Flaps for bulbar urethral ischemic necrosis in pelvic fracture urethral injury
Abstract
Bulbar urethral ischemic necrosis (BUIN) is an iatrogenic entity resulting from repeated attempts at performing anastomotic urethroplasty for pelvic fracture urethral injuries. Etiologically speaking, BUIN is related to a compromised blood supply of the bulbar urethra, which normally relies on anterograde supply from bulbar arteries and retrograde supply from recurrent branches of dorsal penile arteries, through the glans. At each transection of the bulbar urethra, both the anterograde and retrograde supplies are compromised, increasing the risk of BUIN. Even though this term is widely used among reconstructive urologists, BUIN is orphan of an accepted scientific definition. We aim to report our personal perspective on BUIN, to identify factors associated with its occurrence, and to describe the management options in these patients.
Keywords
INTRODUCTION
Pelvic fracture urethral injury (PFUI) occurs in 10% of those who suffer from pelvic fractures. The most common cause of injury is road traffic accidents[1]. While in the Western world PFUI is uncommon and its incidence is decreasing due to higher safety standards on the roads and at work, in developing countries, due to lack of safety rules and uncontrolled environment such as obligation to wear the helmet on road or at work, fasten seat belts, etc., PFUIs are more common and are usually related to work accidents, traffic accidents, falls from high places, or natural catastrophes[2]. The site of urethral injury is the bulbo-membranous junction in the majority of patients. Rarely, the urethral injury is localized to the prostato-membranous junction, intra prostatic, and/or at the bladder neck. Step-wise anastomotic urethroplasty, as described by Webster in 1986, remains the gold standard for treatment of PFUI[3]. The goal of the approach is to reach a tension free anastomosis between the two ends of healthy urethra after transecting the fibrotic segment. The progressive perineal approach is the preferred surgical method. In complex patients with long fibrotic segments or double urethral injuries, the transabdominal approach is also utilized to allow a more extensive pubectomy and expose the prostatic urethra and the bladder neck. Bulbar urethral ischemic necrosis (BUIN) is an iatrogenic entity which results from previous failed anastomotic repairs in patients with PFUI. The term BUIN was used for the first time in 1986 by Turner-Warwick[4]. Historically, BUIN was defined as “long gap” or “unsalvageable” bulbar urethra after previous failed repairs[5,6]. To date, an accepted and standardized definition of BUIN is lacking.
Anatomical considerations
The male urethra is generally classified into two portions: (1) the posterior urethra that extends from the bladder neck to the external urinary sphincter and comprises the prostatic urethra and the membranous urethra; and (2) the anterior urethra, which comprises the pendulous urethra (or penile) and the bulbar urethra, beginning at the peno-scrotal junction and ending at the bulbo-membranous junction.
The bulbar urethra can be divided into distal, mid, and proximal portions. The proximal and mid portions are unique as the spongious tissue is more developed in this region. These segments are also covered by the bulbospongiosus muscle that plays an important role in micturition and ejaculation. The proximal two-thirds of the muscle is circumferential around the urethra. The distal one-third of bulbospongio-cavernous muscle surrounds the corpus cavernous at the base of the penis[7].
Antegrade blood supply to the urethra is derived from the bulbar arteries and perforating vessels of the cavernosal arteries. Retrograde blood supply is derived from the dorsal penile artery. The urethra is also supplied laterally by the circumflex vessels. All these branches are derived from the internal pudendal arteries, which come from the anterior division of the internal iliac arteries.
“Our” definition of BUIN
BUIN is defined as a compromised or complete absence of a segment of bulbar urethra. It can appear as a long narrowing or semi-obliteration of a segment of the bulbar urethra (bulbar urethral fibrosis), or the absence of a segment of bulbar urethral at the retrograde and/or micturition urethrograms (RGU, MCU). The etiology is always vascular.
Pathophysiology of BUIN
On routine retrograde urethrogram, BUIN usually appears as a long bulbar urethra stenosis or as a complete loss of bulbar urethra. For both cases, the etiology of the pathologic condition is vascular ischemia.
In 1986, Turner-Warwick described the causes for this vascular ischemia and spongionecrosis.
1. Over mobilization of distal urethra during anastomotic urethroplasty.
2. Presence of hypospadias and associated lack of retrograde blood supply.
3. Extensive spongiofibrosis resulting from multiple previous surgeries.
How can this happen? To perform the anastomotic urethroplasty, a dissection and mobilization of the distal urethra is required. To mobilize the distal segment of bulbar urethra, the lateral circumflex vessels must be sacrificed, as well as most of the perforating vessels from the cavernosa arteries. When the bulbar urethra is transected, at the site of injury, the bulbourethral arteries also are sacrificed to reach the proximal urethral stump and the perineal diaphragm. After transection and mobilization, the distal segment of the urethra depends on the retrograde blood supply from the dorsal penile vessels. Thus, patients presenting with inadequate retrograde blood supply, due to compromised dorsal penile vessels or hypospadias, might develop BUIN.
In patients who do not present congenital vascular malformations of the blood supply of the genitals (e.g., hypospadias), there are two main reasons for an inadequate retrograde blood supply: (1) It is usually a result of surgery due to the iatrogenic injury of one or both the dorsal penile artery at the time of inferior pubectomy. Indeed, when surgeons approach the pubic bones after crura separation, the excessive use of diathermy, especially on transverse and circumferential fashion, as well as the missed step of periosteum elevation, may increase the risk of vascular injury and subsequently of BUIN[8]. (2) Very rarely, as a result of primary trauma, there can be bilateral pubic bone fractures, causing damage to either the internal pudendal or the dorsal vessels on both sides. Such patients may develop BUIN even without iatrogenic injury. In our large experience of over 1700 cases of PFUI treated with anastomotic urethroplasty, we reported trauma-related vascular deficiency in only three patients, who subsequently developed BUIN. To predict this possibility, we recommend assessing preoperatively the blood supply of the urethra with the color doppler of the cavernosa and dorsal penile arteries. This assessment can guide surgeons during their dissection of the posterior urethral stump, preserving the side where the dorsal penile artery showed a better flow and deepening the dissection on the side with decreased flow.
Awareness of this pathophysiology helps prevent BUIN and its sequelae.
Incidence of BUIN
The exact incidence of BUIN is unknown. Between 1995 and 2021, we treated and managed more than 1700 patients with PFUI. Our success rate at catheter removal for primary anastomosis is 85.4% and for redo cases is 79.1%. Of all patients treated for PFUI, 177 (approximately 9%) had BUIN. The majority of patients with BUIN reported two or more prior failed attempts of PFUI repair[9]. Unfortunately, the incidence of BUIN is rising in recent years, as witnessed by a rising number of flap reconstructions (e.g., pedicle preputial tube). Indeed, the last treatment resource for BUIN is to replace the missing urethral segment with a vascularized flap, which can be tubularized and allows urethral continuity. Table 1 to support this statement show that the number of flaps raised from 3-4 per year during 2010-2015 to more than 15 per year between 2019 and 2021, with 29 flaps in the last 12 months in our units.
Urethroplasties performed for bulbar urethral necrosis
| Procedure | Number | Success rate |
| Pedicled preputial tube and Q flap | 132 | 86.4% |
| Oral mucosal flap | 10 | 58.6% |
| Pedicled prepuce as onlay augmentation with dorsal BMG | 16 | 89.1% |
| Scrotal drop back | 7 | 33.3% |
| Enterourethroplasty | 5 | 100% |
| Forearm flap with microvascular anastomosis | 3 | 100% |
| Pedicled anterolateral thigh flap | 2 | 50% |
| Dorsal BMG with ventral BMG on gracilis muscle transposed to perineum | 2 | 100% |
| Total | 177 | 76.7% |
Evaluation of patients with BUIN
As previously described, the etiology of BUIN is an inadequate blood supply, especially the retrograde blood supply, from the dorsal penile arteries. This inadequate blood supply is more frequently due to an iatrogenic injury of these vessels while performing the anastomotic urethroplasty; more rarely, the deficiency is related to the primary trauma or congenital. During the preoperative work-flow of patients with PFUI, we recommend assessing the presence of erectile dysfunction using standardized questionnaires such as IIEF and the examination of the vascular supply with the penile doppler ultrasound[10,11]. These tests allow a comprehensive evaluation of the genital organ and guide surgeons during the dissection of the posterior urethra reducing the risk of iatrogenic injury of the vessels. The parameters which are assessed during the penile doppler are the peak of systolic velocity (PSV), end diastolic velocity, and resistance index for both cavernosal and dorsal penile arteries before and after administration of intracavernosal Prostaglandin E1. Patients with PSV greater than 35 cm/s were considered to have a normal arterial response, while less than 25 cm/s is associated with severe arterial deficiency. In patients with severe arterial deficiency, we recommend penile revascularization procedure, using microvascular anastomosis between inferior epigastric artery and dorsal penile vessels prior to PFUI repair to avoid BUIN[12].
Diagnosis of BUIN
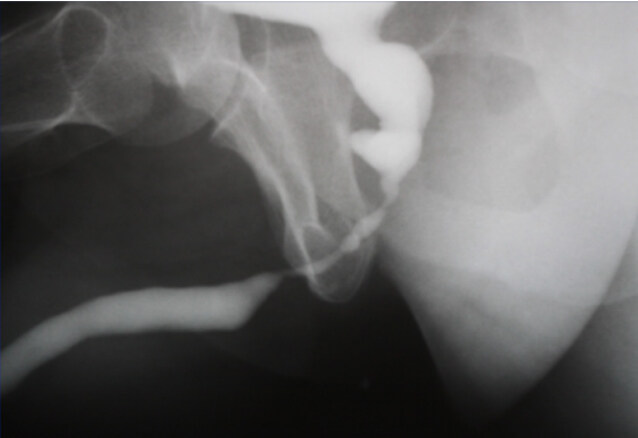
The best way to diagnose the BUIN is performing a retrograde and/or micturition urethrogram (RGU and MCU, respectively) [Figure 1]. These tests would show a long stenotic segment of bulbar urethra (bulbar urethral fibrosis) or a complete gap of bulbar urethra [Figure 2]. MRI[13] and 3D printing models[14] have also proved to be useful for the diagnosis and assessment of patients with PFUI and suspected BUIN.
Figure 1. RGU and MCUG suggestive of bulbar urethral stenosis. RGU: Retrograde urethrogram; MCUG: micturating cystourethrogram.
Figure 2. RGU and MCUG depicting bulbar urethral ischemic necrosis (BUIN). RGU: Retrograde urethrogram; MCUG: micturating cystourethrogram.
In established cases of bulbar urethral ischemia, there are limited options for reconstruction. These patients require care in high volume referral centers to achieve valid functional outcomes and reduce incidence of complications[15]. Initial evaluation includes the identification of the type of injury, the extent of injury, prior number and type of repairs, and preoperative assessment of antegrade and retrograde blood flow to the urethra. All these parameters are of paramount importance.
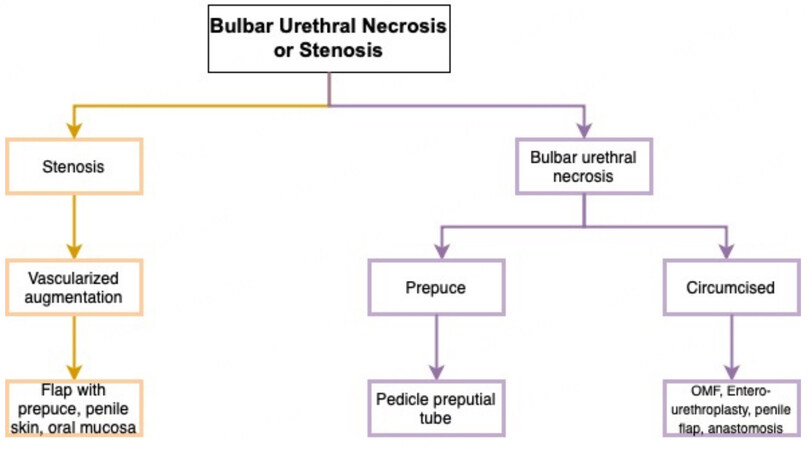
Herein, we present our management algorithm (Figure 3 for BUIN[9]).
Treatment of BUIN
In patients with BUIN presenting with a long stenotic segment, urethral augmentation using a buccal graft, or a penile skin flap is recommended. Presence of lumen and the amount of spongiofibrosis determines the need for a vascularized flap as opposed to a buccal mucosal graft.
The following surgical approaches are available.
1. Pedicle preputial flap/tube:
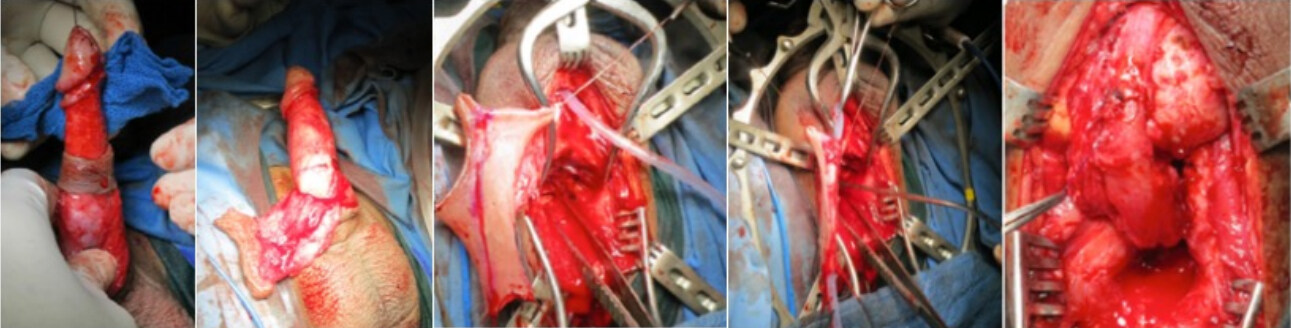
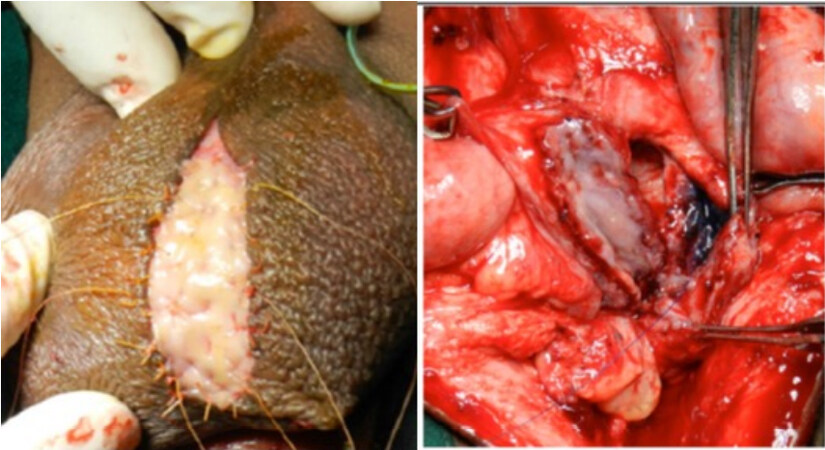
In cases of BUIN, it is important to note preoperatively whether the patient is circumcised or not. In non-circumcised patients, our preferred approach is to use the prepuce to harvest a pedicle preputial tube for repair. Two parallel circumcision incisions are made. The outer circumcision is deep to the skin and superficial to dartos fascia. This dissection is carried forward until the base of the penis. The inner circumcision is performed deep to dartos and superficial to the Bucks fascia. The entire prepuce is then mobilized based on its dartos pedicle. The preputial ring flap is opened ventrally until the penoscrotal junction. Then, the flap is transposed to the perineum through an opening at the basis of the penis. The flap is tubularized to bridge the long gap between the anterior and posterior urethra. The outer prepuce is sacrificed [Figure 4]. This technique can be done using distal penile skin when patient is circumcised[16].
2.“Q” Flap:
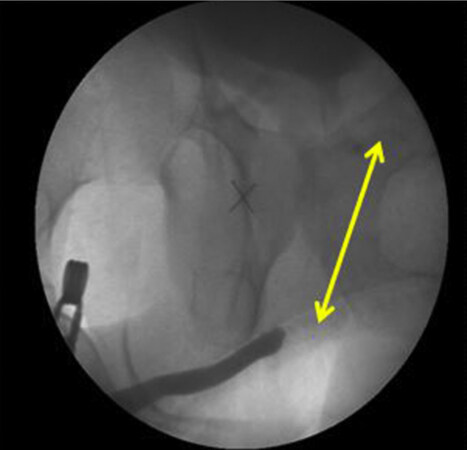
Occasionally, the gap in the bulbar urethra is much longer than anticipated [Figure 5A]. The length of the penis is usually 10-12 cm during erection. Thus, the length of a circumferential flap of prepuce or distal penile skin can reach 10-12 cm at best. In patients with longer gaps, we harvest a “Q”-shaped flap, as described by Morey[17] [Figure 5B]. The length of such flap can be up to 20 cm.
Figure 5. (A) RGU showing long obliterative segment; and (B) “Q-fasciocutaneous flap”. RGU: Retrograde urethrogram.
BUIN in circumcised patients, or for those patients with a small penis with insufficient penile skin, represent a challenging scenario. The options to repair the urethral gap include the following.
3. Oral mucosal flap:
An appropriately sized buccal mucosal graft is harvested[18] and placed and quilted over the dartos of the scrotum. A week later, a flap made from the graft and its underlying dartos can be mobilized with its pedicle and transposed to the perineum serving as an onlay flap or tube for reconstruction [Figure 6].
4. Scrotal drop back (Turner-Warwick):
The scrotal skin is used to bridge the gap between the two ends of the urethra. A midline perineal vertical incision is made. Dissection of the posterior urethra is done. The distal urethra is identified. The scrotal skin is then folded on itself, and an opening is made on the lowest part which is anastomosed to the posterior urethra. This helps the scrotal skin, which is mobile, to form a urethral plate. Six months later, the scrotal skin is tabularized, and urethral continuity is achieved. It has been described in the literature as a surgical procedure for primary perineal urethrostomy but is uncommonly utilized. We have used this technique in 7 patients with complex PFUI with BUIN and reported a success rate of 33.33%.
5. Entero-urethroplasty:
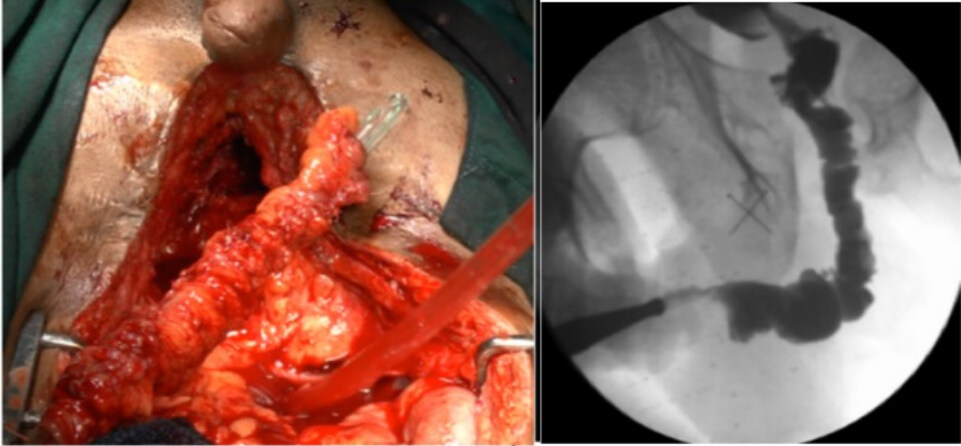
Since 1995, we have performed five entero-urethroplasties following the technique described by Mundy and Andrich[5]. Patient selection for these procedures is of paramount importance. The key is to evaluate which patients could benefit from urethral substitution using a bowel segment. Secondly, it is very important to find the best suited bowel segment for the reconstruction. Usually, we choose the segment of bowel based on the mobility of its pedicle and its proximity to the prostate. Most frequently, the choice falls on the sigmoid. An urethroplasty usually starts with a perineal incision. The healthy distal end of the anterior urethra is identified. Crural separation and inferior pubectomy are performed according to the need to reduce the gap between the distal end and the bowel segment. Frequently, we also approach the area with a suprapubic incision, which allows reaching the extraperitoneal space of Retzius, excising additional wedges of the pubic bones, and removing more scar tissue. The posterior urethra is incised on a bougie passed from the SPC tract though the bladder and the bladder neck. Once the posterior urethra is circumferentially mobilized, we assess the gap length between the two urethral ends. The sigmoid colon is then disconnected from the intestine and mobilized on its mesentery. The more distal portion of the sigmoid is usually sacrificed due to its poor blood supply, while the upper part is swung down on its pedicle. The sigmoid is tailored on a 26-30 Fr nelaton catheter. Then, the enteric segment is transposed to the perineum, and the two anastomoses between proximal sigmoid-end and bulbo membranous junction as well as between the distal sigmoid-end and the anterior urethra are performed. Finally, the omentum is mobilized and wrapped around the proximal anastomosis to offer a vascular support and to prevent from leak and fistulae [Figure 7]. The catheter is removed after six weeks if a peri-catheter urethrogram does not show leakage.
6. Use of buccal mucosa graft[18] (BMG) dorsally and flap ventrally:
This can be performed provided that inferior pubectomy and crural separation are not required. The corpora cavernosa is required to quilt the oral graft dorsally. The penile skin flap is then apposed as ventral onlay to reconstruct the neourethra.
7. Pedicled anterolateral thigh flap for reconstruction for urethral defects[19,20]:
Various vascularized tissues can be used to reconstruct the urethra. This can be a fasciocutaneous flap or a fascia lata flap. Despite its common use for many soft tissue defects in clinical series, there are no reports on the reliability of urethral defects. Ozkan et al.[19] concluded that the flaps could be considered an excellent and ideal alternative to the most used conventional methods for most urethral defects. However, such flaps are technically challenging and should not be used routinely.
8. Forearm flap with microvascular anastomosis to inferior epigastric artery[21]:
This is a prelaminated vascularized flap that can be used as a tube. The forearm skin is incised, and a BMG is inserted onto the fascia. The defect on the forearm skin is closed. The BMG is covered by the overlying skin. The graft takes up on the fascia. Three weeks later, the same incision is made, and the BMG along with the underlying tissues is raised as a flap based on the artery and vein of the forearm. The vessels of the flap are then anastomosed to the vessels of the abdominal wall.
9. Dorsal BMG with ventral BMG on gracilis muscle[22]:
The dorsal wall of the urethra is the BMG quilted to the corpora. The ventral wall is created by mobilizing a gracilis flap and harvesting another BMG that is quilted to the muscle and then transposed into the perineum.
In our series, we managed 177 cases of BUIN. Table 1 outlines our usage of the above techniques and results.
In our experience, the best results are achieved using a vascularized flap with or without buccal mucosal graft.
CONCLUSIONS
BUIN is a catastrophic iatrogenic event that occurs after repeated urethral transection. The best cure is prevention, as surgical reconstruction options are limited and are associated with complications and the risk of failure. BUIN must be managed in high-volume centers by experts to achieve satisfactory long-term outcomes. These patients represent a challenging cohort requiring a versatile approach and a broad knowledge of different reconstructive techniques[23]. Preservation of the dorsal penile arteries should be prioritized and can be achieved by performing periosteal elevation[24] and inferior pubectomy, if necessary. Multi-stage surgery is contraindicated in these patients. Instead, single-stage pedicle flaps from prepuce or distal penile skin are recommended.
DECLARATIONS
Author’s contributionsProtocol/project development, Manuscript writing/editing: Joshi PM, Bandini M, Yepes C
Data collection or management: Bafna S, Bhadranavar S, Sharma V, Cirulli GO
Manuscript writing/editing: Kulkarni SB
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateInstitutional review board approved the study with protocol number KESI/22007.
Consent for publicationInformed consent for performing surgeries, invasive procedures and use of pictures/videos for academic and scientific purpuses is taken.
Copyright© The Author(s) 2022.
REFERENCES
1. Demetriades D, Karaiskakis M, Toutouzas K, Alo K, Velmahos G, Chan L. Pelvic fractures: epidemiology and predictors of associated abdominal injuries and outcomes. J Am Coll Surg 2002;195:1-10.
2. Joshi PM, Kulkarni SB. Management of pelvic fracture urethral injuries in the developing world. World J Urol 2020;38:3027-34.
3. Webster GD, Goldwasser B. Perineal transpubic repair: a technique for treating post-traumatic prostatomembranous urethral strictures. J Urol 1986;135:278-9.
5. Mundy AR, Andrich DE. Entero-urethroplasty for the salvage of bulbo-membranous stricture disease or trauma. BJU Int 2010;105:1716-20.
6. Barbagli G. History and evolution of transpubic urethroplasty: a lesson for young urologists in training. Eur Urol 2007;52:1290-2.
7. Joshi P, Kaya C, Kulkarni S. Approach to bulbar urethral strictures: which technique and when? Turk J Urol 2016;42:53-9.
8. Kulkarni SB, Surana S, Desai DJ, et al. Management of complex and redo cases of pelvic fracture urethral injuries. Asian J Urol 2018;5:107-17.
9. Kulkarni SB, Joshi PM, Hunter C, Surana S, Shahrour W, Alhajeri F. Complex posterior urethral injury. Arab J Urol 2015;13:43-52.
10. Guan Y, Wendong S, Zhao S, et al. The vascular and neurogenic factors associated with erectile dysfunction in patients after pelvic fractures. Int Braz J Urol 2015;41:959-66.
11. Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997;49:822-30.
12. Zuckerman JM, McCammon KA, Tisdale BE, et al. Outcome of penile revascularization for arteriogenic erectile dysfunction after pelvic fracture urethral injuries. Urology 2012;80:1369-73.
13. Joshi PM, Desai DJ, Shah D, Joshi DP, Kulkarni SB. Magnetic resonance imaging procedure for pelvic fracture urethral injuries and recto urethral fistulas: a simplified protocol. Turk J Urol 2021;47:35-42.
14. Joshi PM, Kulkarni SB. 3D printing of pelvic fracture urethral injuries-fusion of technology and urethroplasty. Turk J Urol 2020;46:76-9.
15. Bandini M, Barbagli G, Leni R, et al. Assessing in-hospital morbidity after urethroplasty using the European Association of Urology Quality Criteria for standardized reporting. World J Urol 2021;39:3921-30.
16. Kulkarni S, Joshi P, Surana S, Kulkarni J, Joglekar O, Alkandari M. Pedicled preputial tube urethroplasty for bulbar urethral necrosis after failed anastomotic urethroplasty for pelvic fracture urethral distraction defects. Eur Urol Suppl 2018;17:e1937.
17. Morey AF, Tran LK, Zinman LM. Q-flap reconstruction of panurethral strictures. BJU Int 2000;86:1039-42.
18. Barbagli G, Bandini M, Balò S, et al. Patient-reported outcomes for typical single cheek harvesting vs atypical lingual, labial or bilateral cheeks harvesting: a single-center analysis of more than 800 patients. World J Urol 2021;39:2089-97.
19. Ozkan O, Ozkan O. The prefabricated pedicled anterolateral thigh flap for reconstruction of a full-thickness defect of the urethra. J Plast Reconstr Aesthet Surg 2009;62:380-4.
20. Harris TGW, Manyevitch R, Wu WJ, et al. Pedicled anterolateral thigh and radial forearm free flap phalloplasty for penile reconstruction in patients with bladder exstrophy. J Urol 2021;205:880-7.
21. Chauhan A, Sham E, Chee J. Microsurgical urethroplasty for complex bulbar urethral strictures using the radial forearm free flap prelaminated with buccal mucosa. J Reconstr Microsurg 2016;32:378-85.
22. Nikolavsky D. Prelaminated gracilis flap with buccal mucosal graft for salvage of devastated urethra. Case Rep Urol 2015;2015:490518.
23. Barbagli G, Bandini M, Balò S, Sansalone S, Butnaru D, Lazzeri M. Surgical treatment of bulbar urethral strictures: tips and tricks. Int Braz J Urol 2020;46:511-8.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Joshi PM, Bandini M, Yepes C, Bafna S, Bhadranavar S, Sharma V, Cirulli GO, Kulkarni SB. Flaps for bulbar urethral ischemic necrosis in pelvic fracture urethral injury. Plast Aesthet Res 2022;9:22. http://dx.doi.org/10.20517/2347-9264.2021.98
AMA Style
Joshi PM, Bandini M, Yepes C, Bafna S, Bhadranavar S, Sharma V, Cirulli GO, Kulkarni SB. Flaps for bulbar urethral ischemic necrosis in pelvic fracture urethral injury. Plastic and Aesthetic Research. 2022; 9: 22. http://dx.doi.org/10.20517/2347-9264.2021.98
Chicago/Turabian Style
Joshi, Pankaj M., Marco Bandini, Christian Yepes, Sandeep Bafna, Shreyas Bhadranavar, Vipin Sharma, Giuseppe Ottone Cirulli, Sanjay B. Kulkarni. 2022. "Flaps for bulbar urethral ischemic necrosis in pelvic fracture urethral injury" Plastic and Aesthetic Research. 9: 22. http://dx.doi.org/10.20517/2347-9264.2021.98
ACS Style
Joshi, PM.; Bandini M.; Yepes C.; Bafna S.; Bhadranavar S.; Sharma V.; Cirulli GO.; Kulkarni SB. Flaps for bulbar urethral ischemic necrosis in pelvic fracture urethral injury. Plast. Aesthet. Res. 2022, 9, 22. http://dx.doi.org/10.20517/2347-9264.2021.98
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 3 clicks
Cite This Article 3 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.