Review of cost and surgical time implications using virtual patient specific planning and patient specific implants in midface reconstruction
Abstract
Aim: Summarize the available data on midfacial virtual patient specific planning and patient specific implants, highlighting the financial costs and savings, and additionally emphasize the potential cost implications of transitioning to “in-house” virtual 3D modeling and printing.
Methods: Review of current literature.
Results: Current literature suggests cost benefits of virtual patient specific planning and patient specific implants in the form of improved ischemia time, better boney apposition between flaps, and reduced patient complications. This reduction of complications includes a reduction in blood loss and time spent in the intensive care unit from flap failure. Improved boney apposition results in a higher likelihood of boney union and a further reduction in failure and complications. Subjective benefits of virtual patient specific planning and patient specific implants are shown in the form of improved reconstructive surgeon mental energy. In-house production of 3D models and presurgical planning provides additional cost benefits for providers as they can produce viable models at a fraction of the price of that which is produced by industrial companies. Providers can also construct and use models in an expedient manner compared to industrial models, allowing for the opportunity to be utilized in more acute settings. The foundation of developing an in-house workflow is adequate funding, resources, and clinical volume. Facilities also must focus on appropriate quality and safety measures, as well as appropriate workflow development for adequate production of models.
Conclusion: Virtual patient specific planning and patient specific implants show benefits in midfacial reconstructive outcomes, resulting in realized financial and temporal gains for both patient and provider. These gains may be enhanced by moving to in-house planning and printing.
Keywords
INTRODUCTION
Virtual patient specific planning (VPSP) and patient specific implants (PSI) are emerging surgical tools used in reconstructive surgeries. The most frequently utilized applications are for complex boney reconstructions, including midfacial and maxillofacial reconstructive work. Purported benefits include shorter operative times, shorter ischemia times, more accurate reconstruction, the possibility of performing more complex reconstructions, and decreased complications [Table 1]. Offsetting these benefits is the cost of the materials, which can be thousands of dollars, as well as the cost of labor and time for planning. The required planning time of both the surgeon and the virtual planning team represents another drawback. This paper aims to summarize the available data on midfacial VPSP and PSI, highlighting the financial costs and savings, and additionally emphasize the potential cost implications of transitioning to “in-house” surgical modeling and virtual planning at the point of care [Table 1].
Overview of VPSP and PSI benefits (top). Comparative benefits of industrial vs. in-house VPSP and PSI (bottom)
| VPSP and PSI benefits | |
| Reduced operative and ischemia time | |
| More accurate and complex reconstructions | |
| Reduced complications and costs | |
| Industrial produced models | In-house produced models |
| The gold standard for model production | Promising developmental method |
| More complex reconstructions | Less complex reconstructions |
| Custom plates and guides | Reduced time and cost |
METHODS
A literature review was conducted using the PubMed database in August - October 2021 to search for relevant terms inclusive of that for virtual patient specific planning and patient specific implants. Relevant searched phrases included “3D-printing” as it relates to mid-facial and maxillofacial reconstruction for patient specific surgical guides and implants. Terms such as “computer aided design” (CAD), “computer aided manufacturing” (CAM), “midface”, “maxillofacial”, “customized”, “patient specific”, “models”, “in-house”, “cost”, and “time” were added to the search headings to include relevant articles. Articles were screened to include commercially produced and manufactured models as well as those developed in-house as they pertain to clinical use in human patients. All publications were written in English with the availability of full paper access. Included papers address outcomes such as the impact on patient and provider costs, time savings from reduced operative time as well as planning and hospitalization time, and reconstructive accuracy with resulting complication rates and clinical outcomes. Comparative articles were analyzed and synthesized with institutional procedural outcomes to demonstrate the viability of both commercial and in-house VPSP and PSI.
RESULTS
VPSP and PSI - what is available
VPSP is a broad term encompassing numerous components of digital preoperative planning. A preoperative planning session between the reconstructive surgeons and company engineers allows for the surgeon to plan the desired osseous cuts, and then design the bony reconstruction using patient-specific CT scans of the extremities. Potential options include the design of custom plates and cutting guides based on the anticipated resection, which allows for a cookie-cutter type approach where the free flap bone segments ideally perfectly fit into the defect with minimal alterations. 3D printed models can be sterilized and used in the OR for better understanding of the osseous anatomy or for bending plates, and mirror-image modeling allows for plate bending when exophytic tumors would otherwise prevent plate bending to the patient’s actual anatomy. Industrial options for VPSP and PSI, such as KLS Martin, Synthes, and Stryker, offer different planning packages for certain cost including custom plating, cutting guides for the resection or harvest site, and 3D models. Rather than being all-encompassing, the selection of these components can be a la carte and can be chosen based on specific patient needs. Depending on parts ordered, packages can range from thousands of dollars to ten of thousands of dollars, although costs can be somewhat reduced by a hospital contracting with one of the industrial companies.
Due to the high cost of VPSP and PSI, these procedures create a financial dilemma for providers and hospitals. Can the increased cost be justified due to decreased operative and ischemia time and lower complication rate? Also, what financial trade-offs exist to justify the use of this technology? With the potential for simplifying increasingly complex reconstructive approaches for midface defects, these techniques do require significant cost investment. Next, we aim to investigate the financial and clinical benefits of virtual patient specific planning as compared to the cost of this workflow.
Operative time cost reduction
Previous literature analysis shows a favorable benefit of VPSP and 3D printed models in the form of time and cost savings. Many hypothesize that with a reduced operative time, patients pay less, and hospitals spend less on operation times, which may offset some costs of VPSP and PSI. Current literature seeks to understand how exactly this reduced operative time results in cost savings for both patient and provider.
A review by Ballard et al.[1] poses strong evidence for the time-efficient benefits of virtual planning and 3D printed models in oral and maxillofacial procedures. They evaluated total operative minutes saved in patients undergoing oral and maxillofacial surgeries involving either 3D printed anatomical models or surgical guides in previously completed studies. A reduction in operation time was noted with the use of both the 3D anatomic model and 3D printed surgical guides when compared to controls undergoing the standard procedure[1]. Use of the anatomic model time reduction ranged from 41 to
A meta-analysis, performed by Tack et al.[2], also described the time-saving benefits of virtual planning in maxillofacial procedures with the use of custom implants, anatomical models, and surgical guides. While not entirely backed by numbers, as many authors did not include statistical analysis on operating time, most studies that mention maxillofacial surgery reported an advantage with each of the previously mentioned methods as it relates to operating time. Of the 42 studies that address operating room time, 16 address it as it relates specifically to maxillofacial surgery. They noted a reduction in surgical times of 42.0 min, 43.5 min (SD ± 24.52), and 60.33 min (SD ± 61.58) respectively[2]. The use of custom implants was included in 28 articles, 17 of which reported reduced operating times as well as improved medical outcomes for patients[2]. Implant shaping with anatomical models was addressed in 9 studies, 5 of which reported a time reduction in maxillofacial procedures while all 9 mentioned improved surgical outcomes[2]. The use of surgical guides was reported in 53 articles with respect to neurosurgery, dental surgery, spinal surgery, and maxillofacial surgery, of which 28 demonstrated a reduced operating room time[2]. The authors concluded that the use of 3D-printed trays and plates, surgical guides, and anatomical models are becoming increasingly utilized in maxillofacial surgery to enhance speed of procedures but also improve clinical outcomes[2]. As discussed later, in relation to cost benefits, there is an association between operating room time and surgical complications with patient and hospital costs.
While the previous data shows a significant benefit of VPSP in operative time savings, the true time saving benefit may be even greater. VPSP and PSI are often performed in particularly complex surgeries, some of which may not be possible without this impactful technology. This may result in a skewed overall time of virtual planning surgeries compared to conventional surgical methods as these cases are often of increased complexity. As such, the two comparison groups are difficult to fully compare, as data has been largely retrospective, and there are no randomized trials that have yet to be completed. However, as the literature suggests, operating time is shown to be reduced. Therefore, the true benefit of virtual patient specific planning overall, including in less complex cases, may be even greater. However, the question remains as to how this reduced time per case results in cost savings for patient and provider.
A review completed by Zweifel et al.[3] showed the operating room and cost savings that could be incurred by utilization of VPSP for fibular free flaps in mandibular reconstructions. They reported an average of
Hospitals and patients also incur fewer costs secondarily to the technical advancements and surgical benefits of virtual planning. Mazzola et al.[4] described the cost and time benefit of this in a retrospective matched-pair analysis of 138 patients undergoing microvascular free flap mandible or maxillary reconstruction. Patients were matched based on ablation, site of reconstruction, flap used, age, and surgical complexity score. Comparing length of stay, hospital costs, and operating room costs, VPSP was shown to be statistically significant in all categories[4]. Median hospital cost for VPSP patients showed a reduction of
As described below, significant clinical benefits of VSPS and PSI include reduced complications, and thus reduced provider and patient costs. Most concerning, flap failure and extrusion, are dreaded complications that produce substantial additional emotional and financial stressors for patients and their families, as well as the provider and system. These complications are considered multifactorial, but providers strive to reduce their rates of occurrence, such as with the use of high-quality custom plates and low-profile plates for maxillary and mandibular reconstructions. With the possible reduction in the rate of extrusion, this cost of a more expensive plate can be mitigated by the downstream benefit of successful flap acceptance and negating the costs of complications. As described below, this ability to produce these low-profile, custom engineered plates is an advantage that commercial production has over in-house production. Custom plates are often necessary for use in cases of prior trauma or tumor presence that is unable to be plated as the use of a titanium mill device for plate creation is not available for in-house operations. This makes commercial production an integral piece in reconstruction as in-house modeling currently does not have a good alternative.
By showing a reduced cost for patient and hospital, as well as a shorter length of stay for patients, both hospital and patient benefit financially. This poses a similar dilemma to that of hospitals only significantly benefiting financially from shorter operative times through room utilization. Hospitals would realize benefits largely if they were able to utilize the gained room and beds from shorter patient lengths of stay. The results may be similar to that of the initial dilemma, where benefits come in the form of administrative costs and thus provide minimal overall cost savings.
Clinical benefits
Current use of virtual planning and 3D printing technology suggests that patients and hospitals both incur lower costs through a transitive or indirect property, not directly related to the exact minutes saved from shortened operating times. Critically, the operative time saved is often ischemia time, which is the important time between flap harvest and revascularization. Since placement and orientation of the free flap bone segments requires 3D manipulation, it must be done before flap revascularization. VPSP can minimize the time it takes to get the bone segments cut properly and fit into the ablative defect. There are indirect cost savings associated with shortened ischemia time, better boney contact with flaps, and reduced blood loss in the form of lower rates of complications, flap failures, and time spent in the intensive care unit. This was demonstrated by Crawley et al.[5] and Eskander et al.[6] as they explored the properties of ischemia and operative time in their relation to head and neck complications and flap failures.
A review by Crawley et al.[5] studied 892 subjects and factors, including length of ischemia time, as predictors for flap failures. They reported an odds ratio of 1.062 (CI: 1.019-1.107; P = 0.004) and concluded that for every 5 min increased in ischemia time, there was an increased risk of free flap failure[5].
VPSP and PSI also appear to decrease complications due to improved accuracy and better boney contact with flaps. This was demonstrated by May et al.[7] and Swendseid et al.[8] as they each showed an optimization of boney apposition and its association with a reduction in flap-related complications.
The correlation of optimized boney apposition with reduced postoperative wound complications was also demonstrated by Swendseid et al.[9] as they analyzed 104 patients who underwent mandibular or maxillary reconstruction with osteocutaneous free flaps. They used postoperative CT or PET scans to identify osseointegration and nonunion to compare postoperative wound complications[9]. Their study showed that a larger gap in apposition (≥ 1 mm) between flaps is associated with an increased likelihood of failing to form a complete union (79% vs. 8%, P = 0.0009)[9]. Evidence of radiographic nonunion on CT/PET scan was associated with an increased likelihood of postoperative wound complications (40% vs. 19%, P = 0.025)[9]. The increased boney accuracy using VPSP likely prevents wound complications which ultimately results in a total cost reduction for patient and hospital.
While the above discusses VPSP and PSI in the context of maxillofacial procedures, there is a parallel utilization for dental implant formation and fixation. With previous methodology depended on surgeon experience and intraoperative trial-and-error with 2D imaging, VPSP offers a multitude of benefits in the outcomes and aesthetic outcomes for patients undergoing dental implantation with mandibular reconstruction[10]. With VPSP and the use of patient-specific chest CT scans and custom guides, surgeons are now able to assess and utilize the scapular tip, a boney segment not traditionally thought to accommodate dental implants[8]. Based on scapular thickness, the appropriate caliber dental implant can then be accommodated for the flap. Navarro Cuéllar et al.[10] noted similar benefits of VPSP to that of Swendseid et al.[9] with its association to better bony contact and reducing complication rates. However, they also include dental implant-specific benefits such as optimization of implant placement and dental alignment, which enhances facial symmetry, aesthetic contour, and function[10].
VPSP also improves dental implant outcomes transitively through enhanced rehabilitation. When performed simultaneously with maxillofacial free flap reconstructive surgery with computer-aided surgical design and computer-aided manufacturing (CAD/CAM) plates, patients receiving dental implantations are able to undergo functional dental rehabilitation earlier[11]. This is echoed by Allen et al.[12] as they describe the necessity of VPSP for dental implant placements based on its enhanced accuracy and precision of placements. They adopted a protocol to offer every osseous mandibular reconstructive patient preoperative VPSP to incorporate the placement of implants into the flap. This has allowed for earlier aesthetic restoration of the reconstructed mandibular segment as well as an increased number of patients able to expedite achieving oral rehabilitation[12].
Subjective benefits
Subjectively, there are significant benefits to both patient and physician with the use of virtual planning. Preplanning the boney cuts and reconstruction in a low-key environment, rather than in the operating room under ischemia, is significantly less stressful. Previously, with construction and cutting occurring on-the-fly or in the operating room, physicians were under mental and physical constraints from fitting the implant as well as increased ischemia time. As stated previously, with the correlation between ischemia time, complications, operative time, and costs, the stress of these factors can drain a physician of his mental energy.
Establishing in-house VPSP and PSI
Despite the financial benefits of VPSP and PSI, providers still search for ways to reduce both patient and hospital costs. An increasingly popular way to approach this is through the introduction of 3D model printing to hospitals and in-house surgical planning, as described by Daoud et al.[13]. While there was initially some concern regarding the quality of in-house produced models compared to those from industry, as technology improves, numerous clinical facilities have produced operative quality models for a fraction of the cost.
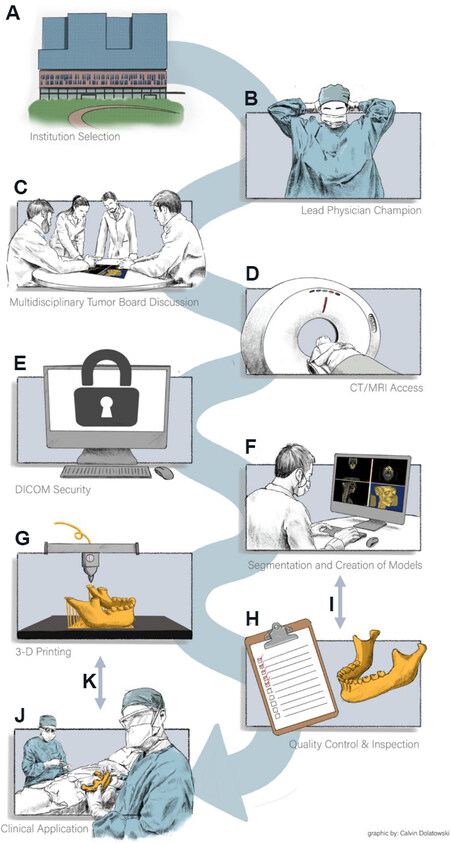
First, there are certain institutional considerations for determining if in-house VPSP and PSI are viable options. Some of the difficult aspects of setting up an in-house model are ensuring that an institution that has ample clinical volume, funding, and resources (both physical space and human resources) necessary to support it. While this review focuses on midface reconstruction, the institutional clinical volume necessary could come from other procedures and specialties that benefit from VPSP and 3D printing. The team that operates the in-house service must be a collaboration between clinicians and engineers. Below is the workflow of an in-house model [Figure 1] and the team member(s) that is responsible for each step[13]:
Figure 1. (A) Identify an institution that has the case volume and proper imaging equipment to support a self-sufficient 3D printing lab; (B) recruit a lead physician (surgeon, radiologist, etc.) to champion 3D printing efforts; (C) utilize tumor board, trauma cases, and other clinical scenarios that identify a wide range of patients who could benefit from 3D models; (D) ensure proper cross-sectional imaging (CT/MRI) Digital Imaging and Communications in Medicine (DICOM) access; (E) ensure DICOM storage for selected patients remaining under the institution’s HIPAA-protected firewall; (F) engineering team manipulates each patient’s specific imaging and creates 3D Computer-Aided Design (CAD); (G) final CAD model print execution per clinical requirements; (H) quality control for proper sterilization and model preparation for clinical use in the operating room; (I) feedback loop between Quality Control & Inspection and Segmentation and Creation of Models to improve future prints; (J) deliver the model for clinical application per institutional requirements; and (K) feedback loop between Clinical Application and 3D Printing to improve future prints. Reprinted by permission from: Springer Nature Journal of Materials Research (Establishing a point-of-care additive manufacturing workflow for clinical use, Daoud et al.[13]), 2021.
1. Primary physician interested in incorporating 3D models into their practice - otolaryngologists, neurosurgeons, facial plastic surgeons, etc.;
2. Identification of surgical candidate - team of physicians (possibly specialized tumor board for an oncology case);
3. Imaging patient anatomy (CT or MRI scans) - radiology;
4. Segmentation of anatomy and design of device from patient scans - engineering team with input from physicians;
5. 3D printing and post-processing of device - engineering team;
6. Delivery of device and sterilization - engineering handoff to surgical team.
Daoud et al.[13] goes into more depth about the technologies available for supporting in-house service. These include software and 3D printers for creating physical devices. Separate software is needed for many steps in the workflow including acquisition of the patient scans, segmenting the anatomy, designing device, and selecting settings for 3D printing (dependent on which 3D printer will be used). The 3D printer needs to be selected based on the type of material desired for the devices and other functionality such as full color or mechanical properties. Some common in-house 3D printers are materials extrusion, powder-based fusion, vat polymerization, and droplet-based printing.
In combination with establishing this workflow, it is important to consider the quality and safety aspects of creating and delivering these devices. Bastawrous et al.[14] discusses the quality controls and development of a quality management system for an in-house printing workflow. The considerations vary based on what the intended use of the device is and the corresponding desired functionality. Determining the intended use is an important first step for determining quality measurements to be taken in the workflow and ensuring that the final product meets the requirements. The workflow below includes possible errors that could happen that need to be addressed by the quality system[14]:
1. Imaging patient anatomy - artifacts, lack of thin-slice datasets;
2. Segmentation and design of device - automatic segmentation errors, incorrect image interpretation, loss of detail in smoothing, inadvertent deletion of structures;
3. 3D printing and post-processing of device - auto resizing of device, poor orientation/placement on printer, printer errors, material irregularities, damage during post-processing, incorrect post-processing resulting in inadequate material properties;
4. Delivery of the device and sterilization - incorrect labeling, material property change after sterilization.
While radiologists are familiar with ensuring the quality of imaging data, other aspects of the workflow are unfamiliar in the hospital setting. Hospitals are familiar with quality systems guidelines as outlined in The Joint Commission developed National Patient Safety Goals (NPSGs); however, many requirements of this workflow are beyond these guidelines and begin to fall under regulatory standards for the manufacturing industry[14]. This includes standards such as ISO 9001, an international standard for QMS, and ISO 13485, an international standard for medical device QMS. As in-house 3D printing grows, increasing the manufacturing activity of hospitals, these regulation standards will also need to be considered.
Benefits of in-house VPSP and PSI
With the emergence of VSPS and PSI as viable options for reconstructive procedures, industrial produced models were the only means for developing and obtaining these materials. They proved to be reliable, high-quality guides and plates that were easily accessible by most providers, thus becoming the gold standard of methodology for these procedures. Currently, some institutions strive to replicate this process within their own system with the goal to replicate the technicality, refinement, and specificity of the commercial models but at a fraction of the cost. Models can be produced for less complex cases, with a significant benefit seen in turn-around time being days, compared to multiple days to weeks seen for commercial options. This technology is in a more primitive phase of development, not widely available, such as commercial options, and requires overhead cost, adequate materials, clinical volume, and annual trainings. The in-house technology is currently not as precise and thus not a viable option for more complex cases. However, with further development, this promising method could provide patients with expedited, high-quality procedures in a more cost-effective format.
The comparison of cost of industry-produced to in-house models was analyzed by Lor et al.[15]. Their study looked at cost savings when using in-house printing models for complex unilateral midface trauma. With the limitation of time when using industry-produced models, their use of in-house models showed a largely reduced price for midfacial reconstruction models when produced by in-house printers[15]. In-house printing costs averaged $236.36 (SD ± 26.17), which was significantly less (P < 0.001) than the Industrial-produced models that ranged from $708.33 to $1995.00 ($1677.82 ± 488.43)[15]. These totals considered Software and disposable feeds, segmentation labor, material costs, print time fees, and production labor.
Sharkh and Makhoul[16] showed the benefit of specific 3D printers and model production with in-house virtual planning techniques for maxillofacial reconstructive procedures. Using both 3DSlicer and Autodesk Meshmixer software, they were able to perform virtual patient specific planning and fabricate cutting guides. They printed a reconstructed jaw model in-house using a commercially available fused deposition modeling-based desk-top 3D-print (Qidi Technology) and used it to prebend the plate. They developed and printed cutting guides using a commercially available resin-based stereolithography apparatus desk-top 3D-printer to sterilize the guides. The Qidi PLA printer costs approximately US $900, while the SLA printer costs approximately US $5000. The average cost of printing the reconstructed model per case was CAD $5.21, and the average cost for printing the cutting guides was CAD $12.80, totaling CAD $18.01 per case[16].
In-house 3D model production with computer aided surgery that utilizes CAD/CAM technology to guide mandibular reconstruction has typically been reported as expensive, requiring investments of time and funds to perform. Dell’Aversana Orabona et al.[17] investigated the reliability and efficiency of homemade cutting guides utilizing a free open-source software for digital planning and 3D layer plastic deposition printer in four cases of patients undergoing mandibular segmental osteotomies and reconstruction with free fibula flap. The initial cost of the 3D printer was approximately 3000 Euro (US $3,509.32) with which they subsequently produced the appropriate models and guides via 6 m of Bioflex medical PLA filament, each at a 3.6 Euro (US $4.21) expense[17]. The authors concluded that using in-house techniques for 3D model and guide production would reduce operative time and cost[17]. A similar cost effectiveness was echoed by
Louvrier et al.[19] notes its importance in cases of midfacial trauma. They examined the turn-around time for industry vs. in-house 3D printed models. They concluded that if not printed in-house, industrially developed implants may take several weeks to print and be delivered, possibly delaying surgery and thus increasing patient cost and hospital space occupancy[19]. This is echoed by Ma et al.[20] as they address this issue in the form of volume at certain medical centers. Medical centers with lower volumes of reconstruction cases are more likely to rely on industry-produced models as the cost of purchasing and maintaining an in-house device may not be feasible[20]. Thus, they argue this could delay the delivery and treatment time, resulting in adverse patient outcomes and increased costs. The specific time concern and treatment delay are described by Lor et al.[15] as they looked at the disparity between patients receiving midface implants constructed in or out of house and their use in acute cases. Their study showed a substantial difference in time for the development of the pieces as, in-house models can be made in as little as 4.65 h, while industrial models take nearly 120 h from order placement to be used[15].
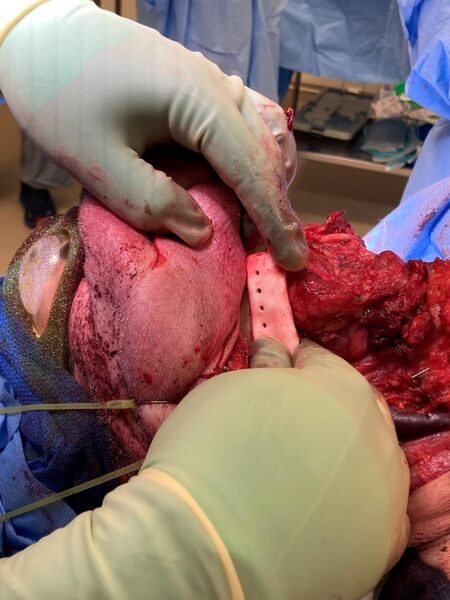
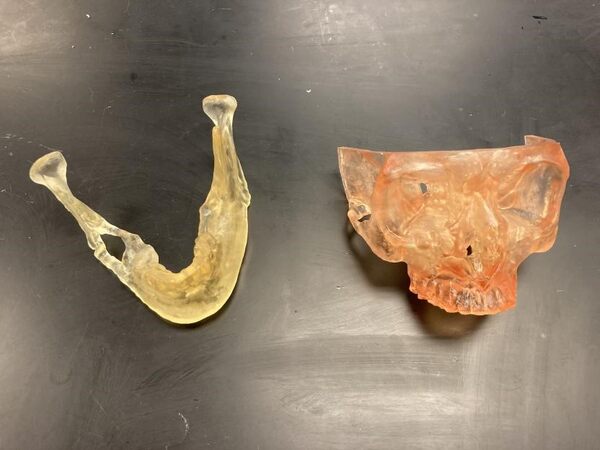
As discussed above, the capabilities of in-house printing in the context of midface reconstruction is an auspicious methodology. Printable items include, but are not limited to, surgical drill guides [Figures 2 and 3], mandible models [Figures 4 and 5], and maxilla models [Figure 5]. The following images are from a point-of-care in-house manufacturing lab and exemplify the feasibility of this process.
Figure 2. In-house 3D-printed Drill Guide[22].
Figure 3. In-house 3D-printed Drill Guide (2)[23].
Figure 4. In-house 3D-printed mandible model with tumor (yellow)[24].
Figure 5. In-house 3D-printed mandible and maxilla models[25].
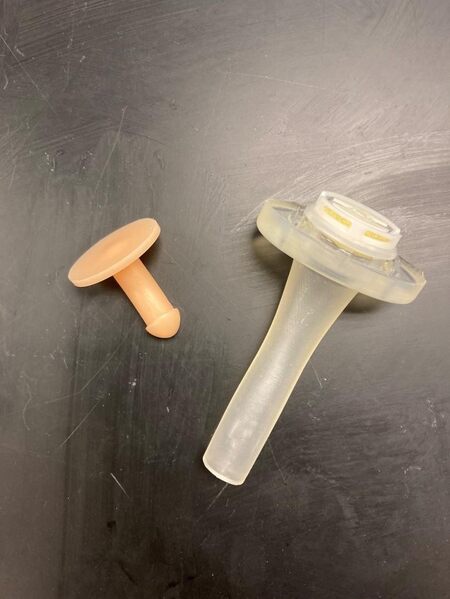
The capability of in-house printing extends far beyond that of VPSP and PSI as models can be produced for a variety of purposes. Those of which are highlighted below include patient care devices [Figure 6], aesthetic design [Figure 7], and models for educational and training purposes [Figure 8].
Figure 6. In-house 3D-printed tracheostomy plug (Left) and laryngectomy tube (Right)[26].
Figure 7. In-house 3D-printed wax nose for prosthetic design[27].
Figure 8. In-house 3D-printed auricular hematoma simulator[28].
DISCUSSION
Current literature and practicing providers suggest a clear time and cost benefit of VPSP and PSI in its use during boney reconstructive surgeries, particularly midfacial reconstructive work. Their utilization allows for more expedient procedures, especially for that of more complex operations. While shorter operation times lead to some cost savings, the benefits are not fully realized until considering a more indirect manner through shorter ischemia times, more accurate reconstructions, and decreased complications. Moving to in-house planning and printing has also led to realized financial gains for reconstruction surgeons. Previous concerns such as the need for preoperative planning time by the surgeon and virtual planning team, as well as the cost of materials have been minimized by current providers who successfully moved to “in-house” development. Providers who utilize in-house planning and printing can produce viable models at minimal cost as well as hold productive planning meetings in a low-key environment to optimize time and cost savings.
DECLARATIONS
Authors’ contributionsAll agree to be responsible for literature review, data collection and analysis, critical editing and composing the manuscript: Lawless M, Swendseid B, von Wildheim N, VanKoevering K, Old M, Seim N
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationFigure 1 Reprinted by permission from: Springer Nature Journal of Materials Research (Daoud GE et al.[13]), 2021. Figures 2-8 in the references section, which are original images and Table 1 in the references section, which is an original table.
CopyrightThe Author(s) 2022.
REFERENCES
1. Ballard DH, Mills P, Duszak R Jr, Weisman JA, Rybicki FJ, Woodard PK. Medical 3D printing cost-savings in orthopedic and maxillofacial surgery: cost analysis of operating room time saved with 3D printed anatomic models and surgical guides. Acad Radiol 2020;27:1103-13.
2. Tack P, Victor J, Gemmel P, Annemans L. 3D-printing techniques in a medical setting: a systematic literature review. Biomed Eng Online 2016;15:115.
3. Zweifel DF, Simon C, Hoarau R, Pasche P, Broome M. Are virtual planning and guided surgery for head and neck reconstruction economically viable? J Oral Maxillofac Surg 2015;73:170-5.
4. Mazzola F, Smithers F, Cheng K, et al. Time and cost-analysis of virtual surgical planning for head and neck reconstruction: a matched pair analysis. Oral Oncol 2020;100:104491.
5. Crawley MB, Sweeny L, Ravipati P, et al. Factors associated with free flap failures in head and neck reconstruction. Otolaryngol Head Neck Surg 2019;161:598-604.
6. Eskander A, Kang S, Tweel B, et al. Predictors of complications in patients receiving head and neck free flap reconstructive procedures. Otolaryngol Head Neck Surg 2018;158:839-47.
7. May MM, Howe BM, O’Byrne TJ, et al. Short and long-term outcomes of three-dimensional printed surgical guides and virtual surgical planning versus conventional methods for fibula free flap reconstruction of the mandible: decreased nonunion and complication rates. Head Neck 2021;43:2342-52.
8. Swendseid BP, Roden DF, Vimawala S, et al. Virtual surgical planning in subscapular system free flap reconstruction of midface defects. Oral Oncol 2020;101:104508.
9. Swendseid B, Kumar A, Sweeny L, et al. Natural history and consequences of nonunion in mandibular and maxillary free flaps. Otolaryngol Head Neck Surg 2020;163:956-62.
10. Navarro Cuéllar C, Tousidonis Rial M, Antúnez-Conde R, et al. Virtual surgical planning, stereolitographic models and CAD/CAM titanium mesh for three-dimensional reconstruction of fibula flap with iliac crest graft and dental implants. J Clin Med 2021;10:1922.
11. Seier T, Hingsammer L, Schumann P, Gander T, Rücker M, Lanzer M. Virtual planning, simultaneous dental implantation and CAD/CAM plate fixation: a paradigm change in maxillofacial reconstruction. Int J Oral Maxillofac Surg 2020;49:854-61.
12. Allen RJ Jr, Shenaq DS, Rosen EB, et al. Immediate dental implantation in oncologic jaw reconstruction: workflow optimization to decrease time to full dental rehabilitation. Plast Reconstr Surg Glob Open 2019;7:e2100.
13. Daoud GE, Pezzutti DL, Dolatowski CJ, et al. Establishing a point-of-care additive manufacturing workflow for clinical use. J Mater Res 2021;36:3761-80.
14. Bastawrous S, Wu L, Strzelecki B, et al. Establishing quality and safety in hospital-based 3D printing programs: patient-first approach. Radiographics 2021;41:1208-29.
15. Lor LS, Massary DA, Chung SA, Brown PJ, Runyan CM. Cost analysis for in-house versus industry-printed skull models for acute midfacial fractures. Plast Reconstr Surg Glob Open 2020;8:e2831.
16. Sharkh H, Makhoul N. In-house surgeon-led virtual surgical planning for maxillofacial reconstruction. J Oral Maxillofac Surg 2020;78:651-60.
17. Dell’Aversana Orabona G, Abbate V, Maglitto F, et al. Low-cost, self-made CAD/CAM-guiding system for mandibular reconstruction. Surg Oncol 2018;27:200-7.
18. Moe J, Foss J, Herster R, et al. An in-house computer-aided design and computer-aided manufacturing workflow for maxillofacial free flap reconstruction is associated with a low cost and high accuracy. J Oral Maxillofac Surg 2021;79:227-36.
19. Louvrier A, Marty P, Barrabé A, et al. How useful is 3D printing in maxillofacial surgery? J Stomatol Oral Maxillofac Surg 2017;118:206-12.
20. Ma H, Shujaat S, Van Dessel J, et al. Adherence to computer-assisted surgical planning in 136 maxillofacial reconstructions. Front Oncol 2021;11:713606.
21. Lawless M. Overview of VPSP and PSI benefits (top). Comparative benefits of industrial vs. in-house VPSP and PSI (bottom). Department of Otolaryngology - Head and Neck Surgery, Columbus. OH USA: The Ohio State University Comprehensive Cancer Center; 2022 February 2. Unpublished.
22. Herster R. In-house 3D-printed Drill Guide [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 275 × 175. Unpublished.
23. Herster R. In-house 3D-printed Drill Guide (2) [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 270 × 251. Unpublished.
24. Herster R. In-house 3D-printed Mandible Model with Tumor (yellow) [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 282 × 242. Unpublished.
25. Herster R. In-House 3D-printed Mandible and Maxilla Models [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 272 × 134. Unpublished.
26. Herster R. In-house 3D-printed Tracheostomy plug (Left) and Laryngectomy Tube (Right) [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 250 × 207. Unpublished.
27. Herster R. In-house 3D-printed Wax nose for prosthetic design [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 272 × 256. Unpublished.
28. Herster R. In-house 3D-printed Auricular Hematoma simulator [digital]. Columbus (OH): Department of Otolaryngology - Head and Neck Surgery. Columbus, OH USA: The Ohio State University Comprehensive Cancer Center; 2022, February 2. 1 Image: color, 215 × 271. Unpublished.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Lawless M, Swendseid B, von Windheim N, VanKoevering K, Seim N, Old M. Review of cost and surgical time implications using virtual patient specific planning and patient specific implants in midface reconstruction. Plast Aesthet Res 2022;9:26. http://dx.doi.org/10.20517/2347-9264.2021.108
AMA Style
Lawless M, Swendseid B, von Windheim N, VanKoevering K, Seim N, Old M. Review of cost and surgical time implications using virtual patient specific planning and patient specific implants in midface reconstruction. Plastic and Aesthetic Research. 2022; 9: 26. http://dx.doi.org/10.20517/2347-9264.2021.108
Chicago/Turabian Style
Lawless, Michael, Brian Swendseid, Natalia von Windheim, Kyle VanKoevering, Nolan Seim, Matthew Old. 2022. "Review of cost and surgical time implications using virtual patient specific planning and patient specific implants in midface reconstruction" Plastic and Aesthetic Research. 9: 26. http://dx.doi.org/10.20517/2347-9264.2021.108
ACS Style
Lawless, M.; Swendseid B.; von Windheim N.; VanKoevering K.; Seim N.; Old M. Review of cost and surgical time implications using virtual patient specific planning and patient specific implants in midface reconstruction. Plast. Aesthet. Res. 2022, 9, 26. http://dx.doi.org/10.20517/2347-9264.2021.108
About This Article
Copyright
Data & Comments
Data
 Cite This Article 16 clicks
Cite This Article 16 clicks














Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.