Assessment of the lymphedema patient
Abstract
The various elements used in assessing the lymphedema patient are presented. These include the History and Physical examination and the various measurements that include limb circumference, limb volume, and bio-impedence spectroscopy. In addition, the different imaging techniques used to assess the lymphatic system are discussed. Finally, a treatment algorithm is presented for the treatment of the lymphedema patient.
Keywords
INTRODUCTION
As we learn more about lymphedema and have more techniques to treat it, assessment of the lymphedema patient becomes more and more important. With a proper assessment, we can choose the most appropriate treatment for these patients.
History
Detailed history taking is important. First, one wants to know how lymphedema started, was there an instigating factor, or did it arise de-novo? Is this an isolated case in the patient’s family, or do other family members also have lymphedema? In other words, is this congenital or acquired. If there was an instigating factor, what was it, and how long ago did this occur? Has the patient had a lymph node dissection and/or radiation? A travel history is important since we know that the leading cause of lymphedema worldwide is filariasis[1]. Even though I live and practice in the United States, I have several patients in my practice whose lymphedema was caused by filaria. Whether the patient has spent time in an area where filariasis is endemic is, therefore, something one needs to know. The duration of the condition is also important. We know that the best results from lymphedema treatment are obtained early in the course of the disease. Duration will also give an idea of how advanced the condition is likely to be, which may impact treatment. Cellulitis is a common accompaniment of lymphedema, and each episode of cellulitis leads to further damage to the lymphatic system[2]. For this reason, a history of infection and its frequency is important. How does the patient manage the swelling? Do they wear compression and, if so, is it effective? Finally, we want to know what treatment the patient has had and, more importantly, how they responded to that treatment.
Physical examination
On completion of the history, the patient should be examined. There are several things that we look for specifically in the patient with lymphedema:
1. How big is the limb and, if it is unilateral, how does it compare with the contralateral side?
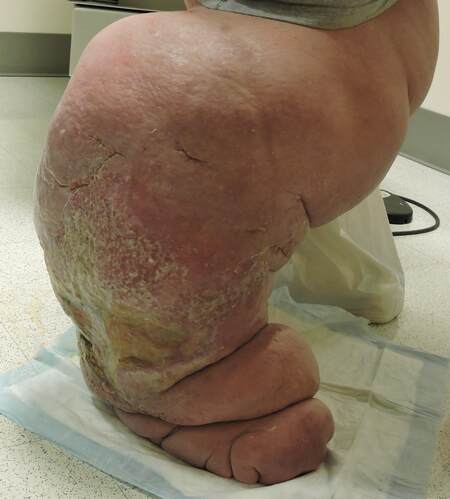
2. Are there trophic skin changes? [Figure 1]
Figure 1. This patient with extreme late-stage lymphedema shows trophic skin changes. There are folds of edematous skin and thick rigorous skin, giving rise to fissures and increasing the risk of infection.
3. Is there any sign of infection?
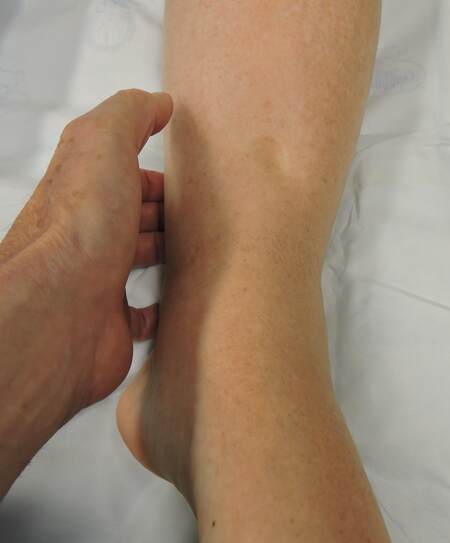
4. Is there pitting edema? [Figure 2]
PATIENT ASSESSMENT
While all these modalities are assessed, no one alone is pathognomonic of lymphedema. However, taken together, the information they give helps develop the diagnosis.
Limb circumference
Most practitioners measure limb circumference, and this is an important tool not only as a static evaluation but also as a dynamic, ongoing measurement to assess the patient’s progress. However, limb circumference needs to be standardized if it is to be of any use. So, the same measurement points need to be chosen.
Limb volume
Limb volume is of greater use than mere limb circumference measurements. Limb volume can, however, be calculated from limb circumference measurements using the inverted cone formula. Brørson has published the formula based on 4 cm increment measurements[3].
The traditional way of measuring limb volume was with water displacement. While this works, not every clinic is equipped to do these measurements.
Optoelectronic volumetry using a Perometer is a very accurate way of measuring limb volume[4]. Perometry uses a frame with light transmitters on two sides and photo-sensors on the opposite sides. The limb to be measured is placed within the frame, and the frame is moved along the limb to give a very accurate measurement of the limb volume. Hands and feet are not measured using this technique.
Bio-impedence spectroscopy
This test measures the time taken for a small electrical current to pass through the tissues. It is based on the premise that the amount of opposition to the flow of electrical current (impedance) through the body is inversely proportional to the volume of fluid in the tissue.
In lymphedema, tissue impedance decreases, which is a useful test that can be used for initial assessment and to measure the patient’s response to treatment[5]. However, it may be misleading in early or pre-clinical lymphedema, where the bio-impedence may be normal. Therefore, it is most useful as a trend measurement over time.
Imaging
Several imaging modalities can be used to assess the lymphedema patient. As with the assessments above, no one imaging modality is used for the complete assessment. Taken together, though, they provide the best information on what is going on with the patient.
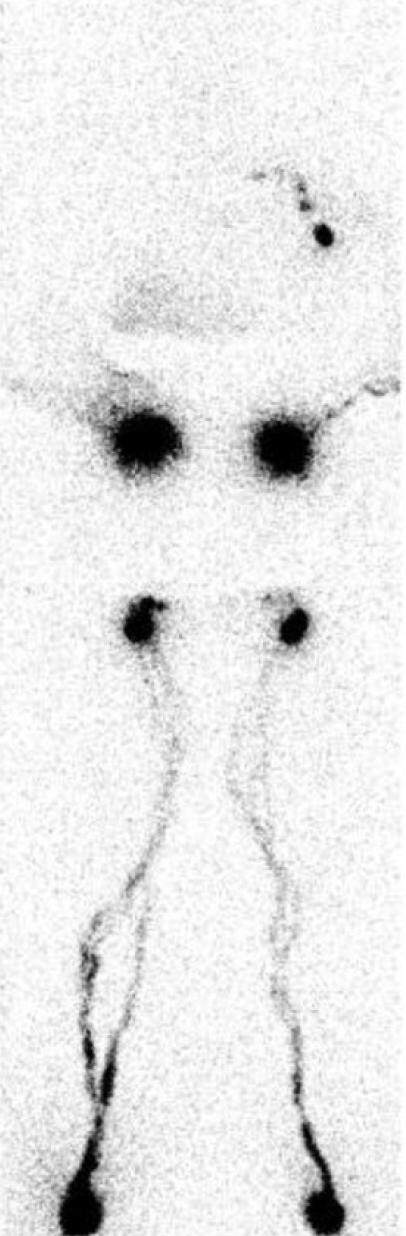
(1) Lymphoscintigraphy [Figure 3]: This involves the injection of filtered sulphur colloid, Technetium 99, into the dermis of the affected limb[6,7]. The Technetium is then transported to the draining lymph nodes. This text gives excellent information on the function of the lymphatic system, and if I have any doubt as to the diagnosis, this is the imaging of choice. It tells you whether the lymphatic system is normal or not. However, it doesn’t give good anatomic information so that if surgery is being planned, more information is needed. In particular, it doesn’t show the superficial lymphatics in any detail.
Figure 3. This is a lymphoscintigram of a post-mastectomy patient. Notice the absence of Technetium pickup in the right axilla indicating distal obstruction of the lymphatic system.
(2) Fluorescent lymphangiography: This is achieved with the injection of indocyanine green (ICG). ICG is a fluorescent dye activated by a laser light source, and the fluorescence can be visualized with a near infra-red camera [Figure 4]. The advantage is that it gives excellent information on superficial lymphatics. Its disadvantage is that it only penetrates about 1.5 cm, so it doesn’t give any information on the deeper lymphatics or other structures in the limb. The significance of the different patterns of ICG that are seen, linear, splash, stardust, and diffuse, have been elucidated and correlate with decreasing circumference of the lymphatics and increasing sclerosis. Apart from its use as a diagnostic tool, ICG is important for mapping the lymphatics at the time of surgery so that incisions can be planned accurately[7,8].
Figure 4. This is the splash pattern seen with ICG lymphangiography in early lymphedema. The lymph is mainly contained within lymphatic channels, but a “splash” of ICG is also seen in the soft tissues. ICG: Indocyanine green.
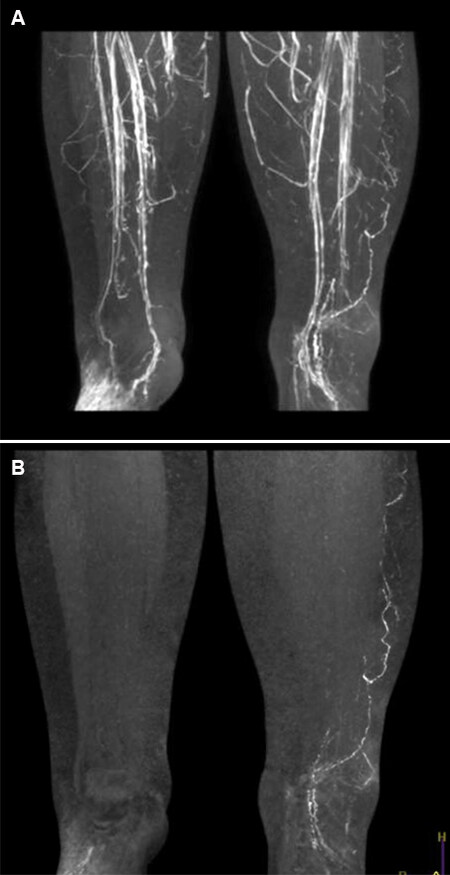
(3) MR lymphangiography: There are several techniques for using MR in assessing the lymphedema patient. Imaging can be achieved with or without contrast. A technique known as the “dark blood” technique can be used [Figure 5]. The contrast used is Gadolinium. The Dark Blood technique involves the intravenous injection of Feraheme, which essentially blacks out the venous signal and eliminates the difficulty of deciding what a vein is and what is a lymphatic[9,10]. However, one of the most significant advantages of MR is that it gives accurate information on other conditions appearing in the limb. Feraheme has been associated with hypersensitivity reactions as well as anaphylactic shock. Hypotension has also been reported[11].
Figure 5. (A) MR Lymphangiogram of a patient with lymphedema of the left leg. Note the difficulty of identifying veins and lymphatics. (B) The same patient with Feraheme producing a “dark blood” image clearly showing lymphatic channels and no veins.
(4) Ultrahigh-frequency ultrasound: This is the newest imaging modality to be used. It has the advantage of identifying lymphatics and veins, and the ultra-high-frequency (up to 70 MHz) probes give an excellent definition[12]. The significant disadvantage of UHF: ultrasound is lack of user-friendliness and the time is taken to do an examination.
DISCUSSION
Once all of this information has been gathered, the provider should understand:
(1) the etiology of the condition;
(2) the extent, or the International Society of Lymphology (ISL) stage;
(3) the condition of the lymphatics;
(4) patient’s previous response to treatment which may predict future response.
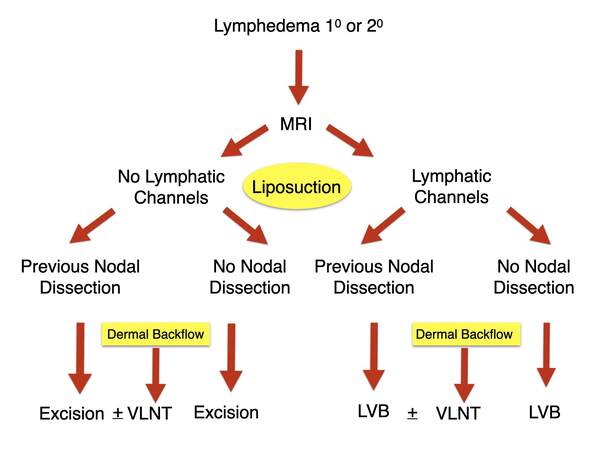
In this way, a treatment plan can be formulated, and as already mentioned, the modalities listed above may be used to assess the patient’s progress on an ongoing basis. Finally, to show the practical application of these assessments, my algorithm for managing the lymphedema patient is presented [Figure 6] in this algorithm, which is used for either primary or secondary lymphedema, all patients who can receive an MR Lymphangiogram. If for some reason, they cannot have an MR, then fluorescent lymphangiography is performed with ICG. If no lymphatic channels are seen, patients are divided into those who have had a previous lymph node dissection and those who have not. The latter group represents primary cases. In either case, the only treatment offered is excisional, the commonest of which is liposuction. The exception is if they have areas of dermal backflow. In that case, a vascularized lymph node transplant (VLNT) to the area of dermal backflow is considered.
Figure 6. Algorithm for treatment of lymphedema, either primary or secondary. LVB: Lymphatico-venous bypass; VLNT: vascularized lymph node transplant.
On the other hand, if lymphatic channels are seen, the patients are divided into those who have or have not had a lymph node dissection. Again, in either case, a lymphatico-venous bypass (LVB) is offered. Areas of dermal backflow may again be eligible for VLNT. Six to twelve months after either LVB or VLNT, patients are offered liposuction as these operations help improve lymphatic flow, but they do not address the accumulated fibro-adipose accumulation, which we see with lymphedema.
Conclusion
Several treatment options exist for patients with lymphedema. Choosing the correct treatment is vital. Patient assessment is of paramount importance in achieving this. There is no one test or finding that is pathognomic for lymphedema, and we need to use multiple modalities to conduct a proper assessment. These assessments are not just useful in the initial evaluation of these patients. They are also extremely useful in following our patients and assessing the results of our treatment.
DECLARATIONS
Authors’ contributionsThe author contributed solely to the article.
Availability of data and materialsThe material is based on the authors’ practice. The bibliography provides all the information on issues described and is available through PubMed.
Financial support and sponsorshipNone.
Conflicts of interestThe author declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
1. Molyneux DH. Filaria control and elimination: diagnostic, monitoring and surveillance needs. Trans R Soc Trop Med Hyg 2009;103:338-41.
2. Rodriguez JR, Hsieh F, Huang CT, Tsai TJ, Chen C, Cheng MH. Clinical features, microbiological epidemiology and recommendations for management of cellulitis in extremity lymphedema. J Surg Oncol 2020;121:25-36.
3. Brorson H, Höijer P. Standardised measurements used to order compression garments can be used to calculate arm volumes to evaluate lymphoedema treatment. J Plast Surg Hand Surg 2012;46:410-5.
4. Campanholi LL, Baiocchi JMT, Batista BN, Bergmann A, Fregnani JHTG, Duprat Neto JP. Agreement between optoelectronic volumetry and circumferential girth measurements to diagnose lymphedema in patients submitted to axillary radical lymphadenectomy for treatment of cutaneous melanoma. Lymphat Res Biol 2021; doi: 10.1089/lrb.2017.0081.
5. Douglass J, Graves P, Gordon S. Intrarater reliability of tonometry and bioimpedance spectroscopy to measure tissue compressibility and extracellular fluid in the legs of healthy young people in australia and myanmar. Lymphat Res Biol 2017;15:57-63.
6. diagnosis and treatment of peripheral lymphedema. Consensus document of the international society of lymphology executive committee. Lymphology 1995;28:113-7.
7. Chowdhry M, Rozen WM, Griffiths M. Lymphatic mapping and preoperative imaging in the management of post-mastectomy lymphoedema. Gland Surg 2016;5:187-96.
8. Narushima M, Yamamoto T, Ogata F, Yoshimatsu H, Mihara M, Koshima I. Indocyanine green lymphography findings in limb lymphedema. J Reconstr Microsurg 2016;32:72-9.
9. Mitsumori LM, McDonald ES, Wilson GJ, Neligan PC, Minoshima S, Maki JH. MR lymphangiography: how I do it. J Magn Reson Imaging 2015;42:1465-77.
10. Maki JH, Neligan PC, Briller N, Mitsumori LM, Wilson GJ. Dark blood magnetic resonance lymphangiography using dual-agent relaxivity contrast (DARC-MRL): a novel method combining gadolinium and iron contrast agents. Curr Probl Diagn Radiol 2016;45:174-9.
11. Lu M, Cohen MH, Rieves D, Pazdur R. FDA report: ferumoxytol for intravenous iron therapy in adult patients with chronic kidney disease. Am J Hematol 2010;85:315-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Neligan PC. Assessment of the lymphedema patient. Plast Aesthet Res 2021;8:45. http://dx.doi.org/10.20517/2347-9264.2021.39
AMA Style
Neligan PC. Assessment of the lymphedema patient. Plastic and Aesthetic Research. 2021; 8: 45. http://dx.doi.org/10.20517/2347-9264.2021.39
Chicago/Turabian Style
Neligan, Peter C.. 2021. "Assessment of the lymphedema patient" Plastic and Aesthetic Research. 8: 45. http://dx.doi.org/10.20517/2347-9264.2021.39
ACS Style
Neligan, PC. Assessment of the lymphedema patient. Plast. Aesthet. Res. 2021, 8, 45. http://dx.doi.org/10.20517/2347-9264.2021.39
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 23 clicks
Cite This Article 23 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.