A personalized nutrition plan based on genetic profile improves outcomes of facial regeneration with Platelet-Rich Fibrin liquid matrices
Abstract
Aim: The importance of nutrition in the prevention of skin aging has been shown by large observational studies. However, there are no studies assessing dietary changes as adjunct procedures to aesthetic interventions. The objective of this study was to assess whether a personalized nutritional plan conveys additional benefits to platelet-rich fibrin (PRF) facial regeneration.
Methods: Forty-seven healthy women (mean age 52.5 years old, SD = 7.7) were offered minimally invasive facial regeneration with the use of PRF liquid matrices, as well as a personalized nutritional plan. The nutritional plan was informed by a nutrigenetic test based on 128 polymorphisms. Horizontal forehead lines, zygomatic wrinkles or mid-cheek furrows, nasolabial folds, perioral expression wrinkles, and marionette line were assessed separately with the use of the Facial Wrinkles Assessment Scale (FWAS).
Results: The total FWAS score change was statistically significantly better in women who reported an at least partial adaptation of nutritional recommendations for at least three months (Z = 2.4, P = 0.008).
Conclusion: Personalized nutritional recommendations based on individual needs as well as generally accepted dietary guidelines can improve treatment outcomes of minimally invasive facial skin aesthetics interventions.
Keywords
INTRODUCTION
Over the last decade, the use of autologous blood concentrates, such as platelet‐rich plasma and platelet‐rich fibrin (PRF), has gained importance in aesthetic medicine for dermal stimulation, augmentation, and rejuvenation[1-3]. Platelet‐rich fibrin liquid matrices based on the low-speed centrifugation concept have shown effectiveness in facial regeneration[4,5] and are being increasingly applied in facial aesthetics either alone or in combination with other techniques.
At the same time, studies of cells in the laboratory, animal models, and human trials also support roles for a variety of nutrients in preventing skin aging[6]. Large observational studies have suggested associations of vitamin C, linoleic acid, and green and yellow vegetables with younger-looking skin[7,8], indicating the need for more holistic approaches that incorporate nutritional advice into aesthetic treatments.
Personalized nutrition plans informed by knowledge of an individual’s DNA are increasingly being utilized for health as well as general well-being purposes. There are numerous studies, particularly in the fields of preventive medicine and cardiovascular diseases, showing that the specification of general dietary recommendation by the inclusion of genetic variants can have a significant impact on health parameters. At the same time, there are thousands of publications based on large cohort or other retrospective studies indicating that the exact amounts of the various nutrients can be specified, among others, by genotype results. Adherence to a nutritional plan that conforms to standard dietary guidelines as well as considers the various individual needs may improve healthy skin parameters. In this study, we sought to investigate whether a personalized nutrition plan conveys additional benefits to a minimally invasive technique for facial regeneration.
METHODS
In total, 47 otherwise healthy women offered PRF facial regeneration based on the low-speed centrifugation concept were included in the study. An ethical approval was granted (acknowledgments No. 1/26/09/2016), and the patients signed an informed consent prior to their treatment. All patients were treated by the same experienced physician (Nacopoulos C) using a standard protocol described in more detail in previous publications[4,5]. Briefly, four sessions of PRF treatments were offered with 2 to 3-week intervals. In each session, 60 mL of venous blood was collected in 10 mL PRF tubes (Orange tubes, Process for PRF, Nice, France) and centrifuged according to the following protocols: 40 mL (four tubes) for 5 min at 1300 rpm (208 g), which results in 10-11 mL of PRF, and 20 mL (two tubes) for 3 min at 700 rpm (60 g), which results in 2 mL of PRF.
A preprogramed centrifuge with a radius of 110 mm (Process for PRF, Nice France) was used to produce a total of 12-13 mL PRF of reduced density. PRF liquid matrices produced by the two abovementioned centrifugation protocols were mixed and a full-face injection technique was applied as described previously[5].
Α personalized nutritional plan was offered by a specialized nutritionist during the treatment. A nutrigenetic test was utilized to aid personalized nutritional recommendations. In more detail, prior to the first PRF session, 5 mL of additional vein blood was collected for genomic DNA analysis using a Pure Link Genomic DNA Mini Kit Assay (Life Technologies), according to the manufacturer’s instructions.
The nutrigenetic test offered to the health professional personalized recommendations based on the genotyping of 128 single nucleotide polymorphisms. Among others, this test examines polymorphisms related to: macronutrient daily intake, e.g., GIPR SNP rs2287019[9], IRS1 SNP rs294364[10], APOA5 SNP rs964184[11], PCSK7 SNP rs236918[12], TCF7L2 SNP rs7903146, and TFAP2B SNP rs987237[13]; fatty acid needs, e.g., APOE SNPs rs429358 and rs7412[14], CETP SNP rs708272[15], and PPARγ2 SNP rs1801282[16]; aging, e.g., FTO SNP rs9939609[17]; supplement response and special supplemental needs, e.g., CYP2R1 SNP rs10741657[18], VDR SNP rs7968585[19,20], and MTHFR SNPs rs1801133 and rs1801131[21]; special antioxidant needs, e.g., COMT SNP rs4680; oxidative stress, e.g., MTHFR SNP rs1801133[22]; micronutrient responses, e.g., GPX1 SNP rs1050450[23]; inflammation response, e.g., IL-6 SNP rs1800795[24], FTO SNP rs9939609, MC4R SNP rs17782313, and TMEM18 SNP rs6548238[25]; choline needs, e.g., PEMT SNP rs12325817[26,27]; susceptibility to injury, e.g., ACTN3 SNP rs1815739[28-30] and WNT16 SNPs rs2908004 and rs2707466[31,32]; and telomere length, e.g., SNPs rs2736100, rs7675998, rs9420907, rs8105767, rs755017, rs11125529, rs894160, and rs4293393[33,34]. Examples of recommendations based on the aforementioned SNPs are provided in Table 1. Of note is that a personalized approach should be focused on the needs and expectations of the patients, which means that recommendations cannot be universally applied each time a specific SNP is found[35-37].
Examples of single nucleotide polymorphisms and related recommendations
| Pathology or health issue | SNP | Genotype-related health effect | Recommendation |
| Folate deficiency | MTHFR rs1801133 | Increased susceptibility to depression | Increase folate intake |
| Hypertension | MTHFR rs1801133 | Elevated blood pressure | Supplementation with dietary riboflavin |
| Prediabetes, hyperglycemia, and risk of developing T2DM | MTNR1B rs10830963 | Reduced rate of insulin release | Morning melatonin supplementation |
| Difficulty in weight loss/control | COMT rs4680 | Reduction in COMT enzymatic activity | Increase catechin intake to increase energy expenditure |
| Cognitive impairment, dementia, and AD | APOE (rs7412, rs429358) risk haplotype e4e4 | Inefficient and/or delayed response to fatty acid intake and ketogenic therapy | Supplementation with DHA in the form of lysoPC121 because it induces ameliorated cognition outcomes when applied prior to the onset of AD dementia |
| Difficulty in weight loss/control | FTO rs1558902 AA | Diet high in protein | Improved weight loss |
| FTO rs9939609 A/AA allele | Diet low in saturated fat Diet high in protein Improvement of aerobic fitness Daily physical activity Vitamin D supplementation |
SNPs
SNPs were genotyped using the TaqMan assays, provided by Life Technologies. We performed genotyping on the Open Array™ SNP Genotyping System (Life Technologies, Carlsbad, CA, USA) using microscope slide-sized plates and the OpenArray Accufill autoloader (Life Technologies, Carlsbad, CA, USA), following the manufacturer’s instructions.
Adaptation of the nutritional recommendations was assessed with a simple questionnaire provided at the time of facial reassessment. In more detail, participants were asked whether they adapted any dietary changes (“none”, “some”, or “all or the majority”) and for how long (“1-3 weeks”, “1-2 months”, “3-4 months”, or “5-6 months”). An at least partial adaptation of recommendations for a three-month period or longer was regarded as adherence to dietary recommendations [Supplementary Table 1].
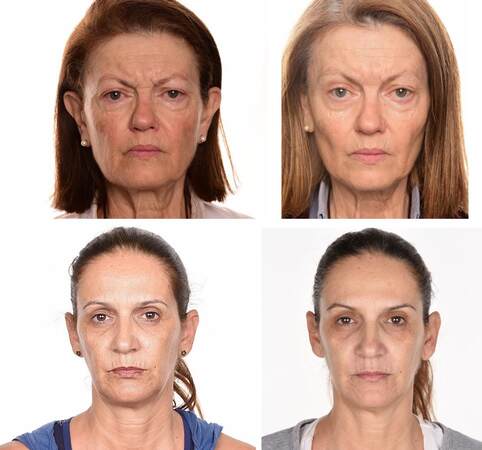
Horizontal forehead lines, zygomatic wrinkles or mid-cheek furrows, nasolabial folds, perioral expression wrinkles, and marionette line were assessed separately with the use of the Facial Wrinkles Assessment Scale (FWAS), a simple and reliable tool for the assessment of wrinkles[38] [Figure 1].Two authors (Nacopoulos C and Vesala AM) blinded to “adherence on recommended diet” scored the scales prior to initial intervention and at a follow-up visit, which took place about six months after the completion of the four PRF sessions.
Figure 1. Horizontal forehead lines, zygomatic wrinkles or mid-cheek furrows, nasolabial folds, perioral expression wrinkles, and marionette line were scored. The first case had deeper wrinkles with a medium score of 4 and despite her weight loss an improvement can been seen in all but perioral lines. In the second case an improvement from a score of 3 to a score of 2 has been marked only for forehead lines (patients have provided consent for publication of their photos).
Statistical analyses
Study participants were divided into two groups: those who reported an at least partial adherence to dietary recommendations for the majority of the time (Group A) and the rest (Group B). Changes in FWAS scores were compared with Mann-Whitney U Test. Unpaired t-tests were utilized for normally distributed variables and chi-squared test for nominal data.
RESULTS
In all, 43 out of 47 patients completed the study. Two patients ceased treatment, one for financial reasons and the other due to the level of perceived pain associated with treatment. Another was lost in the follow up. The fourth patient reported a health problem (gynecological cancer) which has not been reported during the PRF treatment and was excluded from the study.
Group A (subjects who reported an at least partial adherence to dietary recommendations for the majority of the time) consisted of 28 women and Group B of the remaining 15 women [Supplementary Table 1]. Demographic characteristics, total FWAS scores, and their changes for each group are presented in Table 2. An improvement in FWAS scores was recorded in both groups. However, as shown in Table 2, improvement in total FWAS scores was statistically more significant in Group A (adherent to dietary recommendations) (Z = 2.4, P = 0.008).
Facial Wrinkles Assessment Scale and basic confounding parameters assessed in each group
| Group A (n = 28) | Group B (n = 15) | ||
| Mean age (SD) | 52 (8.5) | 53.4 (6.1) | T = 0.53, P = 0.298 |
| Mean initial total FWAS (SD) | 13.7 (3.9) | 14.3 (4.6) | Z = 0.23, P = 0.409 |
| Change of total FWAS (SD) | 2.1 (2.4) | 0.5 (1.9) | Z = 2.4, P = 0.008 |
| Smoking | 9 | 6 | X = 0.2655, P = 0.606 |
| Smoking cessation | 1 | 0 | |
| Average weight loss (kg) | 0.5 | 0 |
DISCUSSION
Blood concentrates are increasingly being utilized as autologous products for aesthetic purposes because they contain platelets for growth factor release, fibrin scaffold for tissue remodeling, plasma proteins for collagen synthesis, white cells for inflammation and recovery, and stem cells for tissue repair and regeneration. Their outcomes regarding rejuvenation of photo-aged facial skin have been shown in small trials[4,39].
This study was not intended to verify previous positive outcomes. In other words, whether surgeons’ reported improvement in wrinkles scores is a biased or a true effect is beyond the scope of this observational study. A more appropriately designed study for this purpose has already been reported previously with positive results[4]. The current study was performed with the intention to see whether a personalized nutritional plan has an impact on PRF facial regeneration outcomes. For this reason, scorers were blinded to adherence to this plan. Thus, the additional skin aesthetics benefits conveyed by the dietary recommendations cannot be attributed to observer bias.
In addition, a relatively long-term adherence to a nutritional plan may be related to factors such as age, smoking, or smoking cessation. All patients initially visited the clinic to have a minimally invasive treatment. Personalized nutrition was offered as an add-on treatment. This may explain the relatively high percentage of the subjects (15/43) who opted not to adhere to a nutritional plan. Since a dietary change for a few days is unlikely to convey any skin changes in the long term (e.g., after six months), we only included those who sustained the recommended diet for at least half the study period. In this context, a difference between the aforementioned factors (age and smoking) is possible, can influence skin appearance and aesthetic results, and needs to be tested in pragmatic studies in which predefined strict entry and exclusion criteria are difficult to apply. Nevertheless, a statistically significant difference in relation to these factors was not noticed between our study groups.
An increasing amount of evidence suggests a protective effect of “healthy diet” on skin aging. Current dietary recommendations promote higher intakes of fruits, vegetables, fish, and PUFAs (polyunsaturated fatty acids)[40,41]. Nevertheless, several studies over the past few years have shown that what constitutes a healthy diet for an individual depends to some extent on his or her physiology and lifestyle[42-45]. Individualized nutrition advice, informed by knowledge of genetic variants, is increasingly favored over standard dietary guidelines based on population-wide averages[46].
Whether a personalized approach based on genotyping results conveys additional benefits over current nutritional recommendations in skin aesthetics, obesity, and other health issues is under study by various teams around the world. Regarding facial skin aesthetics, this study conforms to previous ones indicating a beneficial mid- or long-term effect of healthy nutrition[7,8]. In our cases, the inclusion of genetic variants helped the specification of general dietary recommendation that quite often propose a wide range of concentrations regarding macro- and micronutrients. This means that our personalized nutritional plans conformed to a “healthy diet” and the proposed optimal nutrition intake to stave off skin aging. In addition, the exact amounts of the various nutrients were further specified based on the genotype, as exemplified above.
Any assessment of all potential exposure variables, together with the various determinants of skin aging and appearance, would require hundreds or thousands of subjects. Despite this, the present study shows that nutritional interventions can be an important element to acknowledge, especially if we consider the fact that our era is characterized by minimally invasive and holistic care in facial aesthetics. In general, the relevant data suggest that, when nutritional recommendations are based on individual needs, they can improve the outcome of treatment and, consequently, should be considered as an integral part of any comprehensive care in facial aesthetics.
DECLARATIONS
Authors’ contributionsDesigned the study: Nacopoulos C, Vlastos I, Gkouskou K
Treated the patients: Nacopoulos C
Performed the molecular biology experiments and provided the nutrigenetic results: Gkouskou K
Offered the individualized nutritional plans: Lazou E
Assisted with data recording: Vesala AM
Supervised the whole process: Chaniotis D, Gkouskou K
Wrote and revised the initial draft: Nacopoulos C, Vlastos I, Gkouskou K, Vesala AM, Chaniotis D
Availability of data and materialsThe datasets during and/or analysed during the current study available from the corresponding author on reasonable request.
Financial support and sponsorshipNone.
Conflicts of interestGkouskou K is the owner of Embiodiagnostics laboratory that provides a range of comprehensive genomic services to assist individuals to reach decisions on diet and lifestyles based on their DNA profile. The remaining authors disclose no conflict of interest.
Ethical approval and consent to participateAn ethical approval was granted by the Ethical and Scientific Committee of the Plastic and Reconstructive Surgery Department of Agioi Anargyroi General Oncological Hospital of Kifisia, Athens, Greece (No. 1/26/09/2016) and the patients signed an informed consent prior to their treatment.
Consent for publicationWritten informed consent was obtained from all patients.
Copyright© The Author(s) 2021.
REFERENCES
1. Kim DH, Je YJ, Kim CD, et al. Can platelet-rich plasma be used for skin rejuvenation? Evaluation of effects of platelet-rich plasma on human dermal fibroblast. Ann Dermatol 2011;23:424-31.
2. Toffola ED, Furini F, Redaelli C, Prestifilippo E, Bejor M. Evaluation and treatment of synkinesis with botulinum toxin following facial nerve palsy. Disabil Rehabil 2010;32:1414-8.
3. Sclafani AP. Platelet-rich fibrin matrix for improvement of deep nasolabial folds. J Cosmet Dermatol 2010;9:66-71.
4. Nacopoulos C, Vesala AM. Lower facial regeneration with a combination of platelet-rich fibrin liquid matrices based on the low speed centrifugation concept-Cleopatra technique. J Cosmet Dermatol 2020;19:185-9.
5. Nacopoulos C, Gkouskou K, Karypidis D, et al. Telomere length and genetic variations affecting telomere length as biomarkers for facial regeneration with platelet-rich fibrin based on the low-speed centrifugation concept. J Cosmet Dermatol 2019;18:408-13.
7. Cosgrove MC, Franco OH, Granger SP, Murray PG, Mayes AE. Dietary nutrient intakes and skin-aging appearance among middle-aged American women. Am J Clin Nutr 2007;86:1225-31.
8. Nagata C, Nakamura K, Wada K, et al. Association of dietary fat, vegetables and antioxidant micronutrients with skin ageing in Japanese women. Br J Nutr 2010;103:1493-8.
9. Qi Q, Bray GA, Hu FB, Sacks FM, Qi L. Weight-loss diets modify glucose-dependent insulinotropic polypeptide receptor rs2287019 genotype effects on changes in body weight, fasting glucose, and insulin resistance: the Preventing Overweight Using Novel Dietary Strategies trial. Am J Clin Nutr 2012;95:506-13.
10. Qi Q, Bray GA, Smith SR, Hu FB, Sacks FM, Qi L. Insulin receptor substrate 1 gene variation modifies insulin resistance response to weight-loss diets in a 2-year randomized trial: the Preventing Overweight Using Novel Dietary Strategies (POUNDS LOST) trial. Circulation 2011;124:563-71.
11. Zhang X, Qi Q, Bray GA, Hu FB, Sacks FM, Qi L. APOA5 genotype modulates 2-y changes in lipid profile in response to weight-loss diet intervention: the Pounds Lost Trial. Am J Clin Nutr 2012;96:917-22.
12. Huang T, Huang J, Qi Q, et al. PCSK7 genotype modifies effect of a weight-loss diet on 2-year changes of insulin resistance: the POUNDS LOST trial. Diabetes Care 2015;38:439-44.
13. Martinez JA, Navas-Carretero S, Saris WH, Astrup A. Personalized weight loss strategies-the role of macronutrient distribution. Nat Rev Endocrinol 2014;10:749-60.
14. Chouinard-Watkins R, Conway V, Minihane AM, Jackson KG, Lovegrove JA, Plourde M. Interaction between BMI and APOE genotype is associated with changes in the plasma long-chain-PUFA response to a fish-oil supplement in healthy participants. Am J Clin Nutr 2015;102:505-13.
15. Kalantar Z, Eshraghian MR, Sotoudeh G, et al. Differences in the interaction between CETP Taq1B polymorphism and dietary fat intake on lipid profile of normolipedemic and dyslipidemic patients with type 2 diabetes mellitus. Clin Nutr 2018;37:270-5.
16. García-Calzón S, Martínez-González MA, Razquin C, et al. Pro12Ala polymorphism of the PPARγ2 gene interacts with a mediterranean diet to prevent telomere shortening in the PREDIMED-NAVARRA randomized trial. Circ Cardiovasc Genet 2015;8:91-9.
17. Shin C, Kim NH, Baik I. Sex-specific association between longitudinal changes in adiposity, FTO rs9939609 polymorphism, and leukocyte telomere length. J Am Coll Nutr 2016;35:245-54.
18. Arabi A, Khoueiry-Zgheib N, Awada Z, et al. CYP2R1 polymorphisms are important modulators of circulating 25-hydroxyvitamin D levels in elderly females with vitamin insufficiency, but not of the response to vitamin D supplementation. Osteoporos Int 2017;28:279-90.
19. Levin GP, Robinson-Cohen C, de Boer IH, et al. Genetic variants and associations of 25-hydroxyvitamin D concentrations with major clinical outcomes. JAMA 2012;308:1898-905.
20. Barry EL, Peacock JL, Rees JR, et al. Vitamin D receptor genotype, vitamin D3 supplementation, and risk of colorectal adenomas: a randomized clinical trial. JAMA Oncol 2017;3:628-35.
21. Steluti J, Reginaldo C, Selhub J, Paul L, Fisberg RM, Marchioni DM. Presence of circulating folic acid in plasma and its relation with dietary intake, vitamin B complex concentrations and genetic variants. Eur J Nutr 2019;58:3069-77.
22. Ribeiro MR, Lima RPA, Lisboa JVC, et al. Influence of the C677T polymorphism of the MTHFR gene on oxidative stress in women with overweight or obesity: response to a dietary folate intervention. J Am Coll Nutr 2018;37:677-84.
23. Mocchegiani E, Costarelli L, Giacconi R, et al. Micronutrient-gene interactions related to inflammatory/immune response and antioxidant activity in ageing and inflammation. A systematic review. Mech Ageing Dev 2014;136-137:29-49.
24. Joffe YT, Collins M, Goedecke JH. The relationship between dietary fatty acids and inflammatory genes on the obese phenotype and serum lipids. Nutrients 2013;5:1672-705.
25. Rode L, Nordestgaard BG, Weischer M, Bojesen SE. Increased body mass index, elevated C-reactive protein, and short telomere length. J Clin Endocrinol Metab 2014;99:E1671-5.
26. Costa KA, Corbin KD, Niculescu MD, Galanko JA, Zeisel SH. Identification of new genetic polymorphisms that alter the dietary requirement for choline and vary in their distribution across ethnic and racial groups. FASEB J 2014;28:2970-8.
27. Fischer LM, da Costa KA, Kwock L, Galanko J, Zeisel SH. Dietary choline requirements of women: effects of estrogen and genetic variation. Am J Clin Nutr 2010;92:1113-9.
28. Baumert P, Lake MJ, Stewart CE, Drust B, Erskine RM. Genetic variation and exercise-induced muscle damage: implications for athletic performance, injury and ageing. Eur J Appl Physiol 2016;116:1595-625.
29. Movérare-Skrtic S, Henning P, Liu X, et al. Osteoblast-derived WNT16 represses osteoclastogenesis and prevents cortical bone fragility fractures. Nat Med 2014;20:1279-88.
30. Kim JH, Jung ES, Kim CH, Youn H, Kim HR. Genetic associations of body composition, flexibility and injury risk with ACE, ACTN3 and COL5A1 polymorphisms in Korean ballerinas. J Exerc Nutrition Biochem 2014;18:205-14.
31. Zheng HF, Tobias JH, Duncan E, et al. WNT16 influences bone mineral density, cortical bone thickness, bone strength, and osteoporotic fracture risk. PLoS Genet 2012;8:e1002745.
32. Medina-Gomez C, Kemp JP, Estrada K, et al. Meta-analysis of genome-wide scans for total body BMD in children and adults reveals allelic heterogeneity and age-specific effects at the WNT16 locus. PLoS Genet 2012;8:e1002718.
33. Mons U, Müezzinler A, Schöttker B, et al. Leukocyte telomere length and all-cause, cardiovascular disease, and cancer mortality: results from individual-participant-data meta-analysis of 2 large prospective cohort studies. Am J Epidemiol 2017;185:1317-26.
34. Codd V, Nelson CP, Albrecht E, et al. CARDIoGRAM consortium. Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet 2013;45:422-7, 427e1.
35. Gkouskou K, Lazou E, Skoufas E, Eliopoulos AG. Genetically guided mediterranean diet for the personalized nutritional management of type 2 diabetes mellitus. Nutrients 2021;13:355.
36. Gkouskou KK, Grammatikopoulou MG, Vlastos I, Sanoudou D, Eliopoulos AG. Genotype-guided dietary supplementation in precision nutrition. Nutr Rev 2020;nuaa132.
37. Gkouskou K, Vlastos I, Karkalousos P, Chaniotis D, Sanoudou D, Eliopoulos AG. The "virtual digital twins" concept in precision nutrition. Adv Nutr 2020;11:1405-13.
38. Lemperle G, Holmes RE, Cohen SR, Lemperle SM. A classification of facial wrinkles. Plast Reconstr Surg 2001;108:1735-50; discussion 1751.
39. Alam M, Hughart R, Champlain A, et al. Effect of platelet-rich plasma injection for rejuvenation of photoaged facial skin: a randomized clinical trial. JAMA Dermatol 2018;154:1447-52.
40. Cao C, Xiao Z, Wu Y, Ge C. Diet and skin aging-from the perspective of food nutrition. Nutrients 2020;12:870.
41. Schagen SK, Zampeli VA, Makrantonaki E, Zouboulis CC. Discovering the link between nutrition and skin aging. Dermatoendocrinol 2012;4:298-307.
42. Franzago M, Santurbano D, Vitacolonna E, Stuppia L. Genes and Diet in the Prevention of Chronic Diseases in Future Generations. Int J Mol Sci 2020;21:2633.
43. Mendes-Soares H, Raveh-Sadka T, Azulay S, et al. Model of personalized postprandial glycemic response to food developed for an Israeli cohort predicts responses in Midwestern American individuals. Am J Clin Nutr 2019;110:63-75.
44. Phillips SA, Ali M, Modrich C, et al. Advances in health technology use and implementation in the era of healthy living: implications for precision medicine. Prog Cardiovasc Dis 2019;62:44-9.
45. Aruoma OI, Hausman-Cohen S, Pizano J, et al. Personalized nutrition: translating the science of nutrigenomics into practice: proceedings from the 2018 American College of Nutrition Meeting. J Am Coll Nutr 2019;38:287-301.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Nacopoulos C, Vlastos I, Vesala AM, Lazou E, Chaniotis D, Gkouskou K. A personalized nutrition plan based on genetic profile improves outcomes of facial regeneration with Platelet-Rich Fibrin liquid matrices. Plast Aesthet Res 2021;8:56. http://dx.doi.org/10.20517/2347-9264.2021.46
AMA Style
Nacopoulos C, Vlastos I, Vesala AM, Lazou E, Chaniotis D, Gkouskou K. A personalized nutrition plan based on genetic profile improves outcomes of facial regeneration with Platelet-Rich Fibrin liquid matrices. Plastic and Aesthetic Research. 2021; 8: 56. http://dx.doi.org/10.20517/2347-9264.2021.46
Chicago/Turabian Style
Nacopoulos, Cleopatra, Ioannis Vlastos, Anna-Maria Vesala, Evgenia Lazou, Dimitrios Chaniotis, Kalliopi Gkouskou. 2021. "A personalized nutrition plan based on genetic profile improves outcomes of facial regeneration with Platelet-Rich Fibrin liquid matrices" Plastic and Aesthetic Research. 8: 56. http://dx.doi.org/10.20517/2347-9264.2021.46
ACS Style
Nacopoulos, C.; Vlastos I.; Vesala A.M.; Lazou E.; Chaniotis D.; Gkouskou K. A personalized nutrition plan based on genetic profile improves outcomes of facial regeneration with Platelet-Rich Fibrin liquid matrices. Plast. Aesthet. Res. 2021, 8, 56. http://dx.doi.org/10.20517/2347-9264.2021.46
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 3 clicks
Cite This Article 3 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.