Use of the multiplane internal mastopexy for ptosis correction revision-augmentation mammoplasty
Abstract
Aim: Augmentation mammoplasty is a commonly performed procedure with a high satisfaction rate. Multiplane pocket was described for simultaneous internal mastopexy and augmentation using inframammary crease incision for selected primary and secondary mammoplasties. The use of the technique is presented with a larger experience for correction of ptosis in a patient presenting for revision surgery following subglandular augmentation mammoplasty.
Methods: A retrospectively collected data were analyzed using the Excel Spread Sheet. A total of 25 patients had multiplane augmentation with the internal mastopexy following augmentation mammoplasty in subglandular pocket. Data of 25 patients who had their revision surgery in multiplane were analyzed.
Results: The group included 25 patients with a mean age of 36.6 years (range: 25-54 years) with mean implant duration of 6.4 years (range: 1.5-13 years). Twenty-three of the patients were nonsmokers, 1 smoker and 1 patient’s smoking status was not mentioned. Eighteen patients presented with grade I capsular contracture, 3 patients with grade II contracture and 4 patients had a combination of grade I and II capsular contracture. Pseudoptosis was present in 6, class B ptosis in 6, A/B ptosis in 3, water-down deformity in 5 and rippling in 5 patients. Average preoperative size of implant used initially was 334.4 mL (range: 250-340 mL) and the mean implant size selected for revision surgery was 416 mL (range: 260-525 mL). Mean follow-up time was 18 months (range: 6-48 months). Of 25 patients, 21 had a bilateral procedure whereas the technique was used unilaterally in 4 patients for the correction of asymmetry. All patients had a single dose of intravenous antibiotics and followed by an oral course for 5 days, there was no infection noted in the series. In the current series, no patient required revision surgery following the multiplane internal mastopexy.
Conclusion: Multiplane internal mastopexy can be useful in selected cases of revisionary augmentation mammoplasty.
Keywords
Introduction
Primary and revision-augmentation mammoplasty is a commonly performed procedure. The incidence of implant related mammoplasties for both primary and revision mammoplasties is on the rise and is due to the information available on the product, premarket surveys, enhanced implant safety, and regular quality checks.[1] It is not surprising that augmentation mammoplasty is one of the most commonly performed procedure and in 2012 alone 330,631 implant related mammoplasties were performed in USA.[2] However, the data represent implant-related surgeries performed both for primary and secondary surgery making it difficult to obtain number of secondary or revision mammoplasties performed during the same period of time.[3] Secondary procedures following mammoplasties can be divided into early or late. Early reasons for redo are uncommon and include infection and bleeding, but both may necessitate urgent attention or an emergency surgery.[3] The late complications are more common and do not generally constitutes emergency procedures.[1] These late complications include capsular contracture, change for implant size and shape, implant rippling, asymmetry of shape, implant rupture or ptosis. Revision surgery for these complications is either performed alone or in combination, depending on the presentation. Revision surgery in these complications often requires a change of implant. These complications and their corrective surgery can often be challenging for a surgeon and requires a detailed history of previous surgery, thorough examination, patient’s wishes for a desired results and a well thought, clear and meticulous plan. Patients presenting with these complications may have an associated ptosis that may require simultaneous mastopexy using either periareolar, vertical scar, wise pattern markings or their modifications.[4] The pocket for implant replacement can be subglandular, partial submuscular, subfascial or muscle splitting.[5] Multiplane internal mastopexy (MIM) or use of more than one pocket for augmentation and the internal mastopexy using an inframammary crease has been described and was used in selected patients. The technique allows avoidance of scar in the border line ptosis and especially suits those patients who are not interested in obvious scarring associated with conventional nipple mobilization. The technique was used for primary cases with a limited experience in revision mammoplasties.[6] The current article describes a larger experience in selected patients who had their initial augmentation mammoplasty in subglandular pocket. The technique allows an addition to the armamentarium of surgeons for patients who present for revision surgery with minor to moderate ptosis following augmentation in subglandular pocket.
Methods
A retrospectively collected data were analyzed in the Excel Spread Sheet (Microsoft). Between January 2008 and October 2013, 25 patients had MIM following their augmentation mammoplasty in subglandular pocket. Relevant data of 25 patients who had their revision surgery in multiplane pocket was further analyzed. Six months postoperatively, patients were asked whether they were very satisfied, satisfied or dissatisfied with the outcome of the surgery.
Examination
All patients are examined in standing and supine position. Supine position allows any excess pocket extension in lateral dimension. Breast ptosis with or without upper and medial quadrant rippling is an indication for the conversion technique. Lower and lower lateral skin envelope rippling is unlikely to be improved by muscle splitting conversion or any other submuscular technique. Degree of capsular contracture is noted, and information is gathered about the size and profile of the existing implants.
Technique
All procedures are performed under general anesthetic with full muscle relaxation and as a day case. Patients were placed in the supine position with arms abducted in less than 90°. Inframammary crease was used for the pocket access.
After explantation of the device, pocket was examined for its dimensions and extent and nature of the capsule. In grade 1 or 2 capsular contractures, only lower pole capsulotomies were performed. Breast presenting with advanced capsules, partial or complete capsulectomy was performed. The next step was to identify Pectoralis Major and marked by light scoring on the posterior layer of capsule, starting from the junction of the middle and lower third of sternum medially and going up and laterally to the anterior axillary fold. This line of the muscle split was transposed and marked anteriorly by scoring the anterior layer of the capsule, this scoring should ideally be at or just below the nipple level in the midline.
Pectoralis split was commenced medially at the junction of the middle and lower third of the sternum. Pectoralis muscle is pinched and lifted off the sternal margin using Gillis toothed forceps, and a small incision is made using cutting diathermy. The incision should be large enough to allow index finger, and once the finger is inserted, submuscular dissection was performed using index finger extending up to the 2nd intercostal space and to the anterior axillary line laterally. Once the submuscular dissection is completed, incision is usually large enough allowing the breast retractor to be inserted with its distal end pointing towards anterior axillary fold. Muscle split begins medially using cutting diathermy, and once the split gets closer to the anterior axillary fold, the dissection is slowed down. Here, coagulation of the muscle is performed before splitting or cutting it up further. The maneuver avoids inadvertent bleeding resulting from damage to thoracoacromial axis branches.
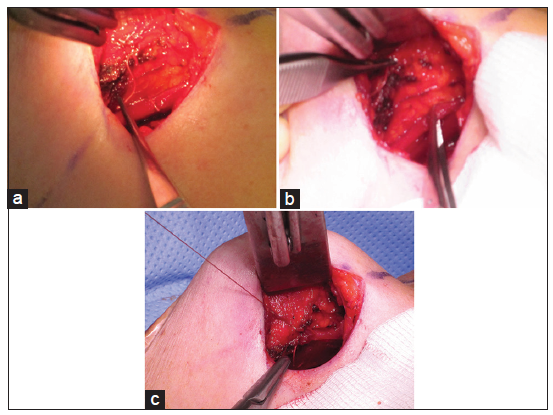
Once pectoralis split is completed, the lateral capsulotomy is extended upward to join the lateral extent of muscle split. The lower border of the upper split pectoralis is now stitched to the breast envelope below the marked and scored anterior capsule using 2-0 Vicryl interrupted stitches vicryl (Ethicon) [Figure 1a-c]. The level at which the anterior capsule is stitched to the lower border of upper split pectoralis depends on the degree of ptosis but should not be less than 2 cm [Figure 1a]. Hemostasis is achieved, and a preoperatively selected implant is placed in its new pocket. Before wound closure and once the new implant is in place, the flat of the hand is run over the skin envelope sliding the skin inferiorly over the mound of the implant. Creation of a crease or fold, due to an internal stitch placed too low inside the skin envelope, is an indication of replacing the stitch to a little higher position. The head end of the table can also be raised to confirm the fold, which depends from the anchoring suture inside and can also be felt by doing a bimanual digital examination using index finger and thumb through inframammary incision. The incorrect positioning of the stitch is not visible without this maneuver when patient is lying in a supine position. More commonly, the sliding or tilting of the table may show minor puckering or dimpling of the skin envelope that can be left alone. Once suture incorrect position is established, implant is removed, and sutures are repositioned at a slightly higher level, using previously scored anterior capsule as a reference point and implant is replaced. The procedure can be repeated to assess the position of the newly position stitch.
Figure 1. (a) Intraoperative picture showing scored anterior skin envelope marked with a Vicryl suture held at its loose end. Below and to the right in the picture, lower edge of the upper split muscle is also marked with Vicryl suture; (b) anterior capsule/wall of the pocket on the left and lower free edge of upper split muscle on the right, held separately in forceps before suturing; (c) tied suture knot between the marked anterior capsule/wall of envelope and lower edge of the upper split pectoralis major in place
Wound closure is done using continuous 2-0 Vicryl to deep fascial layer, subcutaneous 3-0 Vicryl interrupted and intradermal 4-0 continuous Monocryl stitch (Mononcryl (Ethicon). Once the wound closed and dressed, external support to breast envelope is provided using adhesive dressings. The external supportive dressings are applied starting from the lower pole and pulling, supporting, and stabilizing the breast envelope at a higher and desirable position. Support garments are applied, and patients are discharged on the same day.
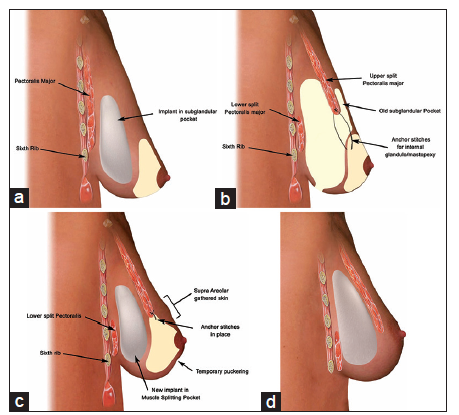
Postoperatively, there is often some puckering of the skin envelope due to internal stitches. This puckering almost always disappears within 4-6 weeks after surgery [Figure 2a-d].
Figure 2. (a) Illustration showing side profile of an implant in subglandular pocket; (b) illustration showing dissected muscle splitting pocket with anchoring stitch placed between lower border of upper split muscle and breast envelope at a level just under the nipple areolar complex (NAC). Note the relative position of the sixth rib and the nipple areolar complex; (c) illustration showing the implant placed in muscle splitting pocket with a tied anchoring stitch between muscle and breast envelope. Note the puckering of the skin below NAC, gathered skin above NAC and relative position of the sixth rib; (d) implant in its new muscle splitting position with puckering and skin gathering settled
Patients are reviewed 2 and 4 weeks later to check for wound healing. Patients are generally allowed to drive and return to work 10 days following surgery. Patients involved in physically demanding work are advised to take 3 weeks off work.
Results
The group included 25 patients with an average age 36.6 years (range: 25-54 years) with mean implant duration 6.4 years (range: 1.5-13 years), 23 of the patients were nonsmokers, 1 smoker and 1 patient’s smoking status was not mentioned. Eighteen patients presented with grade I capsular contracture, 3 patients with grade II ptosis and 4 patients had a combination of grade I and II capsular contracture. Pseudoptosis was present in 6, class B ptosis in 6, A/B ptosis in 3, sliding ptosis or water-down deformity in 5 and rippling in 5 patients. Average size implant from the initial surgery was 334.4 mL (range: 250-340 mL) and the mean implant size selected for revision surgery was 416 mL (range: 260-525 mL). Of 25 patients, 21 patients had a bilateral procedure whereas the technique was used unilaterally in 4 patients for the correction of asymmetry. Mean follow-up time was 18 months (range: 6-48 months). All patients had a single intravenous dose of predominantly Augmentin and followed by an oral course for 5 days, there was no infection noted in the series. In the current series, no patient required revision surgery following MIM. Patient satisfaction data were retrieved from the spreadsheet, 20 patients (80%) were very satisfied with the outcome and 5 patients were satisfied with the results, no patients showed dissatisfaction with the procedure.
Discussion
Augmentation mammoplasty is primarily done either in front or behind the muscles.[7,8] All modifications are the extensions of these 2 primary pockets. The existence of these 2 planes in each subject has the potential of these 2 pockets being used at the same time. Breast ptosis is the slackening and downward descent of the nipple areolar complex (NAC) and breast envelope in relation to the inframammary crease as defined by Regnault.[9] The ptosis correction is commonly performed by using periareolar, vertical scar or wise pattern markings, depending on the presentation of the breast, wishes of the patient and the experience of the surgeon.[4,10] Markings for mastopexy used are independent of the pocket, and choice of the pocket can be independent of markings.[4,5] Even though breast lies in front of the muscle, most of the patients can have satisfactory breast volume restoration or further enhancement in partial submuscular pocket. Muscle splitting augmentation is a pocket that can be used primarily where an implant is placed behind the muscle in its upper part and front of the muscle in lower part of the breast pocket. The advantage of this technique is that the results have a more natural appearance with the advantage of muscle support in the ever-changing upper breast envelope.[11-14] The use of muscle splitting pocket is also described for secondary procedures where partial submuscular and subglandular pockets are converted into muscle splitting pocket.[15-18] Muscle splitting augmentation allows an immediate natural outcome, and the longevity of the results has been reported with a satisfactory outcome and reduced revision rate when compared with other commonly used techniques.[19] The Multiplane technique is a procedure where muscle splitting procedure is used for submuscular implant placement and subglandular pocket is used for breast lift or mastopexy. In a previously published article, postoperative suprasternal notch to NACs distance was reported to be reduced when augmentation mammoplasty with multiplane technique distances was compared with its preoperative measurements.[6] On the other hand, suprasternal notch to NAC distances was increased postoperatively following mammoplasty in subglandular and partial submuscular augmentation, with their respective preoperative measurements.[6] The changes and distances are measured more following sub glandular than sub muscular mammoplasty and are primarily due to the support of an extra muscle layer added to the breast skin envelope when sub muscular pocket is used.[20] In current series, average size of the implants used for the initial procedure was 334 mL as compared to 416 mL selected for the revision cases, a trend normally seen in revision mammoplasties.[3,21] In revisionary aesthetic mammoplasty, patients almost always request for a larger implant size. The larger size of implant used in MIM acts as an internal splint and put an even pressure on the skin envelope that helps to stabilize the draped skin in this form of mastopexy. This internal splinting is supported by external supportive dressings while envelope is settling down in its relocated position. Since this form of mastopexy does not involve skin reduction, necessary tightening of the skin envelope is achieved when a larger implant is used. When subglandular to muscle splitting submuscular site change or pocket changed was performed for rippling alone without an internal mastopexy, and in a patient without ptosis or skin excess, moderate reduction in implant sizes did not show any untoward skin laxity or puckering, when skin envelope finally settled down. However, when a patient presents with breast ptosis and skin envelope excess and wishes to choose a smaller implant for replacement or go down in breast cup size, conventional skin reduction mastopexy with NAC mobilization is the recommended procedure of choice.
The multilane technique was initially described for primary cases with a limited experience for ptosis and rippling correction in patients following augmentation mammoplasty in subglandular pocket.[6] The concept of the use of the two planes is not new and submuscular pocket for implant placement, and subglandular pocket dissection for breast envelope draping has been described in the past. However, the draping of mobilized breast envelope in Multiplane pocket is secured internally while the technique described by Hilton Becker relied on external support using dressings and adhesives bandages alone. Becker[22] also used an expander prosthesis with an occasional combination of periareolar mastopexy in certain cases. Similarly, implant site change or pocket change from subglandular to submuscular, submuscular to neopectoral or subfascial is not new, and the idea has been frequently used and documented.[17] Subglandular, dual plane and partial submuscular to muscle splitting biplane has also been reported for revisionary surgery with acceptable long-term results in various forms of aesthetic breast revisionary surgery.[23-25] With the high number of aesthetic revisionary performed today, preexisting pockets conversion to muscle splitting biplane submuscular pocket, a combination of submuscular and subglandular pocket, remains a suitable option. The use of acellular dermal matrix (ADM) in revisionary aesthetic breast surgery has introduced another horizon to deal with various problems encountered in secondary aesthetic breast procedures.[26] In small case series of three patients, the preemptive use of ADM in lower pole of poor quality breast tissue has been described for internal mastopexy in order to minimize the risk of ptosis in primary cases and in one patient ADM was used for internal mastopexy to correct an established ptosis following augmentation mammoplasty with mastopexy.[27] The report is promising, however, a larger series with longer follow-up will be required to evaluate the efficacy of the technique. In a review article regarding the use of biological and synthetic meshes used in implants surgery, the use of these materials was predominantly limited to breast reconstruction following mastectomy. Even though the use of ADM has gained some popularity following the safety of skin or nipple sparing mastectomies, a high number of seroma, higher infection rate and the cost of the product has restricted its use in aesthetic secondary augmentation mammoplasties.[28] The use of long-term synthetic mesh has shown more promising results in breast reconstructive and cosmetic surgery, however, the available data of its use in primary or secondary augmentation mammoplasties and augmentation mastopexies are limited.[29] Breast implant capsule flaps are reported quite frequently, and various techniques have been described for its use in primary and secondary cosmetic and reconstructive surgeries with very good results.[30] However, the use of these implants flaps, biological matrices and synthetic meshes is limited to support breast envelope, following mastectomies. These alternatives are also aimed to correct implant malposition, redefine or reconstruct inframammary crease both in cosmetic, as well as reconstructive surgery.[28-30] The author also has described the use of existing capsules to recreatenew pockets for the correction of bottoming out, double bubble deformity and animation deformities.[15-16] The use of these materials or techniques as supplementary breast supporting products are limited to reinforce or reconstruct breast dimensions, to support weak breast envelope or to prevent explant exposures but without the ability of reversing the NAC-inframammary crease (IMC) relationship seen following breast ptosis and as defined by Regnault.[9] On the other hand, MIM has a unique ability to restore or improve the altered NAC-IMC relationship and without extra scarring in selected cases.
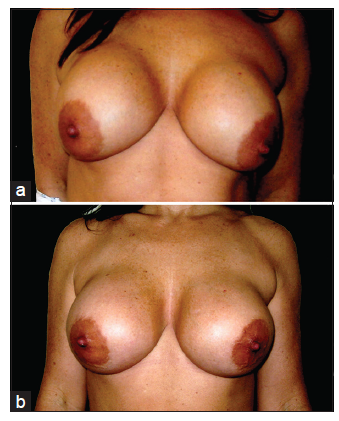
The augmentation mammoplasty with the internal mastopexy in prepectoral or subglandular pocket in revisionary cases has a marked advantage over simultaneous augmentation mammoplasty with the internal mastopexy in primary cases. In primary cases, especially those presenting with large size breasts, initial acceptable results may later show sliding ptosis of the NAC over the mound of the implant. However, when MIM is performed in secondary cases, initial mammoplasty in sub glandular pocket has generally compressed the breast tissue over a period of time. This comparatively thinner layer of the breast envelope is far easier to be elevated, anchored, and secured at a higher position on the muscle, in a predictable way and with longevity of results. The current series has a mean follow-up of 18 months (range: 6-48 months) with high satisfactory results. Despite the much desired scar-less MIM in selected cases, a longer follow-up will be desirable for a comparison with other conventional mastopexy techniques used today. The obvious disadvantage of MIM is the indirect measurements for a nipple areolar repositioning as compared to precise markings used in conventional skin reducing and nipple areolar mobilizing techniques. Minor asymmetry, if present, is well-tolerated and accepted by patients due to the normally occurring asymmetries in breast and NAC.[31-33] The other disadvantage with MIM is that the technique does not allow areolar reduction that may overshadow the true lift achieved in such cases presenting with large size NAC [Figure 3a and b]. In some cases, breast envelope puckering along the lower edge of the upper split muscle can be obvious in the early period of healing but resolves in time [Figure 4]. The added use of external supportive dressings stabilizes the mobilized skin envelope and conceals the temporary puckering that can be worrying for the patients in the early stage of healing. Removal of the dressings in 2 weeks’ time almost always leaves behind a smoother skin envelope and muscle expansion and relaxation allows the implant to settle with more natural three-dimensional results [Figures 5 and 6].
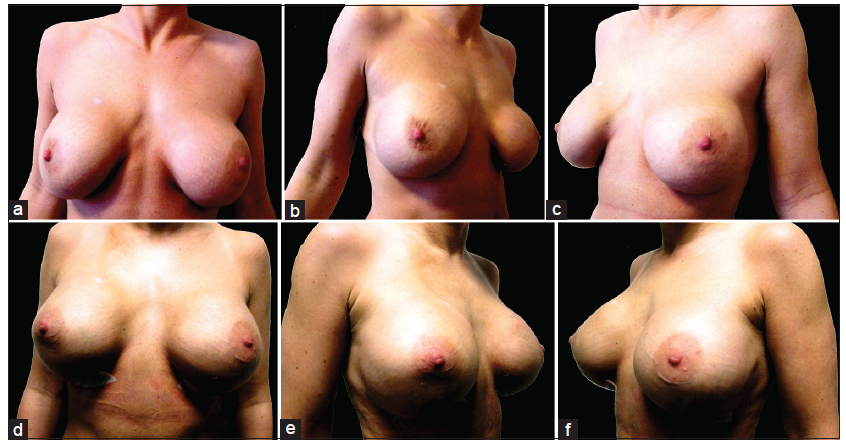
Figure 3. (a) Preoperative anterior view of a 39-year-old patient 9 years following her mammoplasty in subglandular pocket. Patient had 260 mL high profile Perouse Plastie (540T3) cohesive gel silicone implants with preoperative sternal notch to nipple areolar complex (NAC) of 24.5 cm; (b) three months following augmentation mammoplasty using multiplane technique. Patient had 380 g MHP CUI Allergan Prosthesis. The improvement of ptosis is masked by a large size NAC even after postoperative reduction in the sternal notch to NAC distance to 23.5 cm
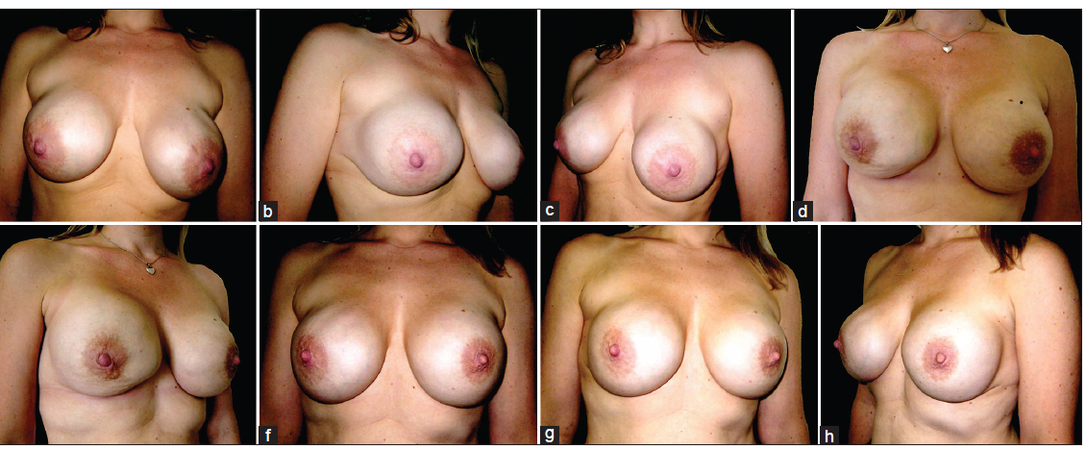
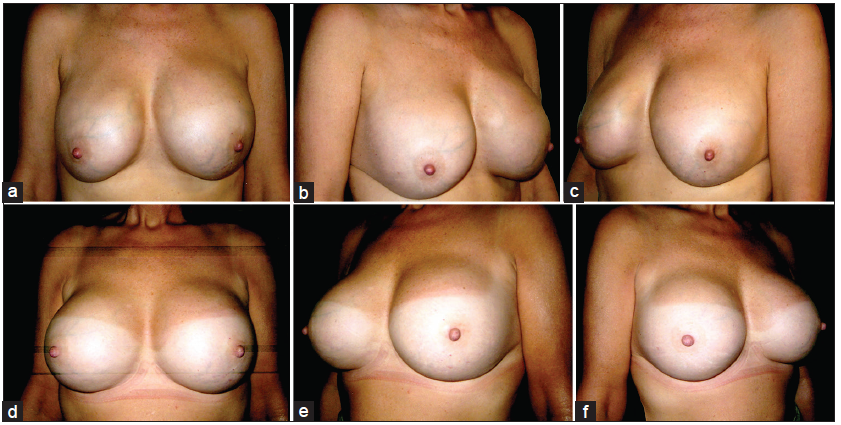
Figure 4. (a-c) Preoperative views of a 29-year-old patient who had 380 mL cohesive gel silicone implants placed in subglandular pocket with preoperative suprasternal notch to nipple areolar complex (NAC) distance of 23 cm right and 24 cm on left side. Patient presented with marked ptosis, rippling and asymmetry of breasts; (d-e) two months following the corrective surgery using 460 mL cohesive gel silicone. There is marked puckering of the right breast during early postoperative period; (f-h) postoperative pictures taken 5 months following surgery with good breast lift and symmetry. Her postoperative suprasternal notch to NAC distance was measured 20.5 cm both sides
Figure 5. (a-c) A 43-year-old patient, following augmentation mammoplasty with 430 mL Poly Implant Prothese implants, in subglandular pocket. Patient presented with rippling, breast asymmetry associated with noticeably lowered nipple areolar complex (NAC) along with short NAC to the inframammary crease distance. Her preoperative suprasternal notch to NAC distance was 24.5 bilaterally; (d-f) ten months following the revision surgery with nice symmetry of the breasts and an adequate NAC to inframammary crease interrelationship. Her postoperative suprasternal notch to NAC distance was measured as 24 cm bilaterally
Figure 6. (a-c) Preoperative views of a 41-year-old patient following augmentation mammoplasty in sub glandular plane with 350 mL cohesive gel silicone implants. Patient presented with ptosis, rippling and absolute lack of the upper pole fullness; (d-f) postoperative views 6 weeks after revision surgery in multiplane internal mastopexy using 350 mL cohesive gel silicone implants. Patient showing upper pole fullness, lack of rippling along with rejuvenated breasts appearance
Even though the study did not include a very large number of patients, the outcome showed a very high satisfaction rate. A larger series with a longer follow-up, comparison of breast morphometrics with other conventional skin reducing and nipple mobilizing mastopexies will be desirable.
The technique allows avoidance of external scars in selected patients and can be a good choice especially in those who are not keen on conventional external scarring. With a mean follow-up of 18 months (range: 6-48 months) all patients had an acceptable results, and no further corrective surgery has been performed in the series analyzed.
Financial support and sponsorship
Nil.
Conflict of Interest
There are no conflicts of interest.
REFERENCES
1. Khan UD. Combining muscle-splitting biplane with multilayer capsulorraphy for the correction of bottoming down following subglandular augmentation. Eur J Plast Surg 2010;33:259-69.
2. The American Society for Aesthetic Plastic Surgery Cosmetic Surgery National Data Bank Statistics; 2012. Available from: http://www.cynosureaustralia.com/wp-content/blogs.dir/7/2013/05/ASAPS-2012-Stats.pdf. [Last accessed on 2014 Sep 10].
3. Khan UD. Secondary augmentation mammoplasties and periprosthetic infection. A three year retrospective review of 92 secondary mammoplasties performed by a single surgeon. Aesthet Surg J 2012;32:465-73.
4. Khan UD. Vertical scar mastopexy with a cat's tail extension for prevention of skin redundancy: an experience with 17 consecutive cases after mastopexy and mastopexy with breast augmentation. Aesthetic Plast Surg 2012;36:303-7.
5. Khan UD. Augmentation mastopexy in muscle-splitting biplane: outcome of first 44 consecutive cases of mastopexies in a new pocket. Aesthetic Plast Surg 2010;34:313-21.
6. Khan UD. Multiplane technique for simultaneous submuscular breast augmentation and internal glandulopexy using inframammary crease incision in selected patients with early ptosis. Eur J Plast Surg 2011;34:337-43.
7. Cronin TD, Gerow FJ. Augmentation mammoplasty: new "natural feel" prosthesis. In: Thomas Ray Broadbent, American Association of Plastic Surgeons, American Society of Plastic and Reconstructive Surgeons, International Confederation for Plastic Surgery, Transactions of the International Society of Plastic Surgeons, editors. Transactions of the Third International Congress of Plastic Surgery. Amsterdam: Excerpta Medica; 1964. pp. 41-9.
10. Khan UD. Vertical scar with the bipedicle technique: a modified procedure for breast reduction and mastopexy. Aesthetic Plast Surg 2007;31:337-42.
12. Khan UD. Subglandular to muscle splitting biplane conversion for revision augmentation mammoplasty. In: Mugea TT, Schifmann MA, editors. Aesthetic Surgery of the Brest. 1st ed. New York: Springer-Verlag, Berlin Heidelberg; 2015. pp. 535-41.
13. Khan UD. Selection of breast pocket using the pinch test in augmentation mammoplasty: can it be relied on in the long term? Aesthetic Plast Surg 2009;33:780-1.
14. Lang Stümpfle R, Figueras Pereira-Lima L, Alves Valiati A, da Silva Mazzini G. Transaxillary muscle-splitting breast augmentation: experience with 160 cases. Aesthetic Plast Surg 2012;36:343-8.
15. Khan UD. Dynamic breasts: a common complication following partial submuscular augmentation and its correction using muscle-splitting biplane technique. Aesthetic Plast Surg 2009;33:353-60.
16. Khan UD. High transverse capsuloplasty for the correction of malpositioned implants following augmentation mammoplasty in partial submuscular plane. Aesthetic Plast Surg 2012;36:590-9.
18. Baxter RA. Update on the split-muscle technique for breast augmentation: prevention and correction of animation distortion and double bubble deformity. Aesthetic Plast Surg 2011;35:426-9.
19. Khan UD. Muscle-splitting, subglandular, and partial submuscular augmentation mammoplasties: a 12-year retrospective analysis of 2026 primary cases. Aesthetic Plast Surg 2013;37:290-302.
20. Khan UD. Breast expansion in augmentation mammoplasty: comparative data analysis in submuscular and subglandular planes. J Muhammad Med Coll 2012;3:8-10.
21. Khan UD. Poly Implant Prothèse (PIP) incidence of device failure and capsular contracture: a retrospective comparative analysis. Aesthetic Plast Surg 2013;37:906-13.
22. Becker H, van Leeuwen JB. The correction of breast ptosis with the expander mammary prosthesis. Ann Plast Surg 1990;24:489-97.
23. Khan UD. Correction of acquired synmastia with muscle-splitting biplane implant replacement. Aesthetic Plast Surg 2009;33:605-10.
24. Khan UD. Breast autoinflation with sterile pus as a marker of implant rupture: single-stage treatment and outcome for five consecutive cases. Aesthetic Plast Surg 2009;33:58-65.
25. Khan UD. Unilateral breast autoinflation and intraprosthetic collection of sterile pus. An unusual operative finding of silicone gel bleed with silicone lymphadenitis. Aesthetic Plast Surg 2008;32:684-7.
26. Maxwell GP, Gabriel A. Acellular dermal matrix in aesthetic revisionary breast surgery. Aesthet Surg J 2011;31:S65-76.
27. Kornstein A. Porcine-derived acellular dermal matrix in primary augmentation mammoplasty to minimize implant-related complications and achieve an internal mastopexy: a case series. J Med Case Rep 2013;7:275.
28. Dieterich M, Faridi A. Biological matrices and synthetic meshes used in implant-based breast reconstruction - a review of products available in Germany. Geburtshilfe Frauenheilkd 2013;73:1100-6.
29. Becker H, Lind JG, 2nd. The use of synthetic mesh in reconstructive, revision, and cosmetic breast surgery. Aesthetic Plast Surg 2013;37:914-21.
30. Persichetti P, Segreto F, Pendolino AL, Del Buono R, Marangi GF. Breast implant capsule flaps and grafts: a review of the literature. Aesthetic Plast Surg 2014;38:540-8.
31. Khan UD. Breast and chest asymmetries: classification and relative distribution of common asymmetries in patients requesting augmentation mammoplasty. Eur J Plast Surg 2011;34:375-85.
32. Rohrich RJ, Hartley W, Brown S. Incidence of breast and chest wall asymmetry in breast augmentation: a retrospective analysis of 100 patients. Plast Reconstr Surg 2006;118:S7-13.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Khan UD, Riaz M. Use of the multiplane internal mastopexy for ptosis correction revision-augmentation mammoplasty. Plast Aesthet Res 2015;2:120-6. http://dx.doi.org/10.4103/2347-9264.157104
AMA Style
Khan UD, Riaz M. Use of the multiplane internal mastopexy for ptosis correction revision-augmentation mammoplasty. Plastic and Aesthetic Research. 2015; 2: 120-6. http://dx.doi.org/10.4103/2347-9264.157104
Chicago/Turabian Style
Khan, Umar Daraz, Muhammad Riaz. 2015. "Use of the multiplane internal mastopexy for ptosis correction revision-augmentation mammoplasty" Plastic and Aesthetic Research. 2: 120-6. http://dx.doi.org/10.4103/2347-9264.157104
ACS Style
Khan, UD.; Riaz M. Use of the multiplane internal mastopexy for ptosis correction revision-augmentation mammoplasty. Plast. Aesthet. Res. 2015, 2, 120-6. http://dx.doi.org/10.4103/2347-9264.157104
About This Article
Copyright
Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.