Allogeneic epidermal substitutes in the treatment of chronic diabetic leg and foot ulcers
Abstract
Aim: Diabetic foot ulcers are the most common cause of nontraumatic lower extremity amputations in the industrialized world. Tissue-engineering products offer a lower extremity salvage strategy when healing does not proceed according to the standard of care. New allogeneic sheets are available for the management of diabetic leg and foot ulcers.
Methods: The endpoints of this case series study regard preliminary outcomes of the application of allogeneic keratinocytes composed of benzyl ester of hyaluronic acid to 16 diabetic foot and leg ulcers in 11 patients with type 2 diabetes mellitus.
Results: Between 21 and 70 days after cellular therapy, 6 out of 16 lesions were completely healed, reducing the wound dimension by 70% and improving the wound bed score by 52%.
Conclusion: The clinical results of the new allogeneic sheets indicate that allogeneic keratinocytes may represent an effective and safe therapy for diabetic foot and leg ulcers in the multidisciplinary approach to this diabetes-related complication.
Keywords
Introduction
The burden of diabetes-related complications is an inevitable consequence of the rise in the prevalence of diabetes mellitus worldwide. The lifetime probability of diabetics to develop a diabetic chronic ulcer is estimated at 10-25%.[1] The risk for patients, in particular with type 2 diabetes, of undergoing a lower extremity amputation is 23-fold higher than that of a nondiabetic.[2] Diabetic chronic ulcers definitively represent a significant cause of morbidity, hospitalization, and a huge financial cost.
Risk factors for diabetic chronic ulcers include vascular anomalies, peripheral neuropathy, imbalanced foot mechanical forces, impaired joint mobility, high body mass, foot instability, and history of previous ulceration or amputation. The standard of care for these wounds, as defined by the International Working Group on the Diabetic Foot, requires multidisciplinary management including control of systemic glucose, extremity vascularization, off-loading, debridement of necrotic tissue, control of local infection, and patient compliance.[3,4]
In this holistic approach, for lesions where the healing process is unsatisfactory, and no other underlying cause exists, there is an increasing need for more effective therapies that will stimulate healing of diabetic chronic ulcers. Tissue-engineered products, especially skin substitutes, both cellular and acellular, are emerging as new local therapy for the treatment of nonhealing diabetic chronic ulcers.[5]
This case-series study was performed to determine wound reduction and healing following use of a new cultured allogeneic keratinocyte sheet in the management of chronic ulcers in diabetic patients. Cultured allogeneic keratinocytes on a hyaluronic acid scaffold have recently been demonstrated to be effective in the treatment of chronic ulcers, but specific studies on a diabetic group have not yet been described.[6] We reviewed sixteen chronic ulcers in diabetic patients treated with these novel epidermal substitutes, and discussed the potential benefits, scientific evidence, and safety in the management of this complication. The review board of University of Milan approved this study.
Methods
From donor cadavers (brain-dead), without infectious microorganisms (hepatitis B virus, hepatitis C virus, human immunodeficiency virus, human T-lymphotropic virus, cytomegalovirus, and negative to the treponema pallidum hemagglutination test), autoimmune, genetic, or infective skin pathologies, a 2 cm × 2 cm biopsy is performed from a sample of glabrous skin. The transfer of the biopsy is performed with a temperature of 4 °C, in a sterile container with gentamicin and amphotericin B, to Niguarda Ca’ Granda Hospital, the Regional Tissue Bank of Lombardia, Italy. The Skin Bank qualifies, collects, processes, validates, cryopreserves, and distributes skin taken from donor subjects. The biopsy is then sterilized and placed in Dulbecco’s modified Eagle’s medium (DMEM) containing Dispase II (bacterial enzyme) for 18 h at 4 °C or 37 °C for about 4 h, in order to split the epidermis from the dermis. The biopsy is then treated with Trypsin-Ethylenediaminetetraacetic acid (EDTA) in DMEM to isolate keratinocytes. The isolated cells can be cultivated in a specific culture medium.
HYAFF11 (Fidia Advanced Biopolymer S.r.l., Abano Terme, Italy) is the biomaterial, composed of hyaluronic acid totally esterified with benzyl ester. Due to its chemical properties, HYAFF11 releases benzyl alcohol and hyaluronic acid to the wound microenvironment because of hydrolysis of the ester bonds caused by the water in the wound exudate. The scaffold is a porous structure, composed of macropores and micropores, constituting the geometrical mesh of the matrix. Macropores (diameter of 0.5 mm) allow cellular distribution on the scaffold and the drainage of exudate when the sheet is put on the wound bed. Micropores (diameter of 40 µm, 6250 poers/cm2) allow neovascularization of the construct after the application, and cellular migration from the superior to the inferior face of the sheet, that is, the face in contact with a wound bed.[7] HYAFF11 forms a two-dimensional matrix, 20-mm thick, for epidermal substitutes, and a three-dimensional scaffold for dermal constructs.
Skin substitute preparation is divided into two phases: the first phase consists of the primary culture of allogeneic cells in a specific culture medium, and the second consists of the seeding of cells on a hyaluronic acid scaffold for cell expansion with adhesion. The sheets arethen ready for application. Keratinocytes are cultivated through Rheinwald and Green’s described method.[8] Cells are incubated with a specific medium at 37 °C for 20-30 min to inactivate Trypsin-EDTA, and then centrifugated and suspended in a medium without epidermal growth factor (EGF). The cellular pellet is located on a feeder layer of mouse-derived fibroblasts (3t3) inactivated by irradiation, and incubated at 37 °C with 5% CO2. The feeder layer secretes proteins of extracellular matrix and growth factors, promoting adhesion, and proliferation of keratinocytes. Irradiation of 3t3 serves blocks the replication of these cells. EGF is added to the culture medium 72 h following seeding on the feeder layers. The medium is changed every 2 days until semi-confluence is achieved. The sheets can be used fresh, within 21 days of production, or can be cryopreserved in dimethyl sulfoxide and stored at -80 °C, thus guaranteeing viability for 2 years.
Patient anamnestic data and outcomes were reviewed through a case series study of 11 patients with well-controlled diabetes type 2, with 16 legs and ankle chronic ulcers, unresponsive to previous conventional therapies (i.e. repeated use of advanced modern dressing for many cycles with a mean duration of 18 months), treated with the new skin substitutes from 2011.
All patients followed the same surgical protocol: chronic wounds incurable with other reconstructive options, such as wound dressings, acellular skin substitutes, and split-thickness autografts, underwent the application of the novel allogeneic skin substitutes in the operating room by the same surgical team. Before the application of sheets, all the wounds were debrided surgically to achieve wound bed preparation, and accurate hemostasis was performed. The skin substitutes were applied once directly to the wound bed without sutures.
Patients were observed weekly for a follow-up period of at least 40-70 days. The follow-up was performed by the physician team at the outpatient wound healing clinic of the Wound Care Unit (Monza, Italy). The wound dressing was the same for the postoperative period and for every control visits: a multicomponent bandage with nonadherent gauze, and polyurethane foam with silver (Biatain Ag, Coloplast) to prevent bacterial superinfection.
At the entry of the study, anamnestic data collected included age, sex, smoking status, and the presence of hypertension, end-stage renal disease, vascular diseases, autoimmune disorders, neurologic or cardiologic problems, or burns. Every chronic wound was classified at the entry of the study for dimension, location, the presence of local infection (absent, mild, moderate, severe), and wound characteristics such as wound bed, perilesional skin disorders, borders, and exudate, to calculate the wound bed score (WBS), as defined by Falanga et al.[9] Diagnosis of infection was based on culture obtained with a sterile rongeur. The results of this culture guided the appropriate use of systemic antibiotics. Multiple ulcers were considered different chronic wounds.
At the end of follow-up dimensional reduction, the WBS and presence of the adverse reaction were recorded.
The primary endpoint of the study was to determine the variation in ulcer dimension versus t0. Secondary endpoints included the evaluation of variation in the wound bed and exudate to determine the WBS, the percentage of wound reduction, and the percentage and time to healing.
Abstracted data were stored using an Excel Office database (Microsoft Corporation, Washington, USA) containing fields for clinical data entry. The statistical analysis was performed considering the patient as a unit of analysis initially (for anamnestic data), and then the single-chronic wound (for clinical results). The mean reduction of the skin lesion during follow-up was verified with the Wilcoxon Signed-Rank Test. The variation of WBS versus t0 was analyzed with the Friedman test. The level of statistical significance was fixed to 5% (P < 0.05) to reject the null hypothesis.
Results
Between January of 2011 and December of 2013, 11 patients with diabetes type 2 with 16 wounds underwent an application of allogeneic epidermal substitutes on a hyaluronic acid scaffold. Table 1 describes the demographics of all patients. Four out of 11 (36%) patients were females, and the mean age of all patients was 75 ± 8.2 years. Among comorbidities, hypertension (7, 63%) and cardiopathy (3, 27%) were the most frequent.
Anamnestic data, length of previous treatments and HbA1c levels
| Patient | Age (years) | Sex | Comorbidity | Length of previous treatments (months) | HbA1c (mg/dL) at t0 | HbA1c (mg/dL) at end |
|---|---|---|---|---|---|---|
| 1 | 75 | Male | Hypertension | 14 | 6.3 | 6.5 |
| 2 | 73 | Male | None | 22 | 6.6 | 6.4 |
| 3 | 69 | Female | Hypertension | 15 | 6.2 | 6.3 |
| 4 | 65 | Male | None | 17 | 5.9 | 6.2 |
| 5 | 70 | Male | Hypertension, cardiomyopathy | 24 | 6.7 | 6.1 |
| 6 | 68 | Female | Hypertension, autoimmune diseases | 19 | 6.5 | 6.2 |
| 7 | 83 | Male | Hypertension, cardiomyopathy, end-stage renal disease | 24 | 6.6 | 6.5 |
| 8 | 90 | Female | Hypertension | 13 | 5.8 | 6.0 |
| 9 | 81 | Male | Hypertension, cardiomyopathy | 18 | 5.7 | 5.9 |
| 10 | 66 | Female | Vascular disease | 28 | 6.4 | 6.1 |
| 11 | 82 | Female | Autoimmune disease | 32 | 6.0 | 5.7 |
As described by Table 2, 8 out of 16 (50%) chronic ulcers were located on the ankle or foot, and the other 50% were located on the lower third of the leg. Five (32%) ulcers showed a mild to moderate local infection at the entry of the study; only 1 severe infection was present. The mean preoperative wound size for all diabetic chronic ulcers was 14.37 ± 9.29 cm2, and the mean preoperative WBS was 9.6 ± 3.5. During the follow-up period, no wound advanced to require amputation of the foot or lower limb, and only one ulcer developed a local severe infection under the application of the sheet, enlarging the wound dimensions. No other adverse reactions were recorded.
Clinical result
| Patient | Ulcer | Location | Wagner ulcer classification | Infection* t0 | Infection end | Dimension t0 | Dimension end | WBS t0 | WBS end | Percentage wound reduction | Time healing (days) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | #1 | Leg | 1 | Absent | Absent | 12.6 | 0 | 6 | 16 | 100 | 40 |
| 2 | #2 | Leg | 1 | Mild (S. epidermidis) | Sev (P. aeruginosa) | 33.82 | 34.09 | 7 | 16 | −1 | - |
| 3 | #3 | Ankle | 2 | Absent | Absent | 11.31 | 0.8 | 10 | 16 | 93 | - |
| #4 | Leg | 1 | Absent | Absent | 28.91 | 15.01 | 9 | 16 | 48 | ||
| #5 | Leg | 1 | Absent | Absent | 6.03 | 2.44 | 9 | 16 | 60 | ||
| 4 | #6 | Ankle | 2 | Mild (S. aureus) | Absent | 21.32 | 19.27 | 8 | 10 | 10 | - |
| #7 | Ankle | 1 | Absent | Absent | 22.68 | 1.78 | 7 | 16 | 92 | ||
| 5 | #8 | Ankle | 1 | Severe (E. coli) | Mild (E. coli) | 15.68 | 8.41 | 3 | 5 | 46 | - |
| 6 | #9 | Leg | 1 | Absent | Absent | 3 | 0 | 12 | 16 | 100 | 21 |
| 7 | #10 | Ankle | 1 | Moderate (P. aeruginosa) | Absent | 19.65 | 1.35 | 9 | 12 | 93 | - |
| #11 | Leg | 1 | Moderate (S. epidermidis) | Absent | 22.7 | 6.93 | 8 | 15 | 69 | ||
| 8 | #12 | Ankle | 1 | Mild (P. aeruginosa) | Absent | 8.55 | 2.28 | 16 | 16 | 73 | |
| 9 | #13 | Leg | 1 | Absent | Absent | 4.85 | 0 | 14 | 16 | 100 | 40 |
| 10 | #14 | Ankle | 1 | Absent | Absent | 11.78 | 0 | 9 | 16 | 100 | 70 |
| #15 | Ankle | 1 | Absent | Absent | 3.08 | 0 | 16 | 16 | 100 | 40 | |
| 11 | #16 | Leg | 1 | Absent | Absent | 4.07 | 0 | 11 | 16 | 100 | 45 |
| Mean | 14.37 | 5.77 | 9.62 | 14.62 | 70 | 42.66 | |||||
| Ds | 9.29 | 9.52 | 3.51 | 3.09 | 32 | 15.75 | |||||
| Maximum | 33.82 | 34.09 | 16 | 16 | 100 | 70 | |||||
| Minimum | 3 | 0 | 3 | 5 | −1 | 21 | |||||
| P value | 0.0007 | <0.0001 |
Six out of 16 (38%) chronic ulcers healed in a mean time of 42 ± 16 days. The application of the new skin substitutes reduced the mean percentage of the wound dimension by 70% (P = 0.0007). The WBS demonstrated an improvement of 52% (P < 0.0001) at the end of the follow-up period from the score recorded at the entry of the study.
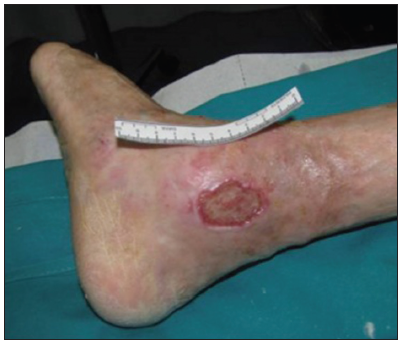
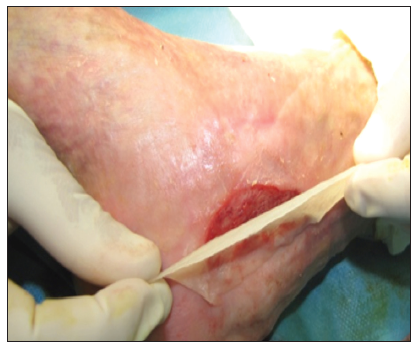
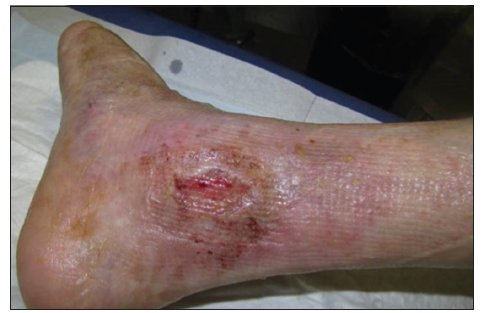
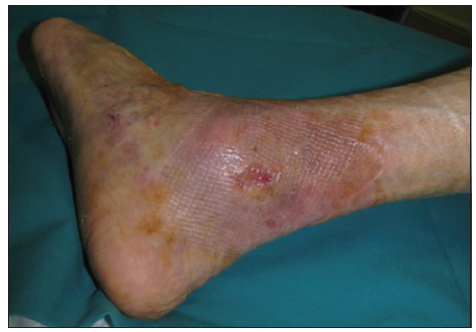
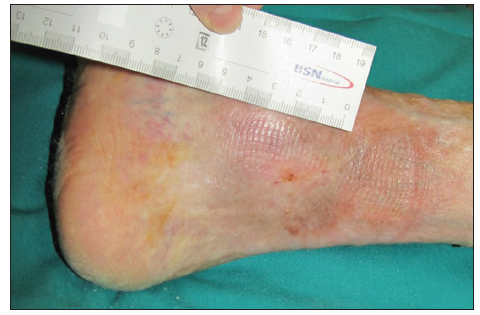
Case example: ankle diabetic ulcers in a 66-year-old patient treated with allogeneic keratinocyte [Figures 1–5].
Discussion
The chronic wound microenvironment is biologically distinct from the acute wound milieu: venous and diabetic chronic ulcers are hypothesized to be trapped in the inflammatory and proliferative phases of normal healing, respectively. Poor wound healing may be a consequence of abnormal insulin signaling and hyperglycemia, affecting skin proliferation and differentiation.[10] Skin biopsies performed in nondiabetic and diabetic subjects from the edges of chronic wounds have revealed increased expression of transforming growth factor (TGF) beta 3 and low expression of TGF-beta 1, resulting in nonhealing.[11] Abnormal expression of insulin-like growth factor type 1 in diabetic skin may also contribute to delayed wound healing.[12]
The damaged biological background of diabetic wounds explains the necessity to modulate therapeutically the unbalanced levels of growth factors, signaling molecules, and extracellular matrix proteins.[13] How these novel skin substitutes work is still not completely understood. Initially, especially with skin substitutes as cultured epidermis and living bilayered skin construct, some degree of permanent engraftment was thought to assist in healing. As shown by DNA and Y chromosome probes, allogeneic constructs are not the same as autografts: a true take of allogeneic sheets has not been demonstrated.[14] The allogeneic skin constructs usually do not stay on the wound for more than a few weeks, and their function is not to replace tissues or cells, but instead to stimulate tissue repair as pharmacologic agents, secreting healing factors in chronic wound microenvironment. The key role of allogeneic constructs seems to be the secretion of growth factors and extracellular matrix proteins, and to attract differentiated or stem cells in the wound milieu.[15]
This clinical case-series study based on the utilization of new cultured allogeneic keratinocyte sheets showed promising results, including safety and tolerability of the allogeneic product, good wound healing rate, a great reduction in wound size in a relatively short period, and preparation of the wound bed for alternative reconstructive treatments (i.e. split-thickness autograft). The application of allogeneic keratinocytes on a hyaluronic acid scaffold may allow improvement of diabetic leg and foot lesions not amenable to other therapies or surgical indications, thereby allowing the closure of nonhealing chronic ulcers, and thus reducing morbidity, cost, and length of hospitaliazation. Allogeneic skin substitutes do not require prolonged operating time or skin biopsy, and are easily applied by the surgeon in contrast to flaps or autograft. In the multidisciplinary approach to diabetic chronic wounds, allogeneic skin substitutes on a hyaluronic acid scaffold may represent a valid alternative when other possibilities have been exhausted. Keratinocyte sheets were also applied when mild to moderate local infection was present, resulting in interesting clinical outcomes. Cultured keratinocytes were, in fact, resistant to bacterial colonization in excised burns and chronic ulcers.[16] In such settings and considering the cost of this new product, allogeneic keratinocytes on a hyaluronic acid scaffold could be considered a second-line treatment in case of prior treatment failure.
Fortunately, we had no cases of immunologic response to these allogeneic products in our case series. Had this occurred, the application of a topical immunologic suppressant like 5-fluorouracil ointment would have been recommended.
In the literature, other studies have reported clinical outcomes for cellular skin substitutes on various other scaffolds in the treatment of chronic leg and foot diabetic ulcers.[17-20] The differences among these studies in results, methods, products, cost-effectiveness ratio, and follow-up period are highlighted in Table 3. However, comparison of the effectiveness is difficult to perform given the extreme variation in protocols (e.g. skin substitutes were used multiple times on the same ulcer in some studies, or the end of the study was not fixed until 100% wound healing was achieved).[21-23] However, some clinical features have emerged from these studies regarding the use of cellular skin substitutes in the management of chronic diabetic ulcers. The role of allogeneic keratinocytes appears to be central in the cellular therapy of diabetic wounds, although good results have been reported with the use of autologous cells.[24,25] Unlike allogeneic substitutes, autologous sheets are not available for use immediately, a skin biopsy is required, and longer times are necessary for cell processing, with the ever-present risk of ischemia or osteomyelitis. Cellular skin substitutes are formed by two elements: cells and scaffold. In the "dynamic reciprocity" model, the extracellular matrix emerges as capable of influencing wound healing, acting on the others two characters, cells and signal factors.[26] Thus, the scaffold and cells are both fundamental in the clinical outcome of skin substitutes. Hyaluronic acid is a central molecule in human skin, and its functions are diverse. Hyaluronan influences hydration of the extracellular matrix, due to its hydroscopic characteristics, and contributes to the physical and mechanical properties of the dermis. Hyaluronic acid interacts with a number of receptors, resulting in the activation of signaling cascades that influence cell migration, proliferation, and gene expression. Further, fetal-like regenerative wound healing is characterized by a large amount of hyaluronic acid deposition. From these observations, a membrane composed of completely esterified hyaluronic acid was developed, and was shown to support growth of keratinocytes in vitro and biocompatibility in vivo.[27] Prior studies on cellular therapy for diabetic wounds have emphasized repeated debridement, control of bacterial growth, careful moisture balance to prevent maceration, blood pressure control, management of blood glucose, and perfusion of the extremity. Wound bed preparation remains central for cellular skin substitutes application and efficacy.
Cellular skin substitutes for diabetic chronic ulcers (costs at the time of writing should be treated as a guide and have been converted into € currency where necessary; the name of the scaffold is present if the product has no commercial name)
| Study | Cells | Scaffold | Product | Sheet | Cost € | Follow-up (weeks) | n cases | Outcome % | Time healing (days) |
|---|---|---|---|---|---|---|---|---|---|
| Vamos et al.[2] | Neonatal allogeneic keratinocytes | Collagen seeded with neonatal allogeneic fibroblasts | Apligraf | 7.5 cm diameter disc | 763.26 | 12 | 82 | 51.5 healing | 84 |
| Veves et al.[18] | Neonatal allogeneic keratinocytes | Collagen seeded with neonatal allogeneic fibroblasts | Apligraf | 7.5 cm diameter disc | 763.26 | 12 | 112 | 56 healing | 65 |
| Harvima et al.[19] | Allogeneic keratinocytes | Nylon | Tegapore | Not commercially available | Not commercially available | 11 | 18 | 100 wound reduction | 64 |
| Bayram et al.[20] | Allogeneic keratinocytes | Polyethylene and silica | Cytoline 1 | Not commercially available | Not commercially available | 4 | 20 | 92 wound reduction | 30 |
| Sams et al.[21] | Neonatal allogeneic keratinocytes | Collagen seeded with neonatal allogeneic fibroblasts | Apligraf | 7.5 cm diameter disc | 763.26 | 24 | 9 | 56 healing | 84 |
| Marston et al.[22] | Neonatal allogeneic fibroblasts | Polyglycolic acid (Dexon™) or polyglactin-910 (Vicryil™) | Dermagraft | 5 cm × 7.5 cm | 326.52 | 12 | 130 | 30 healing | 84 |
| Caravaggi et al.[23] | Autologous fibroblasts | Benzyl ester of hyaluronic acid | Hyalograft-3D | 8 cm × 8 cm | 1200.00 | 11 | 43 | 65.3 healing | 57 |
| You et al.[24] | Allogeneic keratinocytes | - | Kaloderm | Available sizes: 9-16. 25 and 56 cm2 | Not yet licensed | 12 | 27 | 100 wound reduction | 35 |
| Lobmann et al.[25] | Autologous keratinocytes | Benzyl ester of hyaluronic acid | Laserskin | 10 cm × 10 cm | 1200.00 | 12 | 14 | 78 healing | 41 |
| Vaienti et al.[7] | Allogeneic keratinocytes | Benzyl ester of hyaluronic acid | HYAFF11 | 10 cm × 10 cm | 1000.00 | 11 | 16 | 70 wound reduction | 42 |
The present case series study on skin substitutes based on hyaluronic acid scaffold for the therapy of chronic diabetic leg and foot ulcers allows investigation of the clinical results, in order to find evidence for treatment perspectives, and stimulate biochemical research in the field of regenerative medicine. Comprehensive studies will be necessary to evaluate the cost-effectiveness of these therapies before they become acceptable for general use. The goal is to achieve the cellular therapy that "suits" the specific chronic wound microenvironment in diabetic wounds in the future. However, large, randomized, and controlled clinical trials are required to confirm and validate the clinical results of these novel skin substitutes.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Margolis DJ, Hoffstad O, Nafash J, Leonard CE, Freeman CP, Hennessy S, Wiebe DJ. Location, location, location: geographic clustering of lower-extremity amputation among Medicare beneficiaries with diabetes. Diabetes Care 2011;34:2363-7.
2. Vamos EP, Bottle A, Edmonds ME, Valabhji J, Majeed A, Millett C. Changes in the incidence of lower extremity amputations in individuals with and without diabetes in England between 2004 and 2008. Diabetes Care 2010;33:2592-7.
3. Game FL, Hinchliffe RJ, Apelqvist J, Armstrong DG, Bakker K, Hartemann A, Löndahl M, Price PE, Jeffcoate WJ; International Working Group on Diabetic Foot. Specific guidelines on wound and wound-bed management 2011. Diabetes Metab Res Rev 2012;28 Suppl 1:232-3.
4. Bakker K, Apelqvist J, Schaper NC; International Working Group on Diabetic Foot Editorial Board. Practical guidelines on the management and prevention of the diabetic foot 2011. Diabetes Metab Res Rev 2012;28 Suppl 1:225-31.
5. Iorio ML, Goldstein J, Adams M, Steinberg J, Attinger C. Functional limb salvage in the diabetic patient: the use of a collagen bilayer matrix and risk factors for amputation. Plast Reconstr Surg 2011;127:260-7.
6. Pajardi G, Rapisarda V, Somalvico F, Scotti A, Russo GL, Ciancio F, Sgrò A, Nebuloni M, Allevi R, Torre ML, Trabucchi E, Marazzi M. Skin substitutes based on allogenic fibroblasts or keratinocytes for chronic wounds not responding to conventional therapy: a retrospective observational study. Int Wound J 2014; doi: 10.1111/iwj.12223.
7. Vaienti L, Marchesi A, Palitta G, Gazzola R, Parodi PC, Leone F. Limb trauma: the use of an advanced wound care device in the treatment of full-thickness wounds. Strategies Trauma Limb Reconstr 2013;8:111-5.
8. Rheinwald JG, Green H. Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 1975;6:331-43.
9. Falanga V, Saap LJ, Ozonoff A. Wound bed score and its correlation with healing of chronic wounds. Dermatol Ther 2006;19:383-90.
10. Spravchikov N, Sizyakov G, Gartsbein M, Accili D, Tennenbaum T, Wertheimer E. Glucose effects on skin keratinocytes: implications for diabetes skin complications. Diabetes 2001;50:1627-35.
11. Akingboye AA, Giddins S, Gamston P, Tucker A, Navsaria H, Kyriakides C. Application of autologous derived-platelet rich plasma gel in the treatment of chronic wound ulcer: diabetic foot ulcer. J Extra Corpor Technol 2010;42:20-9.
12. Aghdam SY, Eming SA, Willenborg S, Neuhaus B, Niessen CM, Partridge L, Krieg T, Bruning JC. Vascular endothelial insulin/GF-1 signaling controls skin wound vascularization. Biochem Biophys Res Commun 2012;421:197-202.
13. Shevchenko RV, James SL, James SE. A review of tissue-engineered skin bioconstructs available for skin reconstruction. J R Soc Interface 2010;7:229-58.
14. Brain A, Purkis P, Coates P, Hackett M, Navsaria H, Leigh I. Survival of cultured allogeneic keratinocytes transplanted to deep dermal bed assessed with probe specific for Y chromosome. BMJ 1989;298:917-9.
15. Lazic T, Falanga V. Bioengineered skin constructs and their use in wound healing. Plast Reconstr Surg 2011;127 Suppl 1:75S-90S.
16. Maier K, Ehrhardt G, Frevert J. Antibacterial activity of cultured human keratinocytes. Arch Dermatol Res 1992;284:119-21.
17. Kirsner RS, Warriner R, Michela M, Stasik L, Freeman K. Advanced biological therapies for diabetic foot ulcers. Arch Dermatol 2010;146:857-62.
18. Veves A, Falanga V, Armstrong DG, Sabolinski ML, Apligraf Diabetic Foot Ulcer Study. Graftskin, a human skin equivalent, is effective in the management of noninfected neuropathic diabetic foot ulcers: a prospective randomized multicenter clinical trial. Diabetes Care 2001;24:290-5.
19. Harvima IT, Virnes S, Kauppinen L, Huttunen M, Kivinen P, Niskanen L, Horsmanheimo M. Cultured allogeneic skin cells are effective in the treatment of chronic diabetic leg and foot ulcers. Acta Derm Venereol 1999;79:217-20.
20. Bayram Y, Deveci M, Imirzalioglu N, Soysal Y, Sengezer M. The cell based dressing with living allogenic keratinocytes in the treatment of foot ulcers: a case study. Br J Plast Surg 2005;58:988-96.
21. Sams HH, Chen J, King LE. Graftskin treatment of difficult to heal diabetic foot ulcers: one center's experience. Dermatol Surg 2002;28:698-703.
22. Marston WA, Hanft J, Norwood P, Pollak R, Dermagraft Diabetic Foot Ulcer Study Group. The efficacy and safety of Dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care 2003;26:1701-5.
23. Caravaggi C, De Giglio R, Pritelli C, Sommaria M, Dalla Noce S, Faglia E, Mantero M, Clerici G, Fratino P, Dalla Paola L, Mariani G, Mingardi R, Morabito A. HYAFF 11-based autologous dermal and epidermal grafts in the treatment of noninfected diabetic plantar and dorsal foot ulcers: a prospective, multicenter, controlled, randomized clinical trial. Diabetes Care 2003;26:2853-9.
24. You HJ, Han SK, Lee JW, Chang H. Treatment of diabetic foot ulcers using cultured allogeneic keratinocytes – a pilot study. Wound Repair Regen 2012;20:491-9.
25. Lobmann R, Pittasch D, Mühlen I, Lehnert H. Autologous human keratinocytes cultured on membranes composed of benzyl ester of hyaluronic acid for grafting in nonhealing diabetic foot lesions: a pilot study. J Diabetes Complications 2003;17:199-204.
26. Greaves NS, Iqbal SA, Baguneid M, Bayat A. The role of skin substitutes in the management of chronic cutaneous wounds. Wound Repair Regen 2013;21:194-210.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Marchesi A, Brioschi M, Parodi PC, Marchesi M, Brambilla R, Vaienti L. Allogeneic epidermal substitutes in the treatment of chronic diabetic leg and foot ulcers. Plast Aesthet Res 2014;1:74-80. http://dx.doi.org/10.4103/2347-9264.139706
AMA Style
Marchesi A, Brioschi M, Parodi PC, Marchesi M, Brambilla R, Vaienti L. Allogeneic epidermal substitutes in the treatment of chronic diabetic leg and foot ulcers. Plastic and Aesthetic Research. 2014; 1: 74-80. http://dx.doi.org/10.4103/2347-9264.139706
Chicago/Turabian Style
Marchesi, Andrea, Marco Brioschi, Pier Camillo Parodi, Matteo Marchesi, Roberto Brambilla, Luca Vaienti. 2014. "Allogeneic epidermal substitutes in the treatment of chronic diabetic leg and foot ulcers" Plastic and Aesthetic Research. 1: 74-80. http://dx.doi.org/10.4103/2347-9264.139706
ACS Style
Marchesi, A.; Brioschi M.; Parodi PC.; Marchesi M.; Brambilla R.; Vaienti L. Allogeneic epidermal substitutes in the treatment of chronic diabetic leg and foot ulcers. Plast. Aesthet. Res. 2014, 1, 74-80. http://dx.doi.org/10.4103/2347-9264.139706
About This Article
Copyright
Data & Comments
Data
 Cite This Article 2 clicks
Cite This Article 2 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.