A narrative review of outcomes following metoidioplasty: complications and satisfaction
Abstract
Metoidioplasty consists of lengthening and straightening the hormonally hypertrophied clitoris. The goals of the procedure include masculinizing the external genitalia and enabling standing micturition. Metoidioplasty may be performed as a stand-alone procedure or an interval procedure prior to phalloplasty. While most often performed with urethral lengthening, metoidioplasty may also be performed as a “simple release” (i.e., without urethral lengthening). Secondary procedures typically include scrotoplasty and placement of testicular implants. While satisfaction with this procedure is high, complications can occur. Complications are commonly categorized as either urologic (i.e., strictures and fistula) or wound healing (i.e., wound disruption, infection, bleeding, etc.). This narrative review discusses postoperative outcomes, including both satisfaction and complications. Published data on complications include fistula and stricture rates from 0%-50% and 0%-63%, respectively. Overall satisfaction with appearance ranges from 48%-100%, and patient ability to void while standing ranges from 67%-100%. Metoidioplasty is a safe and effective procedure for transgender men. Further research regarding surgical techniques and outcomes will help reduce complications and improve overall patient satisfaction.
Keywords
INTRODUCTION
For transgender individuals, gender affirmation surgery (GAS) helps to align an individual’s anatomy with their gender identity[1]. In doing so, GAS may reduce and/or alleviate gender dysphoria, improve quality of life, and reduce negative health outcomes[2]. In recent years, an increasing number of individuals have requested gender-affirming surgical interventions[3,4]. For transgender men, options for genital gender-affirming surgery (GGAS) include metoidioplasty and phalloplasty. While the goals of these procedures may be somewhat different, genital surgery is typically performed to masculinize the external genitalia, allow standing micturition, and improve gender congruent sexual activity and function with minimal morbidity[5].
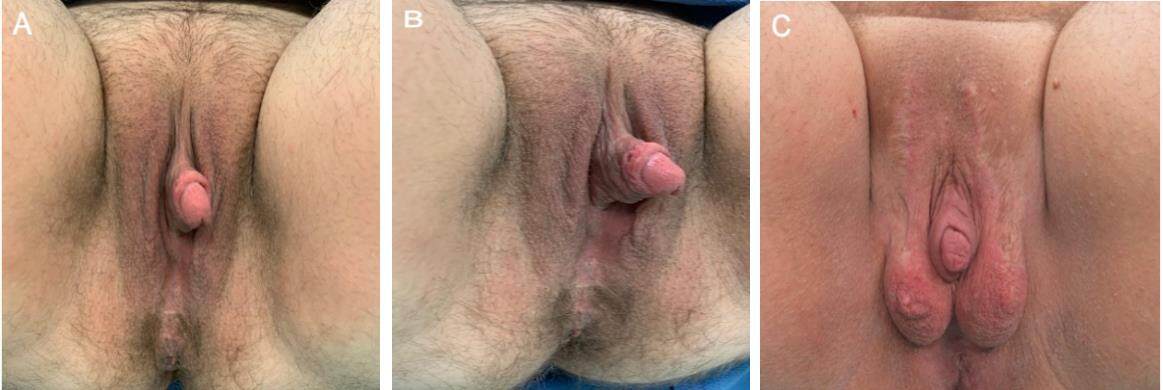
Metoidioplasty involves lengthening the hormonally hypertrophied clitoris and may be performed with or without urethral lengthening (i.e., “simple release”). Metoidioplasty may be performed as an interval procedure prior to phalloplasty for those individuals desiring a larger phallus (whether for appearance, penetration, or standing micturition). Most commonly, hysterectomy with bilateral salpingo-oophorectomy and vaginectomy (colpectomy) with colpocleisis are also performed (either prior to or in conjunction with the metoidioplasty). Scrotoplasty and placement of testicular implants may be performed at the time of metoidioplasty or as a secondary procedure[6] [Figure 1]. Metoidioplasty constructs a phallus typically between 5-9 cm, preserves tactile and erogenous genital sensation, and avoids the donor site morbidity and complexity associated with a phalloplasty. While some individuals report the ability to engage in penetrative intercourse, these reports are largely anecdotal[7].
Figure 1. Postoperative images of staged metoidioplasty. (A and B) First stage metoidioplasty with colpectomy and colpocleisis. (C) Second stage scrotoplasty with the placement of scrotal implants.
While metoidioplasty is a reliable procedure with less technical complexity as compared to a phalloplasty, complications may occur. This review investigates metoidioplasty outcomes with a specific focus on patient satisfaction and complications.
SURGICAL MANAGEMENT
Associated procedures: hysterectomy, salpingo-oophorectomy, colpectomy, and colpocleisis
Hysterectomy alone or with bilateral salpingo-oophorectomy (BSO) is often one of the first surgical steps undertaken by transgender men. While minimally invasive techniques (i.e., laparoscopic, robotic, or transvaginal) are typically performed, there is no reported benefit amongst these techniques[8]. While a transvaginal approach eliminates lower abdominal scarring, surgical access may be difficult in some individuals (i.e., nulliparous, or no prior penetrative sexual activity)[9].
Colpectomy and colpocleisis provide additional pelvic and perineal masculinization and decrease/eliminate vaginal secretions[10]. Colpectomy involves removal and/or ablation of the vaginal lining (anterior, lateral, and posterior vaginal wall epithelium) with closure of the vaginal vault. Colpectomy has been associated with significantly reduced rates of urethral fistula formation in transgender men undergoing urethral lengthening via metoidioplasty or phalloplasty[11].
Metoidioplasty techniques
Prior to undergoing metoidioplasty, the genitalia (clitoris and labia minora) must be sufficiently hypertrophied and of adequate dimensions. This is achieved through the preoperative use of testosterone, often for a minimum of 1-2 years, and possibly clitoral “pumping” (suction applied to the clitoris)[12]. Metoidioplasty may be performed by a variety of techniques which include simple metoidioplasty (no urethral lengthening), ring metoidioplasty, Belgrade metoidioplasty, and combinations/permutations thereof. These techniques typically employ some combination of labia minora flaps, vaginal flaps, and/or buccal mucosal grafts[13-19]. Metoidioplasty techniques historically involve the degloving of the clitoral shaft, release of the suspensory ligament of the clitoris (either completely or sub-totally), transection of the ventral chordae, and rearrangement of the skin of the labia minora and prepuce to construct the penile shaft. In essence, these steps represent the procedure referred to as a “simple release.” Resection of the suspensory ligament is performed on an individualized basis. In some cases, resection of the suspensory ligament, usually subtotal, facilitates lengthening of the penis and improves the appearance of the pubo-phallic angle. When urethral lengthening is requested, additional maneuvers are required to extend the urethra to the level of the glans. Ring metoidioplasty combines the previous steps with that of urethral elongation. The ring flap elongates the urethra by using an anterior vaginal wall flap in conjunction with labia minora flaps[7,13]. The Belgrade metoidioplasty technique follows concepts and techniques used in hypospadias repair[14]. This technique lengthens the urethra by utilizing anterior vaginal wall flaps combined with buccal mucosa and labial skin[15]. While most surgeons have abandoned the use of long vaginal flaps, a short vaginal/periurethral flap (1-2 cm) is still employed by our team.
A staged approach to metoidioplasty employs additional maneuvers performed in a second surgical setting designed to improve the excursion and the appearance of the neophallus. As described by Odeluga et al., the first stage involves phallic and urethral lengthening, vaginectomy, and scrotoplasty[20]. The second stage is tailored to the patient and includes repair of complications from stage one (fistula, stricture, vaginectomy wound complications, release of tethering scars), escutcheonectomy, and/or reduction of scrotal tissue (i.e., former labia majora), reduction to improve appearance.
Surgical staging
Metoidioplasty can be performed as a stand-alone procedure or as a first stage prior to phalloplasty, whether intentionally or due to dissatisfaction with the primary metoidioplasty[21]. In 2019, Al-Tamimi et al. studied 83 patients who underwent phalloplasty following metoidioplasty with a mean of 7.5 years in between[22]. The most common reasons were the desire for a larger phallus (39%, n = 32), the ability to engage in penetrative intercourse (30%, n = 25), those who had planned on a first stage metoidioplasty and a later second stage phalloplasty (20%, n = 17), or improved standing micturition (18%, n = 15). The majority of patients undergoing secondary phalloplasty (87%, n = 83) were dissatisfied with their original metoidioplasty, highlighting the importance of patient counseling prior to undergoing GGAS. Overall, their data demonstrated similar rates of complications between secondary phalloplasty and primary phalloplasty[22]. Metoidioplasty may be performed as a first stage procedure prior to a planned phalloplasty, or a metoidioplasty may be converted to phalloplasty for those individuals desiring a larger phallus. There is no broad consensus as to the staging of phalloplasty. In our opinion, the benefits of performing a metoidioplasty prior to a phalloplasty include reducing the likelihood of simultaneously incurring flap-related and urethral-related complications. While this may not reduce the overall complication rate, it does reduce the magnitude of complications and their impact on the individual’s personal and professional life.
Metoidioplasty may be combined with other procedures such as hysterectomy and bilateral salpingo-oophorectomy, colpectomy with colpocleisis, scrotoplasty (with or without testicular implant insertion), monsplasty, and insertion of corporal body implants[23]. Stojanovic et al. reported 473 patients over a
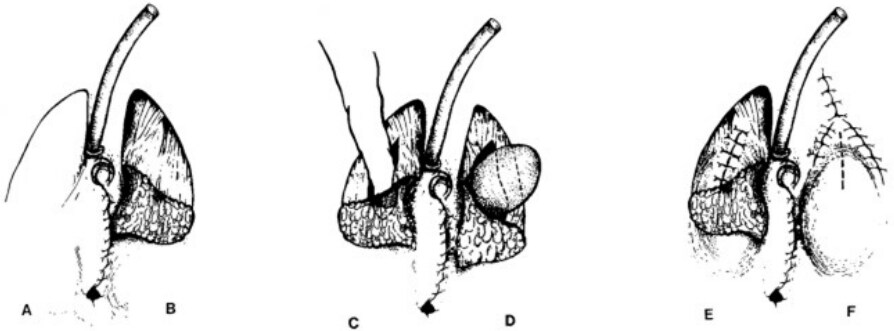
Scrotoplasty with insertion of testicular prostheses may be performed at the time of metoidioplasty or later, during a second-stage procedure[24]. The goals of scrotoplasty include sensation, a fused appearance, and positioning anterior to the thighs[25]. Scrotoplasty typically entails local rearrangement of the labia majora. Subcutaneous labial fat is preserved for bulk and subsequent protection of testicular implants. Other techniques such as non-genital pedicled skin flaps and myocutaneous flaps are less commonly utilized. Pigot et al. describe three common scrotoplasty techniques: the Hage technique, the Hoebeke technique, and a third technique[26]. The Hage technique involves a reverse V-Y flap and is the only technique that allows for concomitant implant insertion [Figure 2]. The Hoebeke technique utilizes medially rotated, cranially based labia majora flaps to create the neoscrotum, with implant insertion occurring during a subsequent procedure. The third technique includes the midline closure of dorsally-based reversed V-Y labia majora flaps followed by later stage implant insertion[26]. While a variety of testicular prostheses are available globally, only one saline-filled prosthesis is approved by the Food and Drug Administration for use in the United States[27].
Figure 2. The technique of scrotum construction in female-to-male transsexuals as applied in our hospital. (A) The labia majora are incised in a reversed V-like manner. (B) Only the skin of the dorsally based skin flaps is undermined. (C) The underlying tunica dartos is incised and undermined bluntly to form a cavity. (D) The large, adult-sized prosthesis is then inserted. (E) The tunica dartos has been closed in such a way that the scar will be fully covered by the skin flap. (F) The skin is closed in a Y-like fashion to lengthen the labia majora caudally.
While some surgeons advocate for immediate placement of testicular implants, this may increase the risk of infection, implant exposure, and/or wound dehiscence[28]. Intraoperative factors such as swelling, tension, insufficient pocket size, etc., may also impact the decision for immediate versus delayed insertion of testicular implants. Other concerns with immediate implant placement include potential implant contamination from a urine leak and/or pressure applied to the urethral reconstruction from the implants. Monsplasty may also be performed in patients with lipodystrophy and/or ptosis of the mons to better exposure to the neophallus; this is similar to the reconstruction of a buried penis[29].
Neuville et al. described the first use of an experimental, semi-rigid device implanted in the corpus cavernosum during a metoidioplasty[24]. The apparent purpose of the device is to support the neophallus during standing micturition (i.e., facilitating the penis exiting the zipper). Implant insertion was undertaken prior to neourethral reconstruction to avoid local contamination. This small study demonstrated no significant increase in neophallic length (mean length 4.1 cm), nor any data regarding penetrative intercourse. Long-term outcomes regarding this technique are yet to be determined[24].
METOIDIOPLASTY COMPLICATIONS
Metoidioplasty complications include urinary complications such as urethral stricture and/or fistula, post-void dribbling, urethral diverticula, urinary stream abnormalities (i.e., spraying) urinary tract infection (UTI), and bladder injury. Non-urologic complications include wound disruptions, bleeding and/or hematoma, and complications associated with adjunct procedures (i.e., monsplasty, colpectomy, colpocleisis, scrotoplasty with/without testicular implant insertion, and corpus cavernosum implant insertion). The following sections detail a systematic approach for the categorization of metoidioplasty-associated complications.
Urinary complications
Urologic complications are common following metoidioplasty[5]. These range from minor complications including urinary dribbling (whether temporary or permanent) and urine spraying to more significant issues such as urethral stricture or fistula. Other urologic complications include diverticula formation, UTI, cystitis, incontinence, urinary retention, and persistent bladder spasms. Table 1 demonstrates the overview of the studies with a listing of the reported minor urinary complication rates.
Overview of studies reporting complications and satisfaction following metoidioplasty
| First author, year | Number of patients | Metotoidioplasty details | Dribbling | Urethral stricture | Urethral fistula | Urethral diverticulum | UTI | Persistent bladder spasms | Cystitis | Urinary retention | Satisfaction |
| Djordjevic | 593 | 1S, Belgrade, UR; LR | 16% | 10 (1.7%) | 48 (8.1%) | Appearance 100% | |||||
| Lin-Brande | 33 | ± UL | 0 | 0 | 4 (2 from each group) | 2 (1 from each group) | |||||
| Takamatsu | 43 | LR | 3 (7%) | 7 (16.3%) | 3 (7%) | VWS 67.4% | |||||
| Jolly et al.[15], 2021 | 403 | 1S, UL | Range 2%-35% | Range 5%-37% | |||||||
| Perovic | 22 | UL | 2 (9.09%) | 3 (13.6%) | Appearance 77.3% | ||||||
| Djordjevic | 82 | 1S, UR | 23 (28%)* | 2 (2.4%) | 7 (8.5%) | VWS 100% | |||||
| Stojanovic | 473 | UL | 3 (3.8%) | 4 (5%) | 1 (1.3%) | Appearance 82.3% (65), VWS 100% | |||||
| Hage et al.[30], 2006 | 70 | 1S, UR | 25 (35.7%) | 26 (37.1%) | 3 (4.3%) | 11 (15.7%) | |||||
| Veerman | LR | 5 (62.5%) | 4 (50%) | 1 (12.5%) | |||||||
| Vukadinovic | 97 | 1S | 17 (17.53%)* | 2 (2%) | 6 (6.18%) | Appearance: 83.5%, VWS 100% (97) | |||||
| Robinson | 32 (Metoidioplasty only) | ± UL | 7 (21.9%) | 13 (40.6%) | 1 (3.1) | Appearance 48.4% | |||||
| Djordjevic | 557 | 1S | 33% | 8 (1.4%) | 39 (7%) | Appearance 100% |
Urethral stricture and fistula may occur following metoidioplasty; they may occur independently or in conjunction with each other. As described by Bizic et al., fistula formation is most likely to occur at the junction between the native urethra and the neourethra[29]. They attributed this location to size discrepancies between the native and the reconstructed urethra (i.e., the elongated, perineal urethra) and/or diminished vascular supply leading to ischemia of the various flaps used in neourethral reconstruction. Fistula formation and subsequent urinary leakage may also result from urinary and soft tissue infections, increased tension at the reconstructed urethra or additional pressure from inserted testicular implants leading to ischemia, flap, and reconstruction failure[30]. Lumen et al. noted that fistulas can occur secondary to increased intraurethral pressure from pre-stenotic dilation[31]. Urethral strictures following metoidioplasty are also related to functional anatomic differences between cis- and trans-men, namely the lack of spongious tissue in the lengthened neourethra.
In their 2019 series of 593 patients undergoing one-stage Belgrade metoidioplasty, Djordjevic et al. reported stricture and fistula rates of 2% (n = 10) and 8% (n = 48), respectively; all cases required surgical revision[7]. Hysterectomy and bilateral salpingo-oophorectomy were performed in approximately 25% of the patients, and urethral lengthening was performed with a buccal mucosa graft and fasciocutaneous labial flaps in over 90% of patients. In an earlier report from 2009 by the same group using the same technique, urethral stricture and fistula rates were 2% and 9%, respectively[19]. Improved surgical technique and experience may be responsible for the decrease in complications.
Other metoidioplasty techniques have yielded different rates of stricture and fistula formation. Hage et al. performed metoidioplasty with urethral elongation in 70 transgender patients[30]. Urethral strictures occurred in 36% of patients (n = 25) and fistulas occurred in 37% of patients (n = 26). Takamatsu et al. utilized the labial ring flap technique for metoidioplasty in 43 patients and reported a 7% stricture (n = 3) and 16% fistula rate (n = 7)[13]. Jolly et al. conducted a meta-analysis of metoidioplasty techniques and complications and found significant differences in stricture and fistula formation rates between different techniques [Table 2][15]. While the Belgrade technique had overall lower rates of stricture and fistula formation, there was insufficient information for the authors to indicate that one technique was superior as compared to the others[15].
Stricture and fistula formation rates between different metoidioplasty techniques[15]
| Metoidioplasty technique | Stricture rate | Fistula rate |
| Labial ring | 5% | 27% |
| Hage | 36% | 37% |
| Belgrade | 3% | 7% |
Waterschoot et al. reported on “additional urethral lengthening” (AUL) performed in 36 (48%) of the 74 metoidioplasties in their study[21]. Although the data suggest metoidioplasty plus AUL is feasible, rates of temporary (resolved in 3 months) and permanent (unresolved in 3 months) fistula and stricture were significantly higher in the AUL group. Furthermore, they note that more permanent fistulas and strictures occur in the AUL segment, described as the “pendulous” urethra.
Veerman et al. compared the rates of urologic complications in metoidioplasty using a labial ring flap technique to that of one stage “big ben” phalloplasty and urethral reconstruction[32]. In their series of 63 patients, eight patients underwent metoidioplasty, and 55 patients underwent one-stage phalloplasty. They employed a variety of combinations to create the neophallus (49% free radial forearm flap (n = 27); 35% anterolateral thigh flap (n = 19), 16% superficial circumflex iliac artery perforator flap (n = 9) and neourethra (49% tube-in-tube free radial forearm flap (n = 27), 33% free radial forearm flap (second fasciocutaneous flap) (n = 18), 9% superficial circumflex iliac artery perforator flap (n = 5), and 9% labial
Stojanovic et al. prospectively reported 473 patients undergoing metoidioplasty from 2007-2016[23]. In this study, 29% of the patients (n = 137) underwent simultaneous hysterectomy and 17% (n = 79) underwent one-stage gender-affirmation surgery (vaginectomy, total transvaginal hysterectomy, metoidioplasty with urethral lengthening, scrotoplasty, placement of testicular prostheses, and chest masculinization)[23]. Collectively, 10% of patients undergoing one-stage surgery experienced urethral complications (n = 8) with a 4% rate of stricture formation (n = 3), a 5% rate of fistula formation (n = 4), and a 1% rate of urethral diverticulum formation (n = 1). Revision surgery was performed in 11% of the 79 patients (n = 9). This included two patients who underwent fistula repair, two patients who underwent stricture repair, and one patient who underwent repair of a urethral diverticulum. Comparing this data to rates of urologic complications in metoidioplasty alone (i.e., without hysterectomy, testicular protheses, etc.), the additional procedures did not seem to increase the risk of urethral complications[23]. However, Danker et al.[33] report that insertion of testicular implants at the time of scrotoplasty and urethroplasty was associated with an increased risk of infection and may lead to an increased rate of stricture and/or fistula formation[34].
While much of the literature regarding metoidioplasty describes the rates of fistula and stricture formation, there is limited research regarding their surgical repair. Lumen et al. reported on the repair of post-metoidioplasty urethral fistulas (fistuloplasty, n = 18) and strictures (urethroplasty, n = 12), noting the rate of failure is approximately 33%[31].
Post-void urine leakage may be related to urethral diverticula, changes in urethral compliance and elasticity, and/or the construction of a static urinary tube (i.e., no bulbospongiosus muscle). In 2019, Djordjevic et al. reported their series of 593 transgender men who underwent metoidioplasty over a 15-year period[7]. They note that dribbling and spraying occurred in 16% of patients; every case resolved spontaneously, and no patients required surgical revision. In their study of 97 patients undergoing single-stage metoidioplasty with vaginectomy, urethroplasty, scrotoplasty, and testicular implant insertion, Vukadinovic et al. reported that 18% of patients (n = 17) experienced urinary dribbling or spraying[34]. All 17 patients were managed non-operatively. The authors reported that by three months, all cases of dribbling and spraying resolved spontaneously. Post-void dribbling may be managed non-operatively using urethral milking.
UTI and cystitis may occur following metoidioplasty. Hage et al. reported UTI in 4% of patients (n = 3)[30]. Lin-Brande et al. reported UTI in 10% of patients undergoing metoidioplasty with urethral lengthening
Lin-Brande et al.[12] and Hage et al.[30] report 6% (n = 2) and 15.7% (n = 11) cases of urinary retention following metoidioplasty, respectively. According to Hage et al., three patients required surgical intervention, and a mechanical obstructive cause of retention was cystoscopically proven in only 3 of the 11 cases[30]. No other causes were identified.
Persistent bladder spasms and urethral diverticula have each been reported once in reference to metoidioplasty[23,35]. In the senior author’s experience, urethral diverticula are relatively common and because many may be asymptomatic, their incidence is likely underreported.
Wound complications
While the genital and perineal areas have a robust blood supply, impaired hygiene may predispose to wound colonization and subsequent infection. In their series of 33 patients, Lin-Brande et al. report that two patients experienced dehiscence of the distal urethral flap (with no reports of either stricture or fistula)[12]. One patient developed granulation tissue which required operative intervention. Additionally, delayed wound healing and cellulitis were each seen in one patient. While overall complications were comparable between the two groups of patients in this study [simple metoidioplasty (50%) and metoidioplasty with urethral lengthening (48%)], wound-related complications were significantly higher in patients who underwent urethral lengthening. The authors posited that labia minora tissue must be of adequate length, which was not quantified, to cover the ventral urethra and shaft, and preoperative assessment of the labia minora is predictive of wound breakdown, urethral flap dehiscence, and even candidacy for metoidioplasty[12].
Djordjevic et al. examined 207 patients undergoing metoidioplasty using a buccal mucosa graft for urethral plate reconstruction combined with either a longitudinal dorsal clitoral skin flap (group I, 40 patients) or a labia minora flap (group II, 158 patients)[18]. Neither group experienced flap necrosis or postoperative infections, and prophylactic antibiotics were given for seven days. Conversely, Hage et al. reported that 23 of 70 patients experienced immediate postoperative complications including four (6%) wound infections[30]. While all patients in the Djordjevic study underwent placement of testicular prostheses at the time of metoidioplasty and reported no postoperative infections, Hage et al. found that simultaneous testicular implant insertion significantly increased the risk of infection[30]. Hage required sufficient labial laxity as a prerequisite for the placement of testicular implants[30]. In addition, in order to reduce the risk of wound dehiscence, patients were required to void via suprapubic cystostomy for seven days. The rationale for urinary diversion was to decrease intraurethral pressureassociated with micturition through an edematous urethra[30].
Bleeding complications
Colpectomy is associated with significant blood loss[36]. However, there are limited reports of estimated blood loss during metoidioplasty procedures. In their case series of 82 patients, Djordjevic et al. reported no clinically significant postoperative bleeding[19]. Lin-Brande et al. reported a median intraoperative blood loss of 400 mL (n = 21) in patients undergoing metoidioplasty with urethral lengthening and 75 mL (n = 12) in patients undergoing metoidioplasty without urethral lengthening[12]. Clinically significant bleeding seems more related to the vaginectomy rather than the metoidioplasty; every patient (n = 21) undergoing urethral lengthening also underwent vaginectomy, while only 25% of patients undergoing simple metoidioplasty also underwent vaginectomy (n = 3). Hougen et al., in their series of 40 patients undergoing vaginectomy, reported a rate of pelvic hematoma, 3% (n = 1), and a rate of significant blood loss necessitating blood transfusion, 3% (n = 1)[37]. Groenman et al. described a median intraoperative blood loss of 75 mL in 36 transgender men undergoing robotic-assisted laparoscopic hysterectomy with BSO and robotic-assisted colpectomy with subtotal closure of the introitus[10]. Subgroup analysis of the 36 cases demonstrated a reduction in mean intraoperative blood loss from 157 mL in the first 18 cases to 30 mL in the last 18 cases.
The reported rate of hematoma following metoidioplasty ranges between 3%-10%. Hage et al., in their series of patients undergoing metoidioplasty without vaginectomy, reported a hematoma in 11% of patients (n = 8 of total 70)[30]. Six of these individuals (9%) required surgical intervention to address postoperative hematoma. Despite hematoma formation in 11% of the patients, there were no reported cases of significant blood loss, though. The use of the anterior vaginal wall flap was associated with donor site bleeding. The authors highlighted the need for meticulous hemostasis to decrease the risk of bleeding from the perivaginal venous plexus. They also advocate for surgical exploration of postoperative bleeding or hematoma, whether immediate or delayed, to prevent flap loss. This recommendation is based on their previous experience, where they describe a severe hematoma resulting in loss of the anterior vaginal wall flap[16].
In a survey of 129 transgender men who underwent genital GAS [phalloplasty (61%), metoidioplasty (25%), or both (14%)], Robinson et al. found that hematoma occurred in 13% of patients (n = 17), and 16 of 17 hematoma cases were managed non-operatively[35]. Rates of hematoma were higher in patients who received metoidioplasty or metoidioplasty followed by phalloplasty at 22% and 17%, respectively, versus phalloplasty alone (9%). They do not specify whether vaginectomy was performed. The limited data and lack of clarity regarding the source of bleeding make definitive conclusions regarding bleeding-associated complications difficult to analyze.
Bladder and lower gastrointestinal injury
Nikkels et al. evaluated 143 transgender patients undergoing colpectomy with or without additional procedures such as metoidioplasty and hysterectomy[36]. A bladder injury was reported in 6 patients (4%) and bowel (rectal) injury in 1 patient (1%). Waterschoot et al. reported three bladder injuries (4%) and two rectal injuries (3%) in their analysis of 74 patients undergoing metoidioplasty[21]. Although bladder and bowel/rectal injury during colpectomy/vaginectomy is a recognized complication, it is variably reported in the literature.
COMPLICATIONS WITH ASSOCIATED PROCEDURES
Scrotoplasty and implant complications
Complications related to testicular implants, such as displacement and/or exposure, are not uncommon. Kang et al. report that the most common implant-associated complications include prosthetic extrusion, displacement/malposition, scrotal contraction (not further defined), pain, hematoma, and infection[25]. The authors noted that approximately 50% of the patients in their series (n = 70 patients) experienced displacement of at least one implant. This necessitated surgical correction in most cases (percentage not further specified by the authors). In addition, 30% of patients required explantation of at least one implant due to infection, obstruction of urinary flow, and/or wound dehiscence.
In their single-stage approach, Stojanovic et al. reported testicular “implant rejection” and implant displacement in 3% and 1% of their patients, respectively[23]. In an eight-year follow-up, Hage et al. reported loss of the testicular prostheses in 29% of patients and displacement in 49% of patients[30]. Despite seeing comparable rates of testicular prosthesis loss in two subgroups (concomitant metoidioplasty with scrotoplasty, 49% vs. staged scrotoplasty, 55%), the authors concluded that the data was subject to selection bias. Patients with insufficient labial skin are at a higher risk of implant erosion and/or displacement. In these patients, a staged scrotoplasty was performed.
Pigot et al. reported their series of 206 patients who underwent testicular implant placement using three different surgical methods (Hage, Hoebeke, and reversed V-shaped)[26] . The most common issues requiring prosthetic explantation were infection, extrusion, discomfort, prosthesis leakage, or urethral problems. The overall explantation rate was 21%, and smoking was the most frequent risk factor. Urethral issues, such as external compression by the prosthesis, were observed in 1% of patients (n = 2 patients), prosthetic rupture or leakage occurred in 2% of patients (n = 4), and neourethral-scrotal fistula occurred in 1 % of patients (n = 2). The authors report an association between scrotoplasty technique and risk of explantation; the Hage technique (concomitant implant insertion) has the highest risk, and the Hoebeke technique (staged implant insertion) has the lowest risk. The authors also found that the rate of explantation was lower in patients who underwent surgery later in the series. This may be attributed to advancements in prosthetic technology (i.e., smaller and lighter prostheses), a trend toward secondary (i.e., delayed) implantation, and surgeon experience. Collectively, the data from Pigot et al.[26] and Danker et al.[33] favor a staged approach to testicular prosthesis insertion.
Delayed rupture of silicone gel prostheses may occur, whether due to mechanical failure and/or pressure on the implant. MRI is considered the gold standard for implant evaluation, although ultrasound may also be used[30]. Selvaggi et al. reported a 5% complication rate on 240 patients who underwent scrotoplasty with V-Y labia majora advancement and rotation flaps[1]. At one year follow-up, no prosthesis required removal. The authors describe the merits of this technique as enhanced erogenous sensitivity and low complication rates, although no comparison group was provided.
Insertion of penile prostheses within the cavernous bodies may also be performed in conjunction with metoidioplasty. Neuville et al. reported on 15 patients who underwent simultaneous metoidioplasty and placement of an experimental semi-rigid penile prosthesis[24]. The authors report wound dehiscence in 33% of patients (n = 5). These complications were managed non-operatively. The authors reported urinary fistula in 13% of patients (n = 2), and hematoma occurred in 7% of patients (n = 1). No complications relating specifically to the penile prosthesis were reported. The authors believe that the prosthesis contributed to higher rates of urethral complications. As unassisted erections are considered a benefit of metoidioplasty (as compared to phalloplasty), it is unclear as to the benefit of penile prostheses in this population.
OUTCOMES OF METOIDIOPLASTY AND ASSOCIATED PROCEDURES
Surgical outcomes
While there is no standard approach for vaginectomy, the most commonly described technique involves resection of the vaginal lining and closure of the vaginal space[37]. This may be performed prior to or in conjunction with metoidioplasty. Hougen et al.[37] performed 40 vaginectomies in conjunction with metoidioplasty using the complete epithelial excision technique[33]. During a 7.7-month median follow-up period, low intra- and postoperative complication rates were observed. 5% (n = 2) of cases required blood transfusion, 3% (n = 1) resulted in pelvic hematoma, and 3% (n = 1) of cases resulted in postoperative C-difficle infection. The authors also reported an increase in intraoperative blood loss proportional to an increase in the patient’s body mass index[37].
Al-Tamimi et al. advocated for primary vaginectomy prior to metoidioplasty or phalloplasty as this led to a decrease in urethral complications such as fistula formation[11]. They also utilized secondary colpectomy plus fistulectomy to treat urethral fistulas following GAS with urethral lengthening. Similarly, Massie et al. evaluated patients who underwent combined metoidioplasty, phalloplasty, and urethral lengthening procedures and found that in patients with prior vaginectomy, there was a decrease in urethral fistula and stricture formation[38]. This decrease was attributed to the use of a labia minora flap with a vascularized vestibular flap to cover the urethroplasty sutures. As described by Hage et al.[16], without a complete vaginectomy, in which the ventral vaginal wall and muscularis are used for neourethra construction, the potential for fistula formation exists between the closed vaginal space remnants[16,37].
Al-Tamimi et al. advocated for primary vaginectomy prior to metoidioplasty or phalloplasty as this led to a decrease in urethral complications such as fistula formation[11]. They also utilized secondary colpectomy plus fistulectomy to treat urethral fistulas following GAS with urethral lengthening. Similarly, Massie et al. evaluated patients who underwent combined metoidioplasty, phalloplasty, and urethral lengthening procedures and found that in patients with prior vaginectomy, there was a decrease in urethral fistula and stricture formation[38]. This decrease was attributed to the use of a labia minora flap with a vascularized vestibular flap to cover the urethroplasty sutures. As described by Hage et al.[16], without a complete vaginectomy, in which the ventral vaginal wall and muscularis are used for neourethra construction, the potential for fistula formation exists between the closed vaginal space remnants[16,37].
Patient satisfaction following metoidioplasty
High rates of satisfaction are reported following metoidioplasty[39]. Patient satisfaction with sexual function and aesthetic outcome has been reported to be greater than 80% and 77%, respectively[40]. Dissatisfaction is generally correlated with unfavorable results, complications, and lower scores on psychological and/or quality-of-life (QoL) outcome indicators. In a series of 63 patients who underwent metoidioplasty (n = 8) and phalloplasty (n = 55), Veerman et al. noted that only 70% of patients were able to void from the tip of their phallus and 73% of patients required multiple additional surgeries to address urologic complications[32]. The authors did not stratify satisfaction rates by the type of procedure. Veerman et al. also concluded that pre- and postoperative quality of life scores were unchanged, showing that unfavorable results and complications deterred any expected score improvements[32].
In their series of 97 patients, Vukadinovic et al. reported standing urination and satisfactory sexual function (reaching orgasm) in all patients undergoing metoidioplasty[34]. These outcomes correlated with overall satisfaction and QoL scores. Similarly, Pigot et al. found that dissatisfaction was mostly associated with sexual function problems, voiding difficulties, and inability to meet aesthetic expectations[41]. In their series of 68 patients [metoidioplasty (52%), phalloplasty (49%)], 45% reported “very satisfactory” or “satisfactory” sexual function, 53% reported satisfactory voiding, and 63% reported satisfactory penis aesthetic with 65% acceptance of neo-scrotum appearance. In this series, 80% of patients would choose to undergo surgery again, and 70% would recommend the surgery to someone else[42] [Table 1].
CONCLUSION
Metoidioplasty preserves genital sensation and allows for unassisted erections and standing micturition. Additionally, metoidioplasty offers the advantage of a less complex procedure as compared to phalloplasty without the need for remote donor sites. While complications may occur, most are treated non-operatively or without a need for revision surgery. Variations in surgical technique and associated procedures make a comparative analysis of reported metoidioplasty outcomes difficult. Future research should be directed at standardizing terminology and outcome measures.
DECLARATIONS
Authors’ contributionsMade substantial contributions to conception of the review and analysis and interpretation of current data: Hamidian Jahromi A, Spellman AM, Horen S, Cherullo EE, Dorafshar AH, Schechter LS
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable. The authors obtain consent for all photographs.
Copyright© The Author(s) 2022.
REFERENCES
1. Selvaggi G, Hoebeke P, Ceulemans P, et al. Scrotal reconstruction in female-to-male transsexuals: a novel scrotoplasty. Plast Reconstr Surg 2009;123:1710-8.
2. Weissler JM, Chang BL, Carney MJ, et al. Gender-affirming surgery in persons with gender dysphoria. Plast Reconstr Surg 2018;141:388e-96e.
3. Canner JK, Harfouch O, Kodadek LM, et al. Temporal trends in gender-affirming surgery among transgender patients in the United States. JAMA Surg 2018;153:609-16.
4. Colebunders B, Brondeel S, D’Arpa S, Hoebeke P, Monstrey S. An update on the surgical treatment for transgender patients. Sex Med Rev 2017;5:103-9.
5. Hadj-Moussa M, Agarwal S, Ohl DA, Kuzon WM Jr. Masculinizing genital gender confirmation surgery. Sex Med Rev 2019;7:141-55.
6. Bizic M, Stojanovic B, Bencic M, Bordás N, Djordjevic M. Overview on metoidioplasty: variants of the technique. Int J Impot Res 2020;33:762-70.
7. Djordjevic ML, Stojanovic B, Bizic M. Metoidioplasty: techniques and outcomes. Transl Androl Urol 2019;8:248-53.
8. Jeftovic M, Stojanovic B, Bizic M, et al. Hysterectomy with bilateral salpingo-oophorectomy in female-to-male gender affirmation surgery: comparison of two methods. Biomed Res Int 2018;2018:3472471.
9. The World Professional Association for Transgender Health. Standards of care for the health of transsexual, transgender, and gender-conforming people, 7th Version. Available from: https://www.wpath.org/publications/soc [Last accessed on 7 May 2022].
10. Groenman F, Nikkels C, Huirne J, van Trotsenburg M, Trum H. Robot-assisted laparoscopic colpectomy in female-to-male transgender patients; technique and outcomes of a prospective cohort study. Surg Endosc 2017;31:3363-9.
11. Al-Tamimi M, Pigot GL, van der Sluis WB, et al. Colpectomy significantly reduces the risk of urethral fistula formation after urethral lengthening in transgender men undergoing genital gender affirming surgery. J Urol 2018;200:1315-22.
12. Lin-Brande M, Clennon E, Sajadi KP, Djordjevic ML, Dy GW, Dugi D. Metoidioplasty with urethral lengthening: a stepwise approach. Urology 2021;147:319-22.
13. Takamatsu A, Harashina T. Labial ring flap: a new flap for metaidoioplasty in female-to-male transsexuals. J Plast Reconstr Aesthet Surg 2009;62:318-25.
14. Stojanovic B, Djordjevic ML. Anatomy of the clitoris and its impact on neophalloplasty (metoidioplasty) in female transgenders. Clin Anat 2015;28:368-75.
15. Jolly D, Wu CA, Boskey ER, Taghinia AH, Diamond DA, Ganor O. Is clitoral release another term for metoidioplasty? Sex Med 2021;9:100294.
16. Hage JJ. Metaidoioplasty: an alternative phalloplasty technique in transsexuals. Plast Reconstr Surg 1996;97:161-7.
17. Perovic SV, Djordjevic ML. Metoidioplasty: a variant of phalloplasty in female transsexuals. BJU Int 2003;92:981-5.
18. Djordjevic ML, Bizic MR. Comparison of two different methods for urethral lengthening in female to male (metoidioplasty) surgery. J Sex Med 2013;10:1431-8.
19. Djordjevic ML, Stanojevic D, Bizic M, et al. Metoidioplasty as a single stage sex reassignment surgery in female transsexuals: Belgrade experience. J Sex Med 2009;6:1306-13.
20. Odeluga N, Reddy SA, Safir MH, Crane CN, Santucci RA. Optimization of second-stage metoidioplasty. Urology 2021;156:303-7.
21. Waterschoot M, Hoebeke P, Verla W, et al. Urethral complications after metoidioplasty for genital gender affirming surgery. J Sex Med 2021;18:1271-9.
22. Al-Tamimi M, Pigot GL, van der Sluis WB, et al. The surgical techniques and outcomes of secondary phalloplasty after metoidioplasty in transgender men: an international, multi-center case series. J Sex Med 2019;16:1849-59.
23. Stojanovic B, Bizic M, Bencic M, et al. One-stage gender-confirmation surgery as a viable surgical procedure for female-to-male transsexuals. J Sex Med 2017;14:741-6.
24. Neuville P, Carnicelli D, Paparel P, Ruffion A, Morel-Journel N. Metoidioplasty with implantation of a specific semirigid prosthesis. J Sex Med 2021;18:830-6.
25. Kang A, Aizen JM, Cohen AJ, Bales GT, Pariser JJ. Techniques and considerations of prosthetic surgery after phalloplasty in the transgender male. Transl Androl Urol 2019;8:273-82.
26. Pigot GLS, Al-Tamimi M, Ronkes B, et al. Surgical outcomes of neoscrotal augmentation with testicular prostheses in transgender men. J Sex Med 2019;16:1664-71.
27. Hayon S, Michael J, Coward RM. The modern testicular prosthesis: patient selection and counseling, surgical technique, and outcomes. Asian J Androl 2020;22:64-9.
28. Kocjancic E, Acar O, Talamini S, Schechter L. Masculinizing genital gender-affirming surgery: metoidioplasty and urethral lengthening. Int J Impot Res 2022;34:120-7.
29. Bizic MR, Stojanovic B, Joksic I, Djordjevic ML. Metoidioplasty. Urol Clin North Am 2019;46:555-66.
30. Hage JJ, van Turnhout AA. Long-term outcome of metaidoioplasty in 70 female-to-male transsexuals. Ann Plast Surg 2006;57:312-6.
31. Lumen N, Waterschoot M, Verla W, Hoebeke P. Surgical repair of urethral complications after metoidioplasty for genital gender affirming surgery. Int J Impot Res 2020;33:771-8.
32. Veerman H, de Rooij FPW, Al-Tamimi M, et al. Functional outcomes and urological complications after genital gender affirming surgery with urethral lengthening in transgender men. J Urol 2020;204:104-9.
34. Vukadinovic V, Stojanovic B, Majstorovic M, Milosevic A. The role of clitoral anatomy in female to male sex reassignment surgery. ScientificWorldJournal 2014;2014:437378.
35. Robinson IS, Blasdel G, Cohen O, Zhao LC, Bluebond-Langner R. Surgical outcomes following gender affirming penile reconstruction: patient-reported outcomes from a multi-center, international survey of 129 transmasculine patients. J Sex Med 2021;18:800-11.
36. Nikkels C, van Trotsenburg M, Huirne J, et al. Vaginal colpectomy in transgender men: a retrospective cohort study on surgical procedure and outcomes. J Sex Med 2019;16:924-33.
37. Hougen HY, Dugi DD 3rd, Berli JU, Sajadi KP. Outcomes of transperineal gender-affirming vaginectomy and colpocleisis. Female Pelvic Med Reconstr Surg 2021;27:300-3.
38. Massie JP, Morrison SD, Wilson SC, Crane CN, Chen ML. Phalloplasty with urethral lengthening: addition of a vascularized bulbospongiosus flap from vaginectomy reduces postoperative urethral complications. Plast Reconstr Surg 2017;140:551e-8e.
39. Djordjevic ML, Bizic M, Stanojevic D, et al. Urethral lengthening in metoidioplasty (female-to-male sex reassignment surgery) by combined buccal mucosa graft and labia minora flap. Urology 2009;74:349-53.
40. Frey JD, Poudrier G, Chiodo MV, Hazen A. An update on genital reconstruction options for the female-to-male transgender patient: a review of the literature. Plast Reconstr Surg 2017;139:728-37.
41. Pigot GLS, Al-Tamimi M, Nieuwenhuijzen JA, et al. Genital gender-affirming surgery without urethral lengthening in transgender men-A clinical follow-up study on the surgical and urological outcomes and patient satisfaction. J Sex Med 2020;17:2478-87.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Hamidian Jahromi A, Spellman AM, Horen S, Cherullo EE, Dorafshar AH, Schechter LS. A narrative review of outcomes following metoidioplasty: complications and satisfaction. Plast Aesthet Res 2022;9:41. http://dx.doi.org/10.20517/2347-9264.2021.111
AMA Style
Hamidian Jahromi A, Spellman AM, Horen S, Cherullo EE, Dorafshar AH, Schechter LS. A narrative review of outcomes following metoidioplasty: complications and satisfaction. Plastic and Aesthetic Research. 2022; 9(6): 41. http://dx.doi.org/10.20517/2347-9264.2021.111
Chicago/Turabian Style
Hamidian Jahromi, Alireza, Ari M. Spellman, Sydney Horen, Edward E. Cherullo, Amir H. Dorafshar, Loren S. Schechter. 2022. "A narrative review of outcomes following metoidioplasty: complications and satisfaction" Plastic and Aesthetic Research. 9, no.6: 41. http://dx.doi.org/10.20517/2347-9264.2021.111
ACS Style
Hamidian Jahromi, A.; Spellman AM.; Horen S.; Cherullo EE.; Dorafshar AH.; Schechter LS. A narrative review of outcomes following metoidioplasty: complications and satisfaction. Plast. Aesthet. Res. 2022, 9, 41. http://dx.doi.org/10.20517/2347-9264.2021.111
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 5 clicks
Cite This Article 5 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.