey PJ, Owen SC. Handbook of pharmaceutical excipients, 5th Edition. United Kingdom: American Pharmacists Association; 2006. Available from: https://www.academia.edu/33834163/Handbook_of_Pharmaceutical_Excipients_Fifth_Edition [Last accessed on 8 Sep 2022]
8. Nogami E, Watanabe I, Hoshi H, et al. D-sorbitol can keep the viscosity of dispersive ophthalmic viscosurgical device at room temperature for long term. Sci Rep 2019;9:16815.
9. Cortet B, Lombion S, Naissant B, Vidovic E, Bruyère O. Non-inferiority of a single injection of sodium hyaluronate plus sorbitol to hylan G-F20: a 6-month randomized controlled trial. Adv Ther 2021;38:2271-83.
10. Heisel J, Kipshoven C. Safety and efficacy findings from a non-interventional study of a new hyaluronic acid/sorbitol formulation (GO-ON® matrix) for intra-articular injection to relieve pain and disability in osteoarthritis patients. Drug Res (Stuttg) 2013;63:445-9.
11. Conrozier T. Viscosupplémentation par acide hyaluronique, associé au mannitol ou au sorbitol. Rhumatos 2017. Available from: https://rhumatos.fr/viscosupplementation-par-acide-hyaluronique-associe-au-mannitol-ou-au-sorbitol/ [Last accessed on 8 Sep 2022]
12. Cassuto D, Delledonne M, Zaccaria G, Illiano I, Giori AM, Bellia G. Safety assessment of high- and low-molecular-weight hyaluronans (Profhilo®) as derived from worldwide postmarketing data. Biomed Res Int 2020;2020:8159047.
13. Safa M, Natalizio A, Hee CK. A prospective, open-label study to evaluate the impact of VYC-12L injection on skin quality attributes in healthy volunteers. CCID 2022;15:411-26.
14. Sundaram H, Rohrich RJ, Liew S, et al. Cohesivity of hyaluronic acid fillers: development and clinical implications of a novel assay, pilot validation with a five-point grading scale, and evaluation of six U.S. food and drug administration-approved fillers. Plast Reconstr Surg 2015;136:678-86.
15. Wong R, Geyer S, Weninger W, Guimberteau JC, Wong JK. The dynamic anatomy and patterning of skin. Exp Dermatol 2016;25:92-8.
16. Lerbret A, Mason PE, Venable RM, et al. Molecular dynamics studies of the conformation of sorbitol. Carbohydr Res 2009;344:222935.
17. Kaushik JK, Bhat R. Thermal stability of proteins in aqueous polyol solutions: role of the surface tension of water in the stabilizing effect of polyols. J Phys Chem B 1998;102:7058-66.
18. Gavard Molliard S, Bon Bétemps J, Hadjab B, Topchian D, Micheels P, Salomon D. Key rheological properties of hyaluronic acid fillers: from tissue integration to product degradation. Plast Aesthet Res 2018;5:17.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Gavard Molliard S, Bon Bétemps J, Hadjab B, Ghazal A, Badi M, Cerrano M. Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality. Plast Aesthet Res 2022;9:52. http://dx.doi.org/10.20517/2347-9264.2022.60
AMA Style
Gavard Molliard S, Bon Bétemps J, Hadjab B, Ghazal A, Badi M, Cerrano M. Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality. Plastic and Aesthetic Research. 2022; 9: 52. http://dx.doi.org/10.20517/2347-9264.2022.60
Chicago/Turabian Style
Gavard Molliard, Samuel, Jérémie Bon Bétemps, Basste Hadjab, Anas Ghazal, Mhd Badi, Marco Cerrano. 2022. "Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality" Plastic and Aesthetic Research. 9: 52. http://dx.doi.org/10.20517/2347-9264.2022.60
ACS Style
Gavard Molliard, S.; Bon Bétemps J.; Hadjab B.; Ghazal A.; Badi M.; Cerrano M. Stabilized composition of 26 mg/mL of high molecular weight HA for subcutaneous injection to improve skin quality. Plast. Aesthet. Res. 2022, 9, 52. http://dx.doi.org/10.20517/2347-9264.2022.60
About This Article
Copyright
Data & Comments
Data

 Cite This Article 11 clicks
Cite This Article 11 clicks













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.