Lateral abdominal wall reconstruction
Abstract
Lateral abdominal wall (LAW) defects are defined as hernias, bulges, or surgical wounds that occur within the anatomic region bounded by the linea semilunaris, costal margin, iliac crest, and paraspinous musculature. Reconstruction of the LAW is complicated by the relatively complex anatomy, asymmetric biomechanical forces on the repair, and progressive nature of concomitant denervation injuries. Furthermore, the relative rarity and varied nature of these defects have complicated comparative analysis and the development of consensus regarding optimal surgical management. Although mesh reinforcement of LAW defects is a universal component of available repair techniques, significant variation exists regarding mesh material selection, anatomic plane utilization, and extent of mesh reinforcement. Special consideration must be given to extirpative defects that extend beyond the aforementioned boundaries of the LAW. In this review, we outline the incidence of LAW defects, pertinent risk factors, common history and physical examination findings, supplementary diagnostic modalities, defect classification systems, surgical indications, and available repair techniques. The outcomes data in this review are presented to help guide surgical management and optimize outcomes for affected patients.
Keywords
INTRODUCTION
Lateral abdominal wall (LAW) defects are relatively complex musculofascial pathologies that often require the expertise of a reconstructive surgeon. A thorough understanding of the strengths and weaknesses of available surgical options is important to increase the probability of reconstructive success for the specific clinical scenario encountered.
Incidence
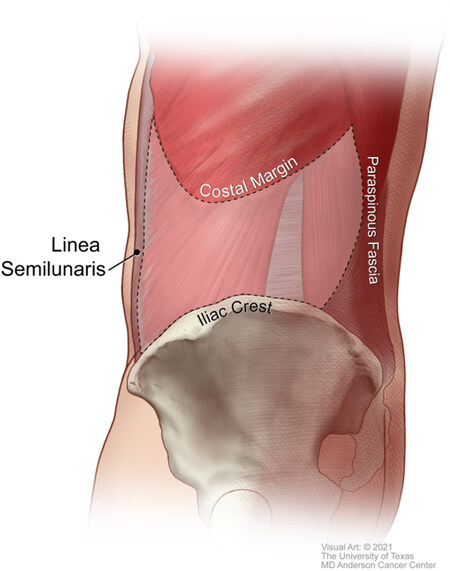
LAW defects are defined as hernias, bulges, or surgical wounds that occur within the anatomic region bounded by the linea semilunaris, paraspinous muscles, costal margin, and iliac crest [Figure 1]. Although these defects may arise from congenital causes, most LAW defects occur as a consequence of traumatic injury or surgical intervention. Structural weakening of the LAW may result from direct injury to the myofascial structures, indirect attenuation caused by disruption of the segmental motor innervation of this region, or combinations thereof[1]. Procedures involving retroperitoneal access, chevron/Kocher incisions, and direct oncologic resection of the abdominal wall are associated with postoperative flank weakness and/or hernia at a reported rate ranging from 8% to 57%[2-4]. In a meta-analysis of 26 studies, Zhou and Carlson[5] identified a flank hernia incidence of 14% following nephrectomy and 20% following retroperitoneal abdominal aortic aneurysm repair.
Risk factors
Patient-specific risk factors that have been reported to be associated with the postoperative development of a flank hernia or bulge include elevated body mass index, history of tobacco use, chronic constipation, abdominal distension, end-stage renal disease, carcinoma, coronary artery disease, and chronic immunosuppression[2,5-7]. Surgical risk factors include retroperitoneal incision length greater than 15 cm, single-layer wound closure, failure to identify and/or preserve the neurovascular bundles, and postoperative surgical site infection[6,7].
Classification
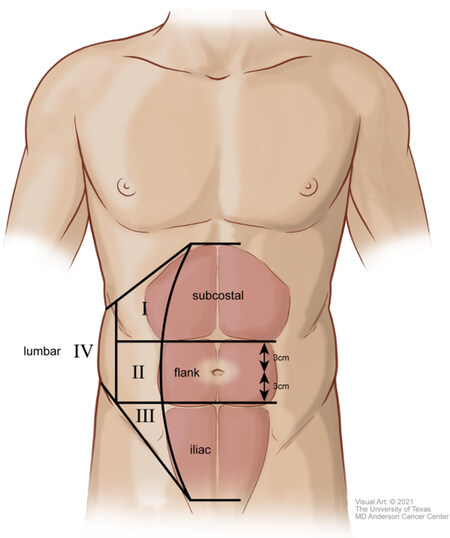
LAW defects can be classified according to their specific anatomic subregion, underlying cause, and morphology. In 2007, the European Hernia Society created a classification system to describe both ventral abdominal wall and LAW incisional hernias[8]. In this classification scheme, the traditional borders of the LAW noted above are divided into four anatomic subregions: subcostal (between the costal margin and a horizontal line 3 cm above the umbilicus), flank (lateral to the rectus sheath extending from 3 cm above to
In addition to anatomic location, LAW defects can be classified as excisional defects, true hernias, or bulges. Excisional defects occur following resection of abdominal wall muscle, fascia, or skin. In contrast, hernias and bulges are a result of myofascial weakening arising from congenital, traumatic, or iatrogenic causes. Hernias and bulges can be differentiated by whether there is a true musculofascial opening (hernia) or not (bulge)[2].
Diagnosis
LAW defects are often diagnosed using a combination of patient history, physical examination, and confirmatory imaging. Affected patients often note an asymmetric protrusion on the affected side. Other patient concerns may include pain of the adjacent oblique or paraspinous musculature, gastrointestinal complaints, psychological distress, and difficulty with ambulation or other activities of daily living[5,9]. Although emergent symptoms such as strangulation and incarceration are possible with flank hernias, these findings are less common when compared with ventral abdominal wall defects. Upon physical examination, one can often identify unilateral flank asymmetry by visual inspection alone. It is important to differentiate LAW hernias from bulges. Hernias involve a true myofascial defect, whereas bulges result from progressive attenuation of the myofascial tissues, most commonly after denervation injury. Myofascial defects, if present, may be difficult to discern in patients with an elevated body mass index. Computed tomography imaging is helpful to differentiate between hernias and bulges, and provides additional detail regarding the size of the defect, affected myofascial structures, and proximity of intra-abdominal or retroperitoneal structures.
SURGICAL MANAGEMENT
Although elective surgical correction is generally recommended in medically fit individuals with symptomatic LAW hernias, surgical intervention is less frequently undertaken in patients with a flank bulge in the absence of a true fascial defect. Whether elective reconstruction is undertaken depends on patient motivation, a thorough assessment of clinical risks and benefits, and the probability of success of the procedure.
Surgical techniques
Several surgical techniques have been proposed that vary regarding incision placement, type of mesh used, location of mesh placement, extent of mesh reinforcement, and surgical approach. Further consideration must be given to defects that cross into adjacent anatomic regions or require additional soft tissue reconstruction.
Incision placement
Given that many LAW defects occur as a result of prior surgical intervention, many techniques involve re-entering the abdomen through the initial incision made during the primary procedure. This approach allows excellent visualization of the defect as well as direct excision of scar tissue, foreign bodies, and previously placed mesh[3]. One must be careful to preserve the cutaneous perforators because failure to do so will likely result in skin necrosis and wound breakdown when large flaps are raised. Zieren et al.[10] championed a midline approach through a median laparotomy incision. Proposed benefits include complete access to the defect through an uncompromised anatomic plane and the potential ability to offset asymmetric abdominal forces via mesh fixation to the contralateral rectus sheath. However, this approach is limited by the significant morbidity of the laparotomy incision and risk of infection and herniation at this site. Access to the LAW through a suprapubic abdominoplasty incision was proposed by Hoffman et al.[9] in 2004. This approach offers visualization and access to both the ipsilateral and contralateral abdominal walls. Removal of excess skin and adipose tissue may offer additional cosmetic and functional benefits for patients. The major drawback is the need to elevate large soft tissue flaps. Furthermore, the utility of this approach may be limited in patients without significant abdominal lipodystrophy.
Type of mesh
Both synthetic and biologic meshes have been successfully used in the reconstruction of LAW defects. Important variables that may influence mesh selection include patient comorbidities, wound contamination, prior radiation, defect size, surgical technique, availability of omentum, and the quality of overlying soft tissue[1]. Synthetic mesh is often believed to have greater prolonged durability than biologic mesh. However, the prolonged foreign-body response associated with synthetic mesh can promote bowel adhesions, obstruction, and erosion through adjacent skin and viscera[1]. The regenerative properties of biologic mesh confer greater resistance to infection and adhesions, and thus these meshes are often favored for use in contaminated wounds. In cases where infection or exposure of the mesh does occur, biologic mesh can often be managed conservatively with dressing changes or local debridement, whereas synthetic mesh is more likely to require surgical explantation[3]. Revisionary procedures following biologic mesh implantation are often less complex owing to fewer and less tenacious adhesions and because these meshes can be incised and closed like native fascia[1]. Although biologic mesh is typically associated with higher per-unit cost compared with synthetic alternatives, no studies so far have provided a long-term cost analysis for LAW reconstruction.
Location of mesh placement
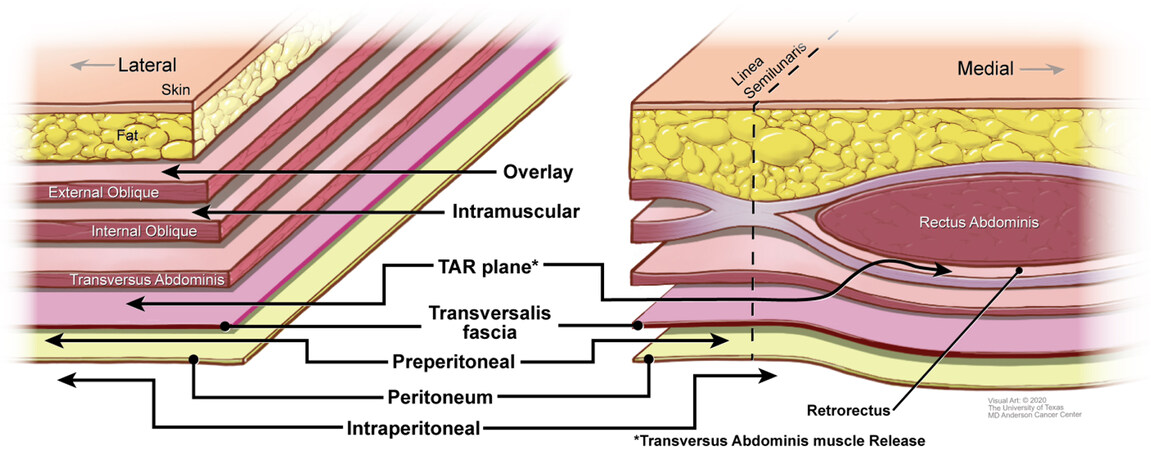
The main anatomic locations of mesh placement used in LAW reconstruction are the onlay (overlay), intramural (also known as intraparietal), transversus abdominus muscle release (TAR), and underlay planes [Figure 3]. The overlay plane is located superficial to the anterior rectus sheath medially and the external oblique muscle laterally. The utility of onlay mesh is limited by an increased risk of recurrence and surgical site infection. Nevertheless, the use of this plane may be indicated in the reconstruction of select retroperitoneal defects where vital structures impede the use of other options and in techniques that use multiple layers of mesh reinforcement[1].
The intramural plane lies between the internal and external oblique muscles. A significant benefit of this approach is the lack of important neurovascular structures in this plane[11]. Reconstruction in the intramural plane often requires imbrication of internal oblique myofascial advancement flaps prior to mesh placement and careful re-approximation of the external oblique muscle over the mesh[3,12,13].
The transversus abdominus muscle release (TAR) plane exists between the transversus abdominus muscle and transversalis fascia and can be extended medially to enter the retrorectus plane. It allows placement of a large piece of mesh superficial to the posterior rectus sheath medially and transversalis fascia laterally[10,14,15].
The underlay plane refers to both the preperitoneal and intraperitoneal planes. The preperitoneal plane exists between the transversalis fascia and the peritoneum. This can be a potentially challenging dissection owing to frequent obliteration of the tissue planes. The intraperitoneal plane lies deep to the peritoneal lining and is a common anatomic plane for minimally invasive approaches. The major limitation of the intraperitoneal plane is potential complications resulting from contact between the mesh and intraperitoneal viscera.
Extent of mesh reinforcement
The extent of mesh reinforcement is another important variable that can significantly influence the ultimate stability of the LAW reconstruction. There are two main categories of mesh reinforcement that vary with respect to the amount of mesh used and the underlying biophysical principles: direct mesh repair and pillar-anchored repair.
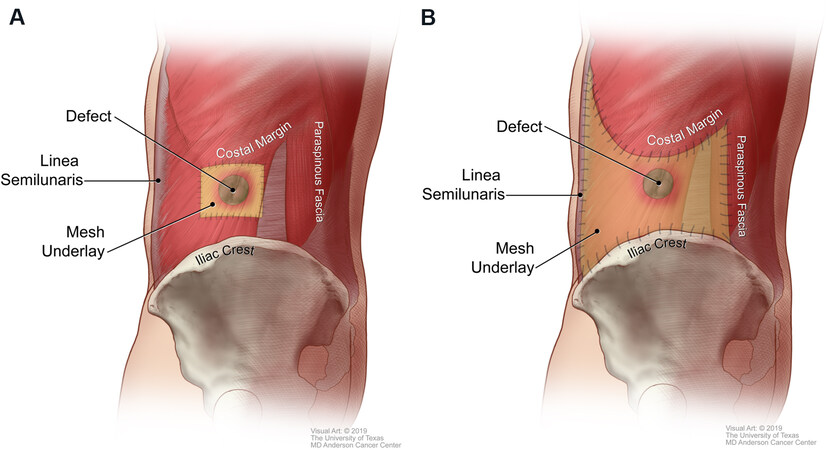
Direct mesh repair refers to the use of an underlay mesh that extends 3-5 cm beyond the immediate boundaries of the defect and is secured locally to healthy musculature [Figure 4A][12]. This technique is considered “load-sharing” because any applied force is transmitted to both the mesh and the muscular abdominal wall. Proponents of this technique suggest that anchoring the mesh to a dynamic structure optimizes abdominal wall compliance and decreases distraction forces at the mesh-tissue interface, which may ultimately translate to improved outcomes. They also cite a potential decrease in the risk of postoperative infection, adhesions, and patient discomfort because a relatively small piece of mesh is used[12]. In contrast, other techniques use a larger piece of mesh that is secured to more distant fixed structural supports.
The pillar-anchored repair involves anchoring an underlay mesh to stable points of fixation along the borders of the LAW: the linea semilunaris, costal margin, inguinal ligament/iliac crest, and posterior paraspinal fascia [Figure 4B][2]. This technique is considered “load-bearing” because most of the applied force is transmitted to the mesh and static supporting structures instead of to the abdominal wall musculature. A recent study by Kapur et al.[2] compared outcomes of LAW reconstruction using direct mesh repair with those of LAW reconstruction using pillar-anchored repair. That study included 106 consecutive patients with a median follow-up of 28 months. Patients who underwent LAW reconstruction with pillar-anchored repair had a 3.5-fold lower risk of hernia recurrence than those who underwent LAW reconstruction with direct mesh repair. Other outcomes such as surgical site occurrences and re-operation for complications did not differ between the two groups.
Minimally invasive surgical techniques
Laparoscopic and robotic techniques have been applied to LAW reconstruction to repair these defects in a minimally invasive fashion. Benefits associated with minimally invasive approaches include reduced infections, less postoperative pain, shorter length of hospital stay, quicker return to normal activities, and the potential for improved cosmesis[16,17]. Minimally invasive repairs often use intraperitoneal onlay mesh or transabdominal pre-peritoneal tissue planes for mesh placement. Although more technically challenging, transabdominal pre-peritoneal repairs offer the added benefit of shielding the mesh from the abdominal viscera[17]. Although less common, successful laparoscopic repair of interparietal flank hernias has also been described[18]. Drawbacks of minimally invasive approaches include the potential for visceral injury during bowel mobilization to improve visualization, difficulty addressing bulge-type defects, the challenge of maneuvering and securing large pieces of mesh, and a potentially steeper learning curve[19,20].
Hybrid techniques
Newer hybrid modalities that combine open and laparoscopic approaches have been proposed with the aim of capitalizing on the relative merits of each approach. Dissatisfied with residual bulges following traditional minimally invasive repair of flank hernias, Amaral et al.[21] in 2019 published a report of their experience with a novel hybrid “sandwich” technique. The procedure begins with laparoscopic adhesiolysis and colonic mobilization to visualize the defect and reduce the intra-abdominal components of the hernia. Following this, an open approach is used to excise the hernia sac, perform fascial closure, and place a reinforcing onlay mesh. Finally, the procedure returns to a laparoscopic approach for an intraperitoneal onlay mesh repair. In their series of 16 patients, the authors reported 1 recurrence (6%) at 12 months after the surgery. The authors acknowledged that their recurrence rate is not superior to that reported in other published series of pure laparoscopic repairs by Moreno-Egea et al.[16] (3%) and Novitsky (0%), but the authors noted that excision of the hernia sac via an open approach may offer potential benefits in terms of reducing postoperative wound complications[14,21]. However, further research is needed to validate this claim and justify the likely increase in operative time and postoperative pain associated with this procedure.
Special boundary considerations
Defects that extend beyond the anatomic boundaries of the LAW and into adjacent territories such as the ventral abdomen, chest wall, retroperitoneum, and pelvis can pose a reconstructive challenge that merits special consideration. This often occurs in the context of oncologic extirpation.
Defects that extend medially beyond the linea semilunaris require reinforcement of both the ventral abdominal wall and the LAW. Because the linea semilunaris has been compromised, it is no longer a stable fixation point for repair. In these instances, a modified pillar-anchored repair can be performed by securing a wide underlay mesh spanning the remaining boundaries of the LAW and the nearest pillar of strength, which is often the linea alba or contralateral linea semilunaris[1,2].
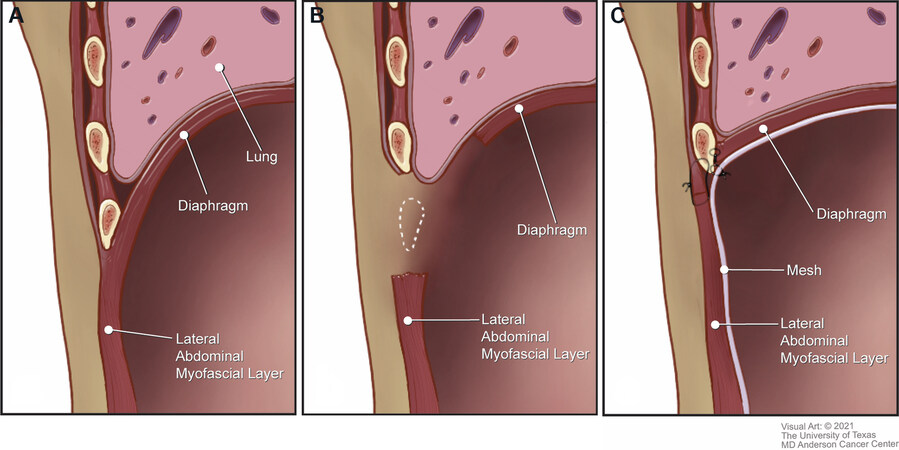
Large chest wall resections can result in destabilization of the superior border of the LAW, loss of separation between the thoracic and abdominal cavities, and compromised respiratory mechanics. Lateral subcostal full-thickness excisions often include components of the ribs, diaphragm, and corresponding connective tissues. Although small peripheral defects may be corrected by suturing the diaphragm to the cut edge of the rib cage, larger resections require a more sophisticated reconstruction to restore the complex three-dimensional anatomy of this region[1]. The remnant LAW and diaphragm are advanced and anchored to the unresected cephalad rib, and an underlay mesh is used to reinforce the repair [Figure 5].
Figure 5. Management of extensive subcostal lateral abdominal wall defects. (A) Anatomic relationship of the diaphragm and the lateral abdominal wall. (B) Subcostal defect. (C) Underlay mesh placement to reinforce and re-establish separation between the thoracic and abdominal cavities. The mesh-diaphragm-abdominal wall complex is affixed to the intact rib.
Resection of the iliac crest during an internal hemipelvectomy compromises the caudal support of the LAW. Reconstruction of these defects can be accomplished by securing an underlay mesh to the cephalad LAW and the superior aspect of the remaining iliac crest. The mesh is suture-anchored to the iliac crest by drilling holes into the osseous margin or by using bone anchors.
Reconstruction of the posterior boundary of the LAW is complicated by the proximity of numerous important retroperitoneal structures. In cases where an underlay mesh cannot be used owing to the risk of neurovascular injury, the mesh may be placed in an onlay fashion. Mesh can also be placed in the transversus abdominus muscle release plane to reinforce this region. The mesh may be further reinforced by a soft tissue flap if significant retroperitoneal dead space is present[1].
Soft tissue reconstruction
Depending on the cause of the LAW defect, additional soft tissue reconstruction may be required to obliterate dead space and attain a tension-free closure. Important variables that influence operative planning include defect size, anatomic location, bacterial contamination, radiation exposure, and prior surgical history[22]. Given that the LAW is perfused by multiple overlapping angiosomes, many defects can be closed by local fasciocutaneous tissue advancement[1]. In certain cases, preoperative tissue expansion can be used to provide additional tissue for advancement. When local options are compromised by excessive skin resection, prior surgery, and radiation-induced fibrosis, pedicled or free flaps are required for wound closure[1,22]. Pedicled flap options for LAW reconstruction include vertical rectus abdominus, latissimus dorsi, anterolateral thigh vastus lateralis, and subtotal thigh flaps. If a pedicled flap would have limited reach or insufficient volume, a free tissue transfer is indicated. Thigh-based fasciocutaneous or musculocutaneous flaps offer large skin paddles and generous volume[1]. Regional recipient vessels include the superior/inferior epigastric, internal mammary, intercostal, and lumbar systems. Arteriovenous loops to the internal mammary and femoral vessels offer an alternative option if the aforementioned recipient vessels are unavailable.
OUTCOMES
Comparative analysis of LAW reconstruction outcomes is complicated by the relative rarity of these defects, variety of available surgical options, and overall heterogeneity of patient-specific factors that influence the reconstructive approach chosen. Much of the published literature on this topic consists of the presentation of novel surgical techniques in a small number of patients. Larger series have demonstrated the safety and efficacy of multiple approaches. In 2013, Moreno-Egea et al.[16] published a comparative analysis of 35 laparoscopic and 20 open lumbar hernia repairs. The recurrence rates for laparoscopic (2.9%) and open (15%) hernia repairs were not statistically different (P = 0.13). In 2016, Patel et al.[4] reported their experience with open mesh repair of LAW hernias in 61 patients. The authors reported an overall recurrence rate of 11.5% at a mean follow-up of 15.4 months. In the largest series of LAW reconstructions to date,
CONCLUSION
Reconstruction of the LAW is a complex undertaking with several different available surgical options. Although recent studies have demonstrated the superiority of a pillar-anchored, load-bearing mesh repair, stronger clinical evidence generated by randomized controlled trials is needed to further inform the most optimal approach for this complex surgical problem. Surgeons should be aware of all available surgical options and choose an approach that maximizes the likelihood of long-term success for the specific patient being treated.
DECLARATIONS
AcknowledgementsWe thank Erica Goodoff, Senior Scientific Editor in the Research Medical Library at The University of Texas MD Anderson Cancer Center, for editing this article.
Authors’ contributionsParticipated in the accumulation of data, literature review, writing, and editing of this manuscript: Smith JM, Kapur SK, Mericli AF, Baumann DP, Butler CE
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
2. Kapur SK, Liu J, Baumann DP, Butler CE. Surgical outcomes in lateral abdominal wall reconstruction: a comparative analysis of surgical techniques. J Am Coll Surg 2019;229:267-76.
3. Pezeshk RA, Pulikkottil BJ, Bailey SH, et al. An evidence-based model for the successful treatment of flank and lateral abdominal wall hernias. Plast Reconstr Surg 2015;136:377-85.
4. Patel PP, Warren JA, Mansour R, Cobb WS 4th, Carbonell AM. A large single-center experience of open lateral abdominal wall hernia repairs. Am Surg 2016;82:608-12.
5. Zhou DJ, Carlson MA. Incidence, etiology, management, and outcomes of flank hernia: review of published data. Hernia 2018;22:353-61.
6. Osman T, Emam A, Farouk A, ElSaeed K, Tawfeek AM, AbuHalima A. Risk factors for the development of flank hernias and bulges following surgical flank approaches to the kidney in adults. Arab J Urol 2018;16:453-9.
7. Matsen SL, Krosnick TA, Roseborough GS, et al. Preoperative and intraoperative determinants of incisional bulge following retroperitoneal aortic repair. Ann Vasc Surg 2006;20:183-7.
8. Muysoms FE, Miserez M, Berrevoet F, et al. Classification of primary and incisional abdominal wall hernias. Hernia 2009;13:407-14.
9. Hoffman RS, Smink DS, Noone RB, Noone RB Jr, Smink RD Jr. Surgical repair of the abdominal bulge: correction of a complication of the flank incision for retroperitoneal surgery. J Am Coll Surg 2004;199:830-5.
10. Zieren J, Menenakos C, Taymoorian K, Müller JM. Flank hernia and bulging after open nephrectomy: mesh repair by flank or median approach? Int Urol Nephrol 2007;39:989-93.
11. Veyrie N, Poghosyan T, Corigliano N, Canard G, Servajean S, Bouillot JL. Lateral incisional hernia repair by the retromuscular approach with polyester standard mesh: topographic considerations and long-term follow-up of 61 consecutive patients. World J Surg 2013;37:538-44.
12. Purnell CA, Park E, Turin SY, Dumanian GA. Postoperative flank defects, hernias, and bulges: a reliable method for repair. Plast Reconstr Surg 2016;137:994-1001.
13. Pineda DM, Rosato EL, Moore JH Jr. Flank bulge following retroperitoneal incisions: a myofascial flap repair that relieves pain and cosmetic Sequelae. Plast Reconstr Surg 2013;132:181e-3e.
15. Gibreel W, Sarr MG, Rosen M, Novitsky Y. Technical considerations in performing posterior component separation with transverse abdominis muscle release. Hernia 2016;20:449-59.
16. Moreno-Egea A, Alcaraz AC, Cuervo MC. Surgical options in lumbar hernia: laparoscopic versus open repair. A long-term prospective study. Surg Innov 2013;20:331-44.
17. Orenstein SB. Robotic flank hernia repair. In: Tsuda S, Kudsi OY, editors. Robotic-assisted minimally invasive surgery. Cham: Springer Nature Switzerland AG; 2019. p. 169-78.
18. Kalmar CL, Bower CE. Laparoscopic repair of interparietal abdominal wall hernias. J Surg Case Rep 2019;2019:rjz319.
19. Pulikkottil BJ, Pezeshk RA, Daniali LN, Bailey SH, Mapula S, Hoxworth RE. Lateral abdominal wall defects: the importance of anatomy and technique for a successful repair. Plast Reconstr Surg Glob Open 2015;3:e481.
20. Phillips MS, Krpata DM, Blatnik JA, Rosen MJ. Retromuscular preperitoneal repair of flank hernias. J Gastrointest Surg 2012;16:1548-53.
21. Amaral PHF, Tastaldi L, Barros PHF, et al. Combined open and laparoscopic approach for repair of flank hernias: technique description and medium-term outcomes of a single surgeon. Hernia 2019;23:157-65.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Smith JM, Kapur SK, Mericli AF, Baumann DP, Butler CE. Lateral abdominal wall reconstruction. Plast Aesthet Res 2021;8:46. http://dx.doi.org/10.20517/2347-9264.2021.49
AMA Style
Smith JM, Kapur SK, Mericli AF, Baumann DP, Butler CE. Lateral abdominal wall reconstruction. Plastic and Aesthetic Research. 2021; 8: 46. http://dx.doi.org/10.20517/2347-9264.2021.49
Chicago/Turabian Style
Smith, J. Michael, Sahil K. Kapur, Alexander F. Mericli, Donald P. Baumann, Charles E. Butler. 2021. "Lateral abdominal wall reconstruction" Plastic and Aesthetic Research. 8: 46. http://dx.doi.org/10.20517/2347-9264.2021.49
ACS Style
Smith, JM.; Kapur SK.; Mericli AF.; Baumann DP.; Butler CE. Lateral abdominal wall reconstruction. Plast. Aesthet. Res. 2021, 8, 46. http://dx.doi.org/10.20517/2347-9264.2021.49
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 11 clicks
Cite This Article 11 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.