Peripheral nerve allograft: how innovation has changed surgical practice
Abstract
The landscape of available technology and surgical technique has changed over the last several decades, thus leading to changes in the peripheral nerve repair surgical algorithm. Neurorrhaphy is a common procedure; however, it is well recognized that nerve repair should be performed tensionless, thus preventing the ability to perform direct repair with a nerve gap. Historically, nerve gaps were repaired with autograft. However, autograft surgery has been associated with complications such as numbness and chronic pain, which left surgeons searching for alternatives. Nerve allografts were first utilized in the 1800s but failed due to the immune response. In the modern era, they were again utilized in the 1980s, but did not gain popularity because of the need for the use of immunosuppressants. It was evident through the 1990s that continued innovation in peripheral nerve repair was needed, as studies showed that only approximately 50% of patients with nerve gap repair achieved good or excellent outcomes. In the 2000s, the advent of an engineered nerve allograft (Avance® Nerve Graft) changed the landscape of peripheral nerve repair. Early clinical evaluation of Avance showed that adequate sensation was able to be achieved in nerve gaps up to 30 mm, providing an alternative to autografts. As engineered nerve allograft use became more conventional, studies showed 87.3% meaningful recovery in nerve gaps up to 50 mm. Furthermore, recent studies have shown that gaps between 50-70 mm have shown 69% meaningful recovery. While technology and surgical technique continue to improve, these results are promising for large nerve gap repair.
Keywords
INTRODUCTION
Hand and wrist injuries occur in 6.6% of emergency room visits in the United States, costing $48.6 billion annually[1]. Peripheral nerve injuries occur in 2.5% of trauma patients[2], with the average number of peripheral nerve procedures at 558,862 annually[3]. The most frequently injured nerves treated within hospitals include ICD-9-CM 955.6, upper extremity digital nerve; ICD-9-CM 955.2, ulnar nerve; ICD-9-CM 955.3, radial nerve; and ICD-9-CM 953.4, the brachial plexus[4]. Peripheral nerve injuries have socioeconomic costs, direct patient costs, and can affect patient quality of life.
The notable socioeconomic costs for the patient include missed work/school due to regular physician appointments, procedures, and hospital visits. These appointments and procedures can result in significant direct costs to the patient, which can be compounded by the loss of wages due to missed work[5]. Additionally, patient quality of life can be impacted by disrupted sleep patterns, social life, extremity function, personal life, professional activities, and mood[6]. Notably, 64% of patients with peripheral nerve injuries have missed at least one month of work or school, and 24% of patients with nerve injuries have missed at least 12 months of work or school[5]. These significant impacts on patients’ economic standing and quality of life highlight the importance of continuing to improve outcomes in the treatment of peripheral nerve injuries.
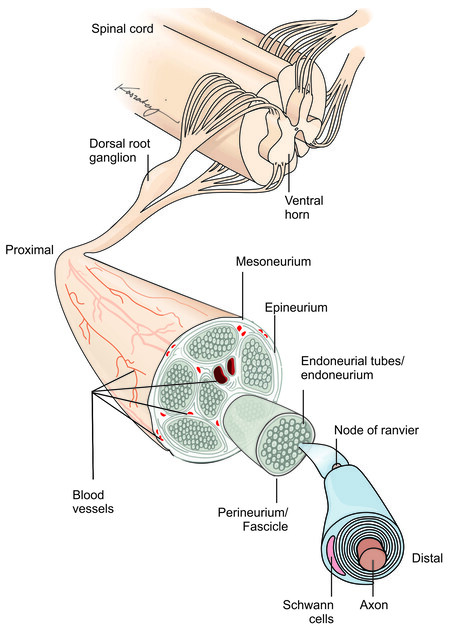
When a peripheral nerve is injured, the resulting injury may lead to varying disruptions in the peripheral nerve anatomy. These varying injury severities result in different functional impacts, which are related to the anatomical structure of peripheral nerves. The peripheral nerve is composed of several layers of connective and functional tissues that support the electrical impulse propagation and the structure of the nerves.
Peripheral nerves extend from the spinal cord and are comprised of both sensory (afferent) and motor (efferent) nerve fibers[7]. These nerve fibers, called axons, are either myelinated or unmyelinated
| Seddon classification[8] | Sunderland classification[9] | Structure injured | Ability to recover |
| Neurapraxia | Type 1 | Myelin | Spontaneous recovery |
| Axonotmesis | Type 2 | Myelin, axon | Spontaneous recovery, but slower than neurapraxia/type 1 |
| Type 3 | Myelin, axon, endoneurium | Spontaneous recovery unlikely, may require surgical intervention | |
| Type 4 | Myelin, axon, endoneurium, perineurium | Spontaneous recovery highly unlikely, requires surgical intervention | |
| Neurotmesis | Type 5 | Myelin, axon, endoneurium, perineurium, epineurium | Spontaneous recovery impossible, requires surgical intervention |
Each nerve fiber is surrounded by the endoneurium, which is a loose collagenous connective tissue layer[7]. Bundles of endoneurium are contained within fascicles, which are surrounded by a connective tissue layer called the perineurium[7]. The perineurium consists of uniformly organized flattened laminae of fibroblasts with alternating sheets of collagen[7]. The outermost layer of the nerve, the epineurium, is composed of irregularly arranged collagenous tissue that provides elasticity and absorption of mechanical forces[7,10]. Peripheral nerve injuries resulting in a complete discontinuity in the peripheral nerve axon and surrounding connective tissue layers are classified as Seddon neurotmesis and Sunderland type 5.
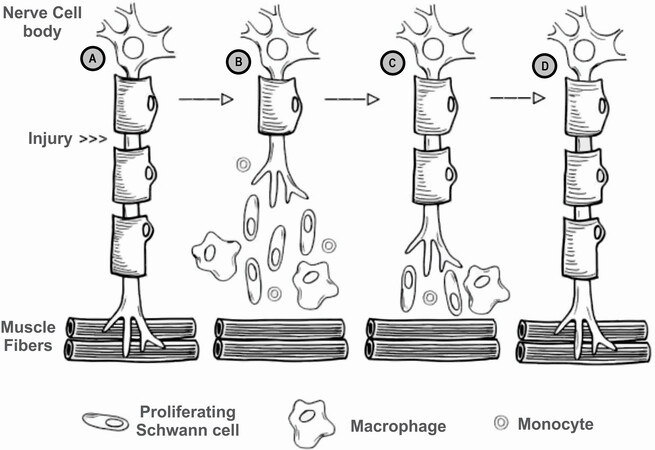
Subsequent to nerve injury, the nerve proximal to the injury undergoes traumatic degeneration up to the first node of Ranvier, while the nerve distal to the injury undergoes Wallerian degeneration [Figure 2A][11]. During Wallerian degeneration, Schwann cells and macrophages degrade myelin in the nerve distal to the injury [Figure 2B][12]. Axonal sprouting occurs from the nerve proximal to the injury within 24 hours of the injury, where the axonal growth cone extends down the basal lamina of the endoneurial tubes [Figure 2C][11] and connects to the distal target organ [Figure 2D]. When a nerve injury classified as Sunderland type IV (axonotmesis) and Sunderland type V (neurotmesis) occurs, surgical intervention is required due to loss of continuity in axons and, at a minimum, the endoneurium and perineurium[8,9]. Surgical treatment is required in 43.5% of patients suffering from peripheral nerve injuries[2], with 56% of patients undergoing direct repair and 44% of patients undergoing nerve gap repair[3]. While direct nerve repair is the historical standard for nerve repair, the nerve ends must be reapproximated without tension at the suture[13]. However, nerve transections may result in a gap between the nerve stumps due to tissue loss from the injury, surgical debridement, or natural retraction of the nerve[14,15].
Figure 2. Process of Wallerian degeneration after peripheral nerve injury (A) intact nerve, prior to nerve injury, location of injury noted; (B) post-injury Wallerian degeneration of the nerve distal to the injury, with traumatic degeneration up to the first node of Ranvier in the nerve stump proximal to the injury; (C) axonal sprouting from the nerve proximal to the injury, where the growth cone extends down basal lamina of the endoneurial tubes; and (D) nerve regeneration complete, with connection established with distal target organ.
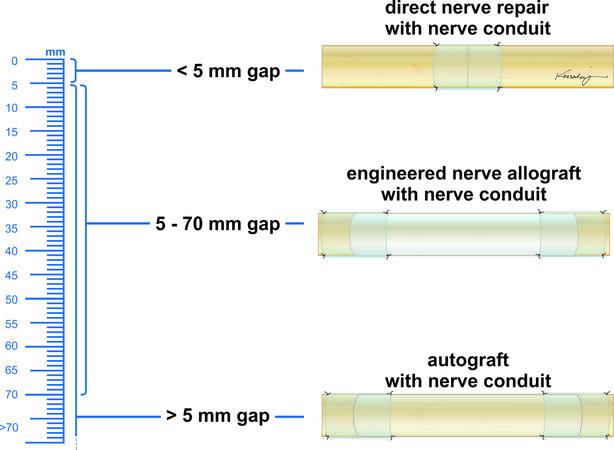
When a nerve gap exists during peripheral nerve regeneration, the endoneurial tubes of the distal nerve stump cannot be accessed by the leading axonal growth cone from the proximal nerve stump due to the physical distance between the nerve stumps, thus leading to the development of a neuroma[16]. The repair of a transected nerve should be performed such that healthy fascicles are reapproximated in a tensionless manner, as tension has been shown to lead to ischemia and decreased axonal outgrowth[17]. In the case of a nerve gap, repair is often performed via nerve grafting using conduit, autograft, or allograft to span the gap between the nerve stumps [Figure 3][18]. Nerve repair has changed notably over the last several decades due to the introduction of microsurgical techniques and off-the-shelf options for bridging nerve gaps.
Off-the-shelf options for bridging nerve gaps include extracellular matrix scaffolds that support axonal regeneration, cellular and non-cellular graft additives. Ideally, a nerve scaffold should: be readily available, be biodegradable over a time appropriate for the application, be able to be revascularized, support cell migration, elicit a limited immunogenic response, allow for oxygen and nutrient diffusion, be adjustable for the nerve injury severity, not lead to long-term nerve compression, and support nerve regeneration[19-21]. Furthermore, Porzionato et al. proposed that the best scaffold for tissue engineering is decellularized extracellular matrix of the same origin as the target tissue[22]. Scaffolds that support axonal regeneration can be engineered from natural or synthetic materials, although natural materials are thought to show improved biocompatibility, decreased toxicity, and better cellular migration[19]. The addition of bioactive factors and cells have also been investigated with the use of nerve scaffolds to stimulate cell migration and provide a preferential substrate for axonal migration[19]. While many of these advancements are not yet clinically available, novel materials for peripheral nerve repair such as engineered nerve allografts and nerve conduits are currently clinically available. The clinical emergence of these materials has inevitably changed the landscape of peripheral nerve repair and the surgical algorithm. This manuscript will explore the advent of nerve conduits and engineered peripheral nerve allografts and how their use has impacted the repair of transected peripheral nerves over time. Furthermore, this manuscript will focus on the clinical application of these technological advancements in extremities, as the most frequently injured nerves are located in the extremities.
HISTORY OF PERIPHERAL NERVE REPAIR
Nerve autograft
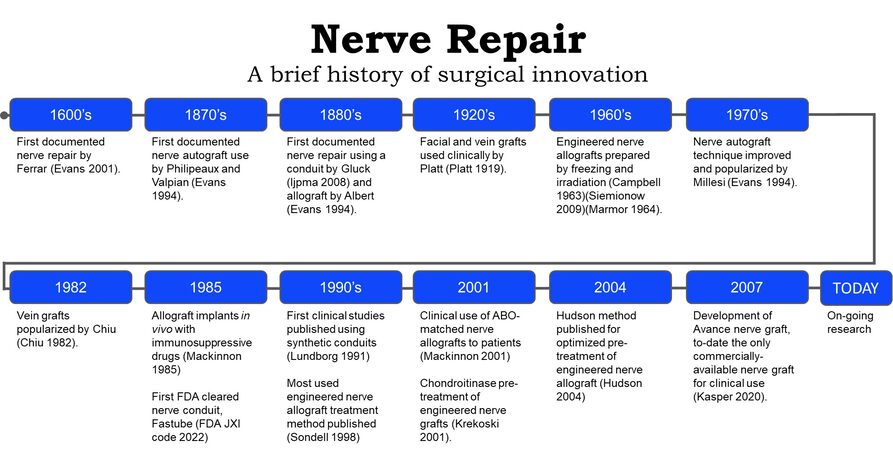
Records of peripheral nerve repair date back to the Hippocratic era[23]; however, the techniques and materials used for nerve gap repair have changed through the years [Figure 4]. In the 1800s, various techniques for peripheral nerve repair were described in the literature, including segmental nerve repair using nerve autograft[38], conduits of various materials[38-40], and cellular nerve allografts[41]. Nerve autograft is considered the historical standard for peripheral nerve gap repair when tensionless direct nerve repair cannot be achieved. Nerve autografts can be successfully utilized as single grafts, cabled, interfascicular, or vascularized grafts[38]. Nerve autografts should be chosen from a donor nerve that is considered expendable and would not lead to undesirable defects after graft harvest[38]. The most common autologous nerve graft is harvested from the sural nerve[38,42]; however, other nerve autograft sources may include medial and lateral antebrachial cutaneous nerve, the dorsal antebrachial nerve, the superficial branch of the radial nerve, dorsal branch of the ulnar nerve, lateral femoral cutaneous nerve, and the posterior interosseous nerve[38]. The selection of the nerve autograft is determined by the surgeon with feedback from the patient regarding the anticipated sensory deficit from the graft harvest. Sural nerve resections, such as sural nerve autograft harvest or sural nerve biopsy, have been shown to cause sensory deficits in 92.9% of patients and sensory symptoms (e.g., tingling cold intolerance, paresthesia, dysesthesia, or irritating sensation) in 41.1% of patients[43]. Additionally, sural nerve autograft harvest has been linked to increased incidents of chronic pain, wound infection, wound complications, impact on daily life, post-operative hematoma, and deep vein thrombosis[43]. Due to these complications, alternative bridging materials have been investigated[6].
Nerve conduits
Nerve conduits were an early alternative to nerve autograft, as nerve conduits circumvent nerve autograft harvest related complications, reduce scar tissue invasion within the nerve gap and help prevent axonal escape, which decreases the likelihood of neuroma formation[25]. In the 1920s, fascial and vein grafts were used clinically by Platt[26,44], and further popularized by Chiu et al.[29] in the 1980s[45].
In the 1990s and early 2000s, various synthetic and biologic nerve conduits were proposed for use in the literature. Early results indicated that conduits could be used to bridge nerve gaps less than 3 cm[46]. However, more recent literature suggests that nerve gaps measuring up to 1 cm are the limit for repair with a nerve conduit[47]. This is due to the lack of structural guidance in hollow conduits and the reliance on the formation of a fibrin cable within the conduit to provide axonal guidance across the gap. At longer gap lengths, the fibrin cable does not provide adequate structure for the regenerating axon throughout the regeneration process[48]. In the repair of peripheral nerve gaps beyond 1 cm, a nerve graft should be utilized[47]. While there are various FDA-cleared nerve conduits, a review in 2021 found that these conduits are mostly used in gaps less than 1 cm and exhibit poorer outcomes in longer gap repairs [Table 2][48].
| Device name | Material | FDA clearance date |
| NervAlign | Porcine pericardium | 02/10/2022 |
| NeuroShield | Chitosan | 05/21/2019 |
| Nerbridge | Polyglycolic acid and porcine dermis-derived collagen | 01/22/2016 |
| Reaxon | Chitosan | 12/02/2015 |
| NeuraGen 3D | Bovine type-1 collagen with inner matrix collagen/chondroitin-6 sulfate | 4/24/2014 |
| Neurolac nerve guide | Poly (DL-lactide-co-ε-caprolactone) | 10/10/2003 |
| Surgisis nerve cuff (Axoguard nerve connector) | Porcine small intestine submucosa | 05/15/2003 |
| Collagen nerve cuff | Collagen | 09/21/2001 |
| NeuroGen nerve guide | Collagen | 07/22/2001 |
| Salumedica nerve cuff | Polyvinyl alcohol hydrogel | 11/24/2000 |
| Neurotube | Polyglycolic acid | 03/22/1999 |
| Fastube nerve regeneration device | Unknown | 07/10/1985 |
Nerve allograft
Nerve allografts were sought as an alternative to autografts, as nerve allografts can be prepared and stored in tissue banks, do not lead to a secondary donor surgical site and provide the proper structural guidance needed for peripheral nerve regeneration[18]. Engineered nerve allografts were first noted in the literature in the 1960s, which were pre-treated by freezing and irradiation[18,27,28]. However, the initial success of engineered nerve allografts faced limitations during early surgical use, as grafts were noted to show delayed axonal outgrowth, elicited an immunologic response and resulted in nerve graft rejection[50-53]. Research to improve nerve allograft outcomes continued, including major histocompatibility complex (MHC) matching, patient immunosuppression and nerve graft processing methods. In 1985, Mackinnon et al. attempted MHC matching in rats and found good regeneration in MHC matched allografts and poor regeneration in the MHC unmatched grafts[30].
In 2001, Mackinnon evaluated the clinical use of donor allograft nerve that was blood-type (ABO) matched to patient recipients[34]. These ABO-matched nerve allografts showed good sensory and motor outcomes in patients[34]. The immunologic response in cellular allogenic nerve grafts was noted to decrease over time as the donor Schwann cells were replaced by host Schwann cells[54], but immunosuppressive treatments, including Cyclosporin-A and tacrolimus (FK506), were still required[18]. Alternative areas of research, such as nerve allograft pre-treatments, were investigated to circumvent the need for immunosuppressives.
Research in engineered nerve allograft pretreatment development has included cryopreservation, lyophilization[55], freeze/thaw cycling[56], cold storage, chemical treatments to extract cellular debris or pre-degenerate the graft[33,35,36,57,58], and irradiation[27]. The most prolific engineered nerve allograft pre-treatment protocol used in literature was proposed by Sondell et al.[33] in 1998, which used Triton X-100 and sodium deoxycholate solution to chemically lyse cells[59]. This pre-treatment protocol resulted in the removal of the myelin sheath and cells from engineered nerve allografts, resulting in a satisfactory nerve regeneration response in vivo[33]. Later research was conducted in 2001 by Krekoski et al. showing that chondroitin sulfate proteoglycan glycosaminoglycan side chains, known to inhibit axonal growth by functional blockade of laminin, could be removed from the nerve by treatment with a chondroitinase ABC enzyme[35]. The methods proposed by Krekoski et al. showed that chondroitinase pre-treated engineered nerve allografts improved the growth-promoting properties of the nerve allografts and resulted in more axons growing at longer lengths through the chondroitinase treated allografts compared to grafts that were not treated with chondroitinase[35]. Several years later, in 2004, Hudson et al. proposed an engineered nerve allograft pre-treatment protocol using Sulfobetaine-16, Triton X-200, and Sulfobetaine-10 to improve cell lysis while maintaining the extracellular matrix[36]. The Hudson et al. study showed that engineered nerve allograft pre-treatment with mild detergents resulted in axon densities that were comparable to nerve isografts (considered to be the equivalent of nerve autografts in pre-clinical studies)[36]. Furthermore, these Hudson pre-treated engineered nerve allografts also showed 910% more axon density than thermally treated engineered neve allografts and 401% more axon density than the pre-treatment process proposed by Sondell[33]. Pre-treating engineered nerve allografts with both the Hudson pre-treatment and the Krekoski pre-treatment has been found to be the most effective pre-treatment for engineered nerve allografts, when compared among other well-established nerve allograft pre-treatment protocols[59]. Avance® nerve graft was developed using both pre-treatment methods[37] outlined by Hudson et al.[36] and Krekoski et al.[35].
The effort to develop an engineered nerve allograft utilizing these two protocols involved over 20 years of research by two research groups, and came to fruition in 2007 when Avance nerve graft was made available as an off-the-shelf engineered nerve allograft for clinical use[37]. To date, Avance nerve graft is the only engineered nerve allograft commercially available in the United States for clinical use. The initial pre-clinical studies showed that Avance nerve allografts did not show an immunogenic reaction and maintained the native extracellular matrix structure of the nerve, including laminin, a protein critical to neurite outgrowth[60].
CLINICAL USE OF AVANCE NERVE GRAFT
The first clinical report of Avance nerve graft use was published in 2009 and involved 8 patients with 10 sensory nerve repairs[61] [Table 3]. The average gap length was 2.23 cm (range 0.5-3 cm), and all patients had sensory improvement by 9 months[61]. Through 2016, several clinical publications demonstrated that adequate sensation was achieved in sensory nerve gap repair using Avance nerve graft in gaps up to 30 mm in the upper and lower extremity, providing an alternative to autografts[64,65,76-79]. Engineered nerve allografts, such as Avance nerve graft, provided advantages over nerve autografts, including circumventing donor-site morbidity, off-the-shelf availability, easy to use, and reduced operative time[61,64,65,76-78]. Further research expanded the gap length and nerve type repaired with Avance nerve graft.
Clinical studies using off-the-shelf commercially available engineered nerve allografts reporting outcomes
| Author, year | Study type | Mean gap length | Nerve type, n-value | Mean follow-up | Implants used/n-value | Outcomes |
| Karabekmez et al.[61], 2009 | Retrospective | 22.3 mm (Range 5-30 mm) | Sensory, n = 10 | 9 months | Allograft, n = 10 | The average static two-point discrimination was 5.5 mm, and the moving two-point discrimination was 4.4 mm. This was the first study to show clinical efficacy for using clinically available engineered nerve grafts to treat sensory defects up to 3 cm in length |
| Brooks et al.[62], 2012 | Retrospective observational | 22 11 mm (Range 5-50 mm) | Sensory, n = 49 Mixed, n = 18 Motor, n = 9 | 264 152 days | Allograft, n = 76 | A meaningful recovery in 87.3% of repairs for motor (> M3) and sensory (> S3) function on MRCC scale. Provides functional recovery in sensory, mixed and motor nerve injuries in gaps up to 50 mm |
| Cho et al.[63], 2012 | Retrospective observational | 23 12 mm (Range 5-50 mm) | Sensory, n = 35 Mixed, n = 13 Motor, n = 3 | 296 160 days | Allograft, n = 51 | Meaningful recovery (≥ S3 and ≥ M3) was achieved in 86% of cases |
| Ducic et al.[47], 2012 | Retrospective observational | Primary, 0 mm Conduit, 9.1 3.7 mm Autograft, 37.5 13.2 mm Allograft, 17.6 7.5 mm | Unknown | Primary 204.0 41.3 weeks Conduit 186.4 56.7 weeks Autograft 250.5 59.3 weeks Allograft 129.7 89.2 weeks | Primary, n = 8 Conduit, n = 27 Autograft, n = 11 Allograft, n = 8 | The recorded average QuickDASH (disabilities of the arm, shoulder, and hand) questionnaire was reported to average 23.2 19.8. There were no significant differences between repair type with respect to outcomes |
| Guo et al.[64], 2013 | Retrospective observational | 23 mm (Range 18-28 mm) | Digital, n = 5 | 13 mon. (Range 12-15 months) | Allograft, n = 5 | All patients reported sensory improvement during the follow-up period after operation. Mean Static two-point discrimination of 6 mm was recorded, and the range of the Semmes-Weinstein Monofilament test was 4.31 to 4.56 |
| Taras et al.[65], 2013 | Prospective | 11 mm (Range 5-30 mm) | Digital, n = 18 | 15 mon. (Range 12-20 months) | Allograft, n = 18 | According to custom Taras[66] scale, excellent results in 39% of repairs, good results in 44% of repairs, fair results in 17% of repairs and poor results in no repairs |
| Isaacs and Safa[67], 2017 | Retrospective observational | 33 10 mm (Range 5-50 mm) | Sensory, n = 2 Mixed, n = 13 | 13 months | Allograft, n =15 | Meaningful recovery for motor (≥ M3) function in 85% of repairs and sensory (≥ S3) function in 67% of repairs, independent of nerve diameter |
| Rinker et al.[68], 2017 | Retrospective observational | 35 8 mm (Range 25-50 mm) | Sensory, n = 50 | 11 months | Allograft, n = 50 | Meaningful recovery for sensory (≥ S3) function in 86% of repairs |
| Zhu et al.[69], 2017 | Retrospective observational | 27 ± 13 mm | Sensory, n = 39 Mixed, n = 19 Motor, n = 6 | 355 158 days | Allograft, n = 64 | Meaningful recovery for motor (≥ M3) function in 66.7% of repairs and sensory (≥ S3) function in 84.6% of repairs |
| Carlson et al.[70], 2018 | Retrospective observational | 65 45 mm (Range 10-140 mm) | Sensory, n = 11 Mixed, n = 3 Motor, n = 2 | 15 5 months | Allograft, n = 19 | Meaningful recovery for motor (≥ M3) function in 33.3% of repairs and sensory (≥ S3) in 91.7% of repairs |
| Nietosvaara et al.[71], 2019 | Case series | 20-50 mm | digital | 10-19 months | Allograft, n = 3 | Resorption of the engineered nerve graft or neuroma proximal to the nerve graft |
| Safa et al.[72], 2019 | Retrospective observational | 33 ± 17 mm (Range 10-70) mm | Mixed and motor, n = 22 | 779 480 days | Allograft, n = 22 | Meaningful recovery was reported in 73% of repairs. No significant differences were noted between gap lengths or mechanism of injury |
| Leckenby et al.[73], 2020 | Retrospective observational | 26.6 ± 16.0 mm (Range 8-100 mm) | Sensory, n = 110 Mixed, n = 25 | 417 214 days | Allograft, n = 135 | Meaningful recovery for motor (≥ M3) function in 36% of repairs and sensory (≥ S3) in 77% of repairs. Inferior prognosis for larger diameter and longer grafts |
| Safa et al.[74], 2020 | Retrospective observational | 24 15 mm (Range 3-70 mm) | Sensory, n = 386 Mixed, n = 77 Motor, n = 12 | 417 days (Range 120-3286 days) | Allograft, n = 624 | Overall meaningful recovery in 82% of repairs in sensory, mixed and motor nerve repairs |
| Peters et al.[75], 2021 | Case series | Range 60-110 mm | Median, n = 3 Ulnar, n = 2 | 9-24 months | Allograft, n = 5 | Subjects had previously experienced an iatrogenic injury. Subjects showed no clinical sensory or motor recovery and showed significant neuropathic pain ranging between 8 and 10 on a 10-score visual analog scale. Histology showed axonal regeneration stalling mid-graft in 3 of 5 grafts and no axonal regeneration in 2 of 5 grafts |
In 2012, Brooks et al. published the first results of nerve gaps repaired with Avance nerve graft up to 50 mm in length, which included 76 nerve gaps averaging 22 mm (range, 5-50 mm) in sensory, mixed, and motor nerves located in the upper and lower extremity[62]. Brooks and colleagues showed meaningful recovery in 87.3% of nerve gaps repaired with Avance nerve graft, where meaningful recovery was defined as a return of motor recovery to M3 or greater and sensory recovery to S3 or greater using the Medical Research Council Classification (MRCC) scale[62]. Additionally, there were no significant differences in sensory and motor outcomes between sensory, mixed, or motor nerve repairs[62]. In late 2012, Cho et al. published on 51 sensory, mixed, and motor nerve gaps repaired with Avance nerve graft in the upper extremity only[63]. Cho et al. and colleagues showed in nerve gaps averaging 23 mm (range, 5-50 mm), 86% of repairs achieved S3 or M4 and above recovery[63]. The adoption of engineered nerve allograft into clinical use and early publications led to the development of an evidence-based algorithm.
In 2012, Ducic et al. discussed that direct repair should be used in nerve gaps less than 5 mm, nerve conduits should be used in gaps 5 mm to 15 mm, engineered nerve allografts should be used in nerve gaps 5 mm to 50 mm, and nerve autografts should be used in nerve gaps 5 mm to greater than 50 mm[47]. This suggested that both engineered nerve allograft and nerve autograft could be used in similar gap sizes. By 2014, it was proposed by Rinker et al. that engineered nerve allografts, such as Avance nerve graft, were the most significant development in peripheral nerve surgery since the introduction of microsurgery[80]. Furthermore, Rinker noted that most major hand centers had updated their nerve repair algorithm as a result of the introduction of these engineered nerve allografts[80]. Additional clinical evidence focusing on long-gap repair was presented by Rinker et al. in 2017, where a patient population of 50 digital (sensory) nerve gaps measuring 25 mm to 50 mm repaired with Avance nerve graft showed S3 or greater recovery in 86% of repairs[68]. Use of Avance nerve graft to repair 15 large diameter (4-5 mm in diameter) nerve gaps averaging 33 mm in length (range 5-50 mm) using a single Avance nerve graft was evaluated in 2017, which showed functional recovery of S3 or M3 and better of sensory and motor function in 67% and 85% of repairs[67].
An additional update to the surgical algorithm was proposed in 2017, when Ducic et al. suggested that utilizing a nerve conduit for connector-assisted repair may help overcome difficulties noted in the literature that may impede peripheral nerve regeneration after repair[81]. This connector-assisted repair was proposed for direct repair as well as using with engineered nerve allograft to prevent misalignment of the fascicles during the repair[81]. This algorithm was supported by further clinical evidence in 2018, when Carlson et al. evaluated 19 sensory, mixed, and motor nerve gaps averaging 65 mm in length (range, 10-140 mm) that were repaired with Avance nerve graft reinforced at the nerve coaptation site with Axoguard® nerve protector[70]. Carlson and colleagues found that 91.7% of repairs showed S3 or better sensory recovery, with 66% meaningful recovery in gap lengths greater than 50 mm[70]. As clinical evidence of the efficacy of Avance nerve graft continued to build, surgeons adapted their surgical algorithm to include the use of Avance nerve graft.
Evidence presented in 2018 by Azouz et al. showed that 70% of hand surgeons used engineered nerve allografts in their surgical practice, as noted by the use of current procedural terminology codes 64910 (nerve repair with synthetic conduit or vein allograft), 64890 (nerve graft, single strand, hand, < 4 cm), 64831 (suture of digital nerve, hand, or foot, 1 nerve), 64911 (nerve repair with autogenous vein graft), 64999 (unlisted procedure nervous system) and 64834 (suture of 1 nerve, hand, common sensory nerve)[82]. Additionally, the surgical algorithm for peripheral nerve repair has continued to be updated, as recently updated algorithms incorporate the use of engineered nerve allograft as well as autograft in gaps up to 7 cm in length[83,84]. The clinical evaluation of 22 nerve repairs using engineered nerve allograft in 2019 showed meaningful motor recovery, as noted by M3 function or above, in 73% of nerve repairs with a median of 33 ± 17 mm (10-70 mm) for nerve graft lengths[72]. Furthermore, motor recovery of M3 or above was reported at 80% for nerve gaps 10-25 mm, 63% for nerve gaps 26-49 mm, and 75% for nerve gaps 50 mm or larger[72]. While this was a small cohort, additional larger studies provided similar evidence. In 2020, Safa et al. reported on meaningful recovery, defined as S3/M3 or greater, in 624 sensory and mixed nerve gap repairs measuring up to 70 mm in length[74]. Safa and colleagues showed that in gap lengths 50-70 mm, repair with Avance nerve graft resulted in 69% meaningful recovery, with no statistical difference between the 50-70 mm, 30-49 mm, and 15-29 mm nerve gap repair groups[74]. Furthermore, there was an overall meaningful recovery of 82% in nerve gaps up to 70 mm in length, which was noted to be comparable to historical data for nerve autograft and exceeding historical literature for conduit[74].
While a plethora of positive clinical evidence has been presented for the use of engineered nerve allografts, additional literature has been published with lower success rates. In 2019, a case series of three patients was presented by Nietosvaara outlining poor results due to engineered nerve graft resorption[71]; however, the failures were attributed to possible infection and host rejection. In 2020, Leckenby et al. presented sub-optimal results for a single-center experience with engineered nerve allograft and found that 77% of patients achieved sensory recovery of S3 or better and 36% of patients achieved motor recovery of M3 or better[73]. However, Leckenby et al. suggested that outcomes with the engineered nerve grafts were similar to nerve autograft in short nerve gaps[73]. Further discussion by Leckenby et al. suggested that issues were encountered with increased length and diameter of engineered nerve grafts[73].
When considering the body of evidence for the clinical use of engineered nerve grafts for the repair of peripheral nerve defects, there is overwhelming support for their application. Furthermore, the use of engineered nerve grafts can also circumvent the comorbidities associated with nerve autograft. With these considerations, there is sufficient support for further clinical use of engineered nerve allografts, such as Avance nerve graft.
DISCUSSION
Peripheral nerve repair, much like other surgical repairs, has undergone a drastic transformation in the last 20 years. This transformation has been largely in part due to the development of innovative peripheral nerve repair materials such as nerve conduits and engineered nerve allografts. Nerve conduits have provided a material to improve peripheral nerve repair by providing a protected environment for peripheral nerve regeneration, moving the suture away from the regenerating axons at the proximal nerve stump, and allowing for selective reinnervation of the distal target[81]. While these advances in the application of nerve conduits have been useful, the application of a nerve conduit should be limited to nerve gaps less than 1 cm in length[47]. Longer nerve gaps require the use of a nerve autograft or engineered nerve allograft. While nerve autografts have shown good outcomes in large gaps, the patient comorbidities associated with nerve autograft harvest often include chronic pain, wound infections, wound complications, and sensory deficits[43]. While recovery from some of these complications may occur, sensory deficits have shown variable outcomes, with 0%-11% of adult patients experiencing complete sensory recovery[43]. The variability in recovery of sensory deficits has been suggested to be correlated with the length of the resected nerve segment, where longer nerve segment resections, such as nerve autograft harvest, show larger areas of chronic sensory deficits[43]. The acute and chronic comorbidities related to nerve autograft harvest should be a consideration for the surgical algorithm of surgeons performing peripheral nerve repair. Furthermore, alternatives to nerve autografts should be considered by evaluating evidence-based outcomes. The advent of engineered nerve allografts has provided one such alternative to nerve autografts that circumvent the associated donor-site morbidities associated with nerve autografts and offer promising outcomes.
The development of engineered nerve allografts has required decades of research, but has been proven to achieve successful clinical outcomes. The early use of pre-treated engineered nerve allografts showed limited successful outcomes[50-53] or required immunosuppression[85]. With later advancements in the field, the advent of an optimized nerve allograft pre-treatment method allowed for the successful implantation of engineered nerve allografts without the use of immunosuppressives and with outcomes that are comparable to autograft[62,63]. The development and clinical use of this off-the-shelf engineered nerve allograft, Avance nerve graft, has finally driven changes in the peripheral nerve repair surgical algorithm. Initial clinical research supported the use of engineered nerve allografts in nerve gaps up to 30 mm, with a later expansion of successful clinical use for nerve gap repair up to 50 mm. However, a recent clinical study supports the use of engineered nerve allografts for both sensory and mixed/motor nerve repair in gaps up to 70 mm in length[74]. With this recent publication, the application of engineered nerve allografts can be confidently used clinically in nerve gaps up to 70 mm in length.
The successful outcomes of peripheral nerve repair with engineered nerve allograft are promising; however, limitations exist in the literature, including the lack of randomized controlled clinical trials comparing engineered nerve allograft to autograft. While retrospective clinical trials lack stringent controls seen in randomized clinical trials, the retrospective studies reviewed in this manuscript provide the best comparative data available to date in the repair of peripheral nerve gaps. By evaluating the clinical data chronologically, it is clear that technological advancements in peripheral nerve repair support the use of engineered nerve grafts with increasing gap lengths over time. This method of chronological presentation serves to present the data as it has been shared with the field, ensuring a balanced presentation of meaningful studies to the clinical community. It is expected that technology will continue to improve, thus changing the future surgical algorithm for peripheral nerve repair.
The clinical application of materials and techniques currently in early-phase research will continue to change the landscape of peripheral nerve repair. Some notable early-phase research includes engineered nerve allografts, engineered nerve conduits with and without fillers, and cellular and non-cellular graft additives. Engineered nerve allografts have included various processing techniques, including cold preservation[51], freeze thawing[86], detergents[36], and irradiation[87]. While various engineered nerve allografts have been researched, only Avance has been made available commercially as an off-the-shelf engineered nerve allograft. This limits the ability to evaluate the clinical efficacy of different engineered nerve allografts. Engineered nerve conduits have been investigated using various synthetic and natural materials, which may be either resorbable or non-resorbable.
A recent meta-analysis found that autograft vein conduits, autograft muscle-vein conduits, engineered collagen tubes (e.g., NeuroMatrix, Neuroflex, NeuraGen® nerve guide), and Neurolac™ (a bioresorbable synthetic material) were the most studied and best nerve conduit options[88]. New materials for conduit fillers and additives have been explored to enhance conduit efficacy and increase the application length of these materials. Research on new conduit filler materials includes fibrin, laminin, collagen, and synthetic aligned matrices[89]. Furthermore, additives have been investigated with various nerve graft materials, including cellular (e.g., Schwann cells, fibroblasts, and bone stromal cells) and non-cellular (e.g., neurotrophic growth factor, fibroblast growth factor, glial growth factor, ciliary neurotrophic factor, vascular endothelial growth factor, and brain-derived growth factor) components[89]. These additives to luminal fillers have shown beneficial nerve regeneration effects across a nerve gap[89].
Engineered nerve allografts have also been evaluated with the use of additives and cellular enhancements, which showed that enhancement with additives[90] or cells[91] improve regenerative potential. However, these results are only available in the pre-clinical phase of research, as to date there is no FDA cleared commercially available biological additives or cells for the clinical application to engineered nerve conduits or allografts[48]. The ability to use this technology clinically will require additional research and clearance by appropriate regulatory bodies. When available, the clinical use of new conduit materials, luminal fillers, additives or cells will likely continue to expand the surgical algorithm in the future.
CONCLUSION
In conclusion, engineered nerve allografts have impacted the peripheral nerve repair surgical paradigm and should be considered as an alternative to nerve autograft for peripheral nerve repair. The use of engineered nerve allografts, such as Avance nerve graft, show meaningful motor and sensory recovery in nerve gaps up to 70 mm in length. Recent data has provided confidence for the use of clinically available engineered nerve grafts even for repair of longer nerve gaps 50-70 mm in length. Furthermore, the use of engineered nerve allograft as an alternative to autograft circumvents nerve autograft comorbidities, such as sensory deficits and chronic pain. While the surgical algorithm for peripheral nerve repair is ever-changing, additional research and clearance for clinical use by regulatory bodies are required to advance current surgical techniques.
DECLARATIONS
AcknowledgmentsThe author would like to acknowledge the support of Dr. Anne Engemann and her team for review of this manuscript prior to submission.
Authors’ contributionsThe author contributed solely to the article.
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestThe author is a paid consultant for Axogen; however, no financial support was provided for the creation of this manuscript.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Colen DL, Fox JP, Chang B, Lin IC. Burden of hand maladies in US emergency departments. Hand (N Y) 2018;13:228-36.
2. Noble J, Munro CA, Prasad VS, Midha R. Analysis of upper and lower extremity peripheral nerve injuries in a population of patients with multiple injuries. J Trauma 1998;45:116-22.
3. Brattain K. Analysis of the peripheral nerve repair market in the United States. Magellan Med Technol Consult Inc Minneap 2014; doi: 10.2172/85042.
4. Karsy M, Watkins R, Jensen MR, Guan J, Brock AA, Mahan MA. Trends and cost analysis of upper extremity nerve injury using the national (nationwide) inpatient sample. World Neurosurg 2019;123:e488-500.
5. Felder JM, Ducic I. Cross-sectional evaluation of the economic burden on patients seeking surgical treatment for chronic nerve injuries. Ann Plast Surg 2022;88:200-7.
6. Felder JM, Ducic I. Chronic nerve injuries and delays in surgical treatment negatively impact patient-reported quality of life. Plast Reconstr Surg Glob Open 2021;9:e3570.
7. Goldstein B. Anatomy of the peripheral nervous system. Physical Medicine and Rehabilitation Clinics of North America 2001;12:207-36.
8. Seddon H. Surgical disorders of the peripheral nerves. Baltimore: Williams and Wilkins; 1975. Available from: https://pesquisa.bvsalud.org/portal/resource/pt/biblio-1085568 [Last accessed on 18 May 2022].
9. Sunderland S. Nerves and nerve injuries. New York: Churchill Livingstone; 1978. Available from: https://xueshu.baidu.com/usercenter/paper/show?paperid=9ca836416bf984ed6a0d2ccb570eba31&site=xueshu_se [Last accessed on 18 May 2022].
10. Maugeri G, D’Agata V, Trovato B, et al. The role of exercise on peripheral nerve regeneration: from animal model to clinical application. Heliyon 2021;7:e08281.
11. Griffin JW, Hogan MV, Chhabra AB, Deal DN. Peripheral nerve repair and reconstruction. J Bone Joint Surg Am 2013;95:2144-51.
12. Stoll G, Trapp B, Griffin J. Macrophage function during Wallerian degeneration of rat optic nerve: clearance of degenerating myelin and Ia expression. J Neurosci 1989;9:2327-35.
14. Ray WZ, Mackinnon SE. Management of nerve gaps: autografts, allografts, nerve transfers, and end-to-side neurorrhaphy. Exp Neurol 2010;223:77-85.
15. Millesi H, Schmidhammer R. How to improve the results of peripheral nerve surgery. Austria: Springer Vienna; 2007.
16. Oliveira KMC, Pindur L, Han Z, Bhavsar MB, Barker JH, Leppik L. Time course of traumatic neuroma development. PLoS One 2018;13:e0200548.
17. Yi C, Dahlin LB. Impaired nerve regeneration and Schwann cell activation after repair with tension. Neuroreport 2010;21:958-62.
18. Siemionow M, Brzezicki G. Chapter 8 current techniques and concepts in peripheral nerve repair. Elsevier; 2009. p. 141-72.
19. Evans GR. Peripheral nerve injury: a review and approach to tissue engineered constructs. Anat Rec 2001;263:396-404.
20. Verreck G, Chun I, Li Y, et al. Preparation and physicochemical characterization of biodegradable nerve guides containing the nerve growth agent sabeluzole. Biomaterials 2005;26:1307-15.
21. Chrząszcz P, Derbisz K, Suszyński K, et al. Application of peripheral nerve conduits in clinical practice: a literature review. Neurol Neurochir Pol 2018;52:427-35.
22. Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R. Tissue-engineered grafts from human decellularized extracellular matrices: a systematic review and future perspectives. Int J Mol Sci 2018;19:4117.
23. Belen D, Aciduman A, Er U. History of peripheral nerve repair: may the procedure have been practiced in Hippocratic School? Surg Neurol 2009;72:190-3.
24. Evans PJ, Midha R, Mackinnon SE. The peripheral nerve allograft: a comprehensive review of regeneration and neuroimmunology. Progress in Neurobiology 1994;43:187-233.
25. IJpma FF, Van De Graaf RC, Meek MF. The early history of tubulation in nerve repair. J Hand Surg Eur Vol 2008;33:581-6.
26. Platt H. On the results of bridging gaps in injured nerve trunks by autogenous fascial tubulization and autogenous nerve grafts. British Journal of Surgery 1919;7:384-9.
27. Campbell JB, Bassett AL, Boehler J. Frozen-irradiated homografts shielded with microfilter sheaths in periphearal nerve surgery. J Trauma 1963;3:303-11.
28. Marmor L. The repair of peripheral nerves by irradiated homografts. Clin Orthop Relat Res 1964;34:161-9.
29. Chiu DT, Janecka I, Krizek TJ, Wolff M, Lovelace RE. Autogenous vein graft as a conduit for nerve regeneration. Surgery 1982;91:226-33.
30. Mackinnon SE, Hudson AR, Falk RE, Hunter DA. The nerve allograft response - an experimental model in the rat. Ann Plast Surg 1985;14:334-9.
31. Devices@Fda - Jxi Code. US Food and Drug Administration; 2022. Available from: https://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm?start_search=1&q=SlhJ&approval_date_from=&approval_date_to=&sort=approvaldatedesc&pagenum=10 [Last accessed on 18 May 2022].
32. Lundborg G, Dahlin LB, Danielsen N. Ulnar nerve repair by the silicone chamber technique. Case report. Scand J Plast Reconstr Surg Hand Surg 1991;25:79-82.
33. Sondell M, Lundborg G, Kanje M. Regeneration of the rat sciatic nerve into allografts made acellular through chemical extraction. Brain Research 1998;795:44-54.
34. Mackinnon SE, Doolabh VB, Novak CB, Trulock EP. Clinical outcome following nerve allograft transplantation. Plast Reconstr Surg 2001;107:1419-29.
35. Krekoski CA, Neubauer D, Zuo J, Muir D. Axonal regeneration into acellular nerve grafts is enhanced by degradation of chondroitin sulfate proteoglycan. J Neurosci 2001;21:6206-13.
36. Hudson TW, Zawko S, Deister C, et al. Optimized acellular nerve graft is immunologically tolerated and supports regeneration. Tissue Engineering 2004;10:1641-51.
37. Kasper M, Deister C, Beck F, Schmidt CE. Bench-to-bedside lessons learned: commercialization of an acellular nerve graft. Adv Healthc Mater 2020;9:e2000174.
40. Bungner O. Ueber die degenerations und regenerations-vorgange am nerven nach verletzungen. Beitr Pathol Anal 1891;10:321.
43. Ducic I, Yoon J, Buncke G. Chronic postoperative complications and donor site morbidity after sural nerve autograft harvest or biopsy. Microsurgery 2020;40:710-6.
45. Chiu DT, Strauch B. A prospective clinical evaluation of autogenous vein grafts used as a nerve conduit for distal sensory nerve defects of 3 cm or less. Plast Reconstr Surg 1990;86:928-34.
46. Mackinnon SE, Dellon AL. Clinical nerve reconstruction with a bioabsorbable polyglycolic acid tube. Plast Reconstr Surg 1990;85:419-24.
47. Ducic I, Fu R, Iorio ML. Innovative treatment of peripheral nerve injuries: combined reconstructive concepts. Ann Plast Surg 2012;68:180-7.
48. Parker BJ, Rhodes DI, O’Brien CM, Rodda AE, Cameron NR. Nerve guidance conduit development for primary treatment of peripheral nerve transection injuries: a commercial perspective. Acta Biomater 2021;135:64-86.
49. Schlosshauer B, Dreesmann L, Schaller HE, Sinis N. Synthetic nerve guide implants in humans: a comprehensive survey. Neurosurgery 2006;59:740-7.
50. Ağaoğlu G, Kayikçioğlu A, Sargon M, Erk Y, Mavili E. Comparison of immune response to nerve allograft segments in fetal and adult rabbits: a histological study. Ann Plast Surg 2000;44:398-404.
51. Evans PJ, Mackinnon SE, Levi AD, et al. Cold preserved nerve allografts: changes in basement membrane, viability, immunogenicity, and regeneration. Muscle Nerve 1998;21:1507-22.
52. Pollard JD, McLeod JG. Fresh and predegenerate nerve allografts and isografts in trembler mice. Muscle Nerve 1981;4:274-81.
53. Strasberg SR, Mackinnon SE, Genden EM, et al. Long-segment nerve allograft regeneration in the sheep model: experimental study and review of the literature. J Reconstr Microsurg 1996;12:529-37.
54. Midha R, Mackinnon SE, Becker LE. The fate of Schwann cells in peripheral nerve allografts. J Neuropathol Exp Neurol 1994;53:316-22.
55. Jacoby W, Fahlbusch R, Mackert B, Braun B, Rolle J, Schnell J. Bridging of peripheral nerve defects with lyophilised and disantigenised homologous grafts. Munch Med Wochenschr 1970;112:586-9.
56. Zalewski AA, Gulati AK. Evaluation of histocompatibility as a factor in the repair of nerve with a frozen nerve allograft. J Neurosurg 1982;56:550-4.
57. Johnson PC, Duhamel RC, Meezan E, Brendel K. Preparation of cell-free extracellular matrix from human peripheral nerve. Muscle Nerve 1982;5:335-44.
58. Hall SM. Regeneration in cellular and acellular autografts in the peripheral nervous system. Neuropathol Appl Neurobiol 1986;12:27-46.
59. Lovati AB, D’Arrigo D, Odella S, Tos P, Geuna S, Raimondo S. Nerve repair using decellularized nerve grafts in rat models. A review of the literature. Front Cell Neurosci 2018;12:427.
60. Whitlock EL, Tuffaha SH, Luciano JP, et al. Processed allografts and type I collagen conduits for repair of peripheral nerve gaps. Muscle Nerve 2009;39:787-99.
61. Karabekmez FE, Duymaz A, Moran SL. Early clinical outcomes with the use of decellularized nerve allograft for repair of sensory defects within the hand. Hand (N Y) 2009;4:245-9.
62. Brooks DN, Weber RV, Chao JD, et al. Processed nerve allografts for peripheral nerve reconstruction: a multicenter study of utilization and outcomes in sensory, mixed, and motor nerve reconstructions. Microsurgery 2012;32:1-14.
63. Cho MS, Rinker BD, Weber RV, et al. Functional outcome following nerve repair in the upper extremity using processed nerve allograft. J Hand Surg Am 2012;37:2340-9.
64. Guo Y, Chen G, Tian G, Tapia C. Sensory recovery following decellularized nerve allograft transplantation for digital nerve repair. J Plast Surg Hand Surg 2013;47:451-3.
65. Taras JS, Amin N, Patel N, McCabe LA. Allograft reconstruction for digital nerve loss. J Hand Surg Am 2013;38:1965-71.
66. Taras JS, Jacoby SM, Lincoski CJ. Reconstruction of digital nerves with collagen conduits. J Hand Surg Am 2011;36:1441-6.
67. Isaacs J, Safa B. A preliminary assessment of the utility of large-caliber processed nerve allografts for the repair of upper extremity nerve injuries. Hand (N Y) 2017;12:55-9.
68. Rinker B, Zoldos J, Weber RV, et al. Use of processed nerve allografts to repair nerve injuries greater than 25 mm in the hand. Ann Plast Surg 2017;78:S292-5.
69. Zhu S, Liu J, Zheng C, et al. Analysis of human acellular nerve allograft reconstruction of 64 injured nerves in the hand and upper extremity: a 3 year follow-up study. J Tissue Eng Regen Med 2017;11:2314-22.
70. Carlson TL, Wallace RD, Konofaos P. Cadaveric nerve allograft: single center’s experience in a variety of peripheral nerve injuries. Ann Plast Surg 2018;80:S328-32.
71. Nietosvaara Y, Grahn P, Sommarhem A. Failed peripheral nerve reconstruction with processed nerve allografts in three patients. J Hand Surg Eur Vol 2019;44:318-20.
72. Safa B, Shores JT, Ingari JV, et al. Recovery of motor function after mixed and motor nerve repair with processed nerve allograft. Plast Reconstr Surg Glob Open 2019;7:e2163.
73. Leckenby JI, Furrer C, Haug L, Juon Personeni B, Vögelin E. A retrospective case series reporting the outcomes of avance nerve allografts in the treatment of peripheral nerve injuries. Plast Reconstr Surg 2020;145:368e-81e.
74. Safa B, Jain S, Desai MJ, et al. Peripheral nerve repair throughout the body with processed nerve allografts: results from a large multicenter study. Microsurgery 2020;40:527-37.
75. Peters BR, Wood MD, Hunter DA, Mackinnon SE. Acellular nerve allografts in major peripheral nerve repairs: an analysis of cases presenting with limited recovery. Hand (N Y) 2021:15589447211003175.
76. Isaacs J, Browne T. Overcoming short gaps in peripheral nerve repair: conduits and human acellular nerve allograft. Hand (N Y) 2014;9:131-7.
77. Means KR Jr, Rinker BD, Higgins JP, Payne SH Jr, Merrell GA, Wilgis EF. A multicenter, prospective, randomized, pilot study of outcomes for digital nerve repair in the hand using hollow conduit compared with processed allograft nerve. Hand (N Y) 2016;11:144-51.
78. Souza JM, Purnell CA, Cheesborough JE, Kelikian AS, Dumanian GA. Treatment of foot and ankle neuroma pain with processed nerve allografts. Foot Ankle Int 2016;37:1098-105.
79. Rinker BD, Ingari JV, Greenberg JA, Thayer WP, Safa B, Buncke GM. Outcomes of short-gap sensory nerve injuries reconstructed with processed nerve allografts from a multicenter registry study. J Reconstr Microsurg 2015;31:384-90.
80. Rinker B, Vyas KS. Clinical applications of autografts, conduits, and allografts in repair of nerve defects in the hand: current guidelines. Clin Plast Surg 2014;41:533-50.
81. Ducic I, Safa B, DeVinney E. Refinements of nerve repair with connector-assisted coaptation. Microsurgery 2017;37:256-63.
82. Azouz SM, Lucas HD, Mahabir RC, Noland SS. A survey of the prevalence and practice patterns of human acellular nerve allograft use. Plast Reconstr Surg Glob Open 2018;6:e1803.
83. Rodriguez-Collazo E, Laube Ward K. Conduit-assisted allograft neurorrhaphy for the treatment of intractable lower extremity pain due to neuromas-in-continuity. Plast Reconstr Surg Glob Open 2021;9:e3867.
84. MacKay BJ, Cox CT, Valerio IL, et al. Evidence-based approach to timing of nerve surgery: a review. Ann Plast Surg 2021;87:e1-e21.
85. Atchabahian A, Doolabh VB, Mackinnon SE, Yu S, Hunter DA, Flye MW. Indefinite survival of peripheral nerve allografts after temporary cyclosporine a immunosuppression. Restor Neurol Neurosci 1998;13:129-39.
86. Gulati AK. Evaluation of acellular and cellular nerve grafts in repair of rat peripheral nerve. J Neurosurg 1988;68:117-23.
87. Mackinnon SE, Hudson AR, Falk RE, Kline D, Hunter D. Peripheral nerve allograft: an immunological assessment of pretreatment methods. Neurosurgery 1984;14:167-71.
88. Silva J, Marchese GM, Cauduro CG, Debiasi M. Nerve conduits for treating peripheral nerve injuries: a systematic literature review. Hand Surg Rehabil 2017;36:71-85.
89. Chen MB, Zhang F, Lineaweaver WC. Luminal fillers in nerve conduits for peripheral nerve repair. Ann Plast Surg 2006;57:462-71.
90. Boriani F, Fazio N, Bolognesi F, Pedrini FA, Marchetti C, Baldini N. Noncellular modification of acellular nerve allografts for peripheral nerve reconstruction: a systematic critical review of the animal literature. World Neurosurg 2019;122:692-703.e2.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Buncke G. Peripheral nerve allograft: how innovation has changed surgical practice. Plast Aesthet Res 2022;9:38. http://dx.doi.org/10.20517/2347-9264.2022.08
AMA Style
Buncke G. Peripheral nerve allograft: how innovation has changed surgical practice. Plastic and Aesthetic Research. 2022; 9(5): 38. http://dx.doi.org/10.20517/2347-9264.2022.08
Chicago/Turabian Style
Buncke, Gregory. 2022. "Peripheral nerve allograft: how innovation has changed surgical practice" Plastic and Aesthetic Research. 9, no.5: 38. http://dx.doi.org/10.20517/2347-9264.2022.08
ACS Style
Buncke, G. Peripheral nerve allograft: how innovation has changed surgical practice. Plast. Aesthet. Res. 2022, 9, 38. http://dx.doi.org/10.20517/2347-9264.2022.08
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 43 clicks
Cite This Article 43 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.