Adipose-derived stem cells in cutaneous wound repair
Abstract
Growing interest in regenerative medicine and advances in adipose tissue research have led to the identification of mesenchymal stem cells in adipose tissue, so called adipose tissue-derived stem cells (ASCs). Due to the simple and safe harvest technique as well as high regenerative capacity, ASCs are regarded as a potential source for various indications including cutaneous wound repair. This review provides a short overview over mechanisms of ASC action in cutaneous wound repair and data regarding its clinical application. Mostly experimental data provide accruing evidence for the supportive effect of ASCs in cutaneous wound healing by secretion of soluble factors, differentiation into keratinocyte and fibroblasts, neovascularization and interaction with myofibroblasts. A number of in vivo experiments also support a positive effect of ASCs in different wound healing models. Furthermore, first clinical data evaluated the feasibility of ASCs in the treatment of different wound healing pathologies, e.g., chronic ulcers and burn wounds. Although the majority of currently available data indicate a beneficial role of ASCs in cutaneous wound repair, additional detailed experimental studies and larger, high-quality clinical trials are required to provide a reliable statement on the true value of ASCs in this context.
Keywords
Fat grafting and adipose-derived stem cells
Ever since the German surgeon Gustav Neuber documented the first autologous fat transfer at the 23rd Congress of the German Surgical Society in 1893 by treating an infrorbital scar by a piece of autologous fat harvested from the upper arm, the interest in adipose tissue as a reservoir for graft material rose[1]. Czerny used a lipoma for autologous breast augmentation in 1895 and documented further cases of autologous fat transfer[2,3]. Case reports by Czerny, Bartlett, Lexer, Passot, Gurney, Peer, May, Miller and more were soon backed by a scientific evaluation of fat grafting with investigators examining the high rate of fat resorption. Marchand performed a histological analysis on transplanted fat and found resorption and necrosis while Neuhof hypothesized that transplanted fat tissue may necrotize and eventually be replaced by a mix of fibrous and adipose tissue[4,5].
Contrary to the general perception that adipose tissue primarily acts as an energy storage with other rather passive functions such as mechanical padding and thermal insulation, investigators revealed its complex structure and its diverse cell composition[6]. The liposuction technique, invented by the Italian gynecologists Arpad and Giorgio Fischer, was soon popularized by Yves-Gerard Illouz[7]. While Sydney Colemans defined fat grafting protocol then established the era of lipofilling, it was the characterization of mesenchymal stem cells within adipose tissue that lay the foundation to adipose-derived stem cell (ASC) therapy[8,9]. ASC therapy is a modern approach in regenerative medicine that utilizes isolated ASCs for various indications including wound repair. In this review, the most fundamental characteristics of ASCs, and insights into their role in cutaneous wound repair based on recent experimental and clinical data will be presented.
Characteristics of ASCs
Enzymatic digestion of harvested fat tissue by collagenase separates adipose tissue into two fractions: a top layer of floating mature adipocytes and a bottom layer of cells, that are collectively called the stromal vascular fraction (SVF). The SVF represents a heterogenous cell mixture including adipose tissue macrophages, smooth muscle cells, T- and B-lymphocytes, endothelial cells, pericytes, myocytes, fibroblasts and importantly ASCs.
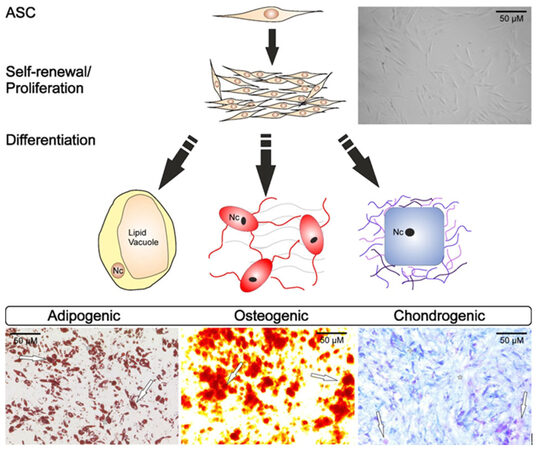
ASCs are mesenchymal stem cells of mesodermal origin with low oxygen consumption and a considerable proliferation rate that differentiate into mesodermal, ectodermal and entodermal cell lines[9][Figure 1]. In recent years, they have evolved as a serious alternative to bone marrow-derived stem cells (BMSC), the major source for mesenchymal stem cells so far[10]. In contrast to BMSCs, the harvest of ASCs is easier, and excitingly, the relative yield of ASCs is reported to be substantially higher when compared to BMSCs[11]. One milliliter of human lipoaspirate will yield approximately (2.5-3.75) × 105 ASCs within a timeframe of 4-6 days[12].
Figure 1. Proliferation and differentiation capacity of adipose tissue-derived stem cells (ASCs). ASCs display a fibroblast like phenotype. Their morphology is characterized by a small cell body, containing a large round nucleus, with long and thin extensions. ASC have have high proliferation and self-renewal potential in vitro. Upon induction by specific medium, ASCs are able to differentiate into different cell lines including the adipogenic, chondrogenic and osteogenic line. According to their cell fate, ASC change their morphology by activation of certain molecular processes during the differentiation processes. This may lead to increased fat synthesis resulting in the generation of lipid droplets (white arrows; adipogenic differentiation is indicated by red droplets upon Oil-Red O staining), which will increase during adipogenesis (white asterisks), and finally merge into one large fat vacuole. During osteogenesis, ASCs secrete a collagen I-rich extracellular matrix that calcifies during the later stages of differentiation. One indicator of osteogenesis is the formation of calcification appearing red after Alizarin red staining (arrow). Chondrogenic differentiation leads to the generation of cell nodules associated with a well-organized extracellular matrix rich in collagen II and sulfated proteoglycans. These proteoglycans can be specifically detected using the stain Alcian Blue under acidic conditions (white asterisk; cross marks nuclei; arrows denote acidic mucosubstances)
The increasing interest in ASC and its applications have led to a non-transparent nomenclature that was addressed by the International Federation for Adipose Therapeutics and Science and the International Society for Cellular Therapy (ISCT). As any other mesenchymal stem cell population, ASCs have to meet the minimal criteria defined by the ISCT which encompass: plastic adherence, differentiation into the adipogenic, chondrogenic, and osteogenic line as well as expression of specific surface genes[13]. In addition, ASCs may be distinguished from SVF cells and BMSCs by other distinct surface markers. ASCs consistently express cluster of differentiation (CD) 73, CD90, CD105, CD44 and are negative for the endothelial markers CD31 and hematopoietic cell marker CD45; the population doubling capacity should be determined by the colony forming unit fibroblast (CFU-F) assay[14].
Isolation of ASCs
The regenerative potential of ASCs has rapidly attracted worldwide attention as their isolation and utilization are easy and do not underlie the same legislative and ethical concerns as applicable to pluripotent embryonic stem cells. In the first step, fat tissue is commonly harvested by lipectomy or liposuction procedures. Although ASCs are not as vulnerable to high negative pressure as mature adipocytes (which tend to rupture), higher suction pressure may significantly decrease ASC yield[15]. Next, the aspirated adipose tissue may undergo washing steps, e.g., with phosphate-buffered saline and collagenase digestion followed by collagenase inactivation, further filtering and washing steps. The resulting SVF, by many authors commonly but inadequately and misleadingly referred to as the ASCs, is expanded in culture.
Commercial suppliers such as Celution® (Cytori Therapeutics, San Diego, CA, USA) and many others offer manual, semi- and fully-automated systems for alternative progenitor cell/cell fraction isolation/enrichment[16]. The cell fractions isolated by pre-manufactured systems generally follow proprietary protocols and are immediately re-injected in the same procedure. As these protocols do not include an in vitro cell cultivation step and it is unclear whether these cell fractions do meet the ISCT minimal criteria, we do not recommend labelling cell fractions isolated by such as pure ASCs.
Mechanisms of action - how do ASCs improve cutaneous wound repair
Cutaneous wound healing comprises four stages termed as the hemostasis, inflammatory, proliferative and remodeling phase[17]. Upon reduction of blood loss by vascular constriction, platelet aggregation and fibrin clot formation (hemostasis phase), the inflammatory phase, characterized by invasion of cells such as neutrophils/monocytes, immunomodulation by release of pro-inflammatory and chemotactic cytokines, sets in. The proliferative phase covers neoangiogenesis, re-epithelialization and reorganization of the underlying dermal layers. During this phase, invading fibroblasts in particular secrete extracellular matrix (ECM) proteins, enzymes and various cytokines that jointly facilitate dermis restoration. Finally, the fibroblasts adopt a contractile myofibroblast phenotype during the remodeling phase which inter alia replaces immature collagen III into contractile collagen I fibres forming the ultimate scar.
ASCs are a natural component of the subcutaneous adipose tissue that lies in immediate proximity to cutaneous wounds and takes part in the delicate physiological course of wound healing. Due to their migratory ability ASCs also are believed to infiltrate the wound and thereby additionally foster wound repair[18].
First of all, the abundant secretome of ASCs may orchestrate wound healing in a paracrine fashion. Nearly all growth factors that participate in cutaneous wound healing including keratinocyte growth factor, hepatocyte growth factor, epidermal growth factor, members of the vascular endothelial growth factor (VEGF) family, basic fibroblast growth factor (bFGF), platelet-derived growth factor-BB, insulin-like growth factor-1 and key enzymes such as matrix metallothioneine-9 (MMP-9) and many more are secreted by ASCs[19-21]. Without going into detail, the mentioned soluble factors instigate fibroblast and keratinocyte migration, proliferation and differentiation. The literature also suggests ASCs to attenuate inflammation by secretion of soluble factors such as interleukin-10 and other cytokines implicated in leukocyte action[22]. Thereby, ASCs also may exert beneficial effects on cutaneous diseases or wounds such as non-healing ulcers that involve a pathologically pro-longed inflammatory state.
In addition to their remarkable secretome, ASCs are known to directly differentiate into keratinocytes and fibroblasts to regenerate epidermal and dermal layers that further promote cuntaneous wound repair in vitro[23,24]. However, until the afore-mentioned experimental in vitro studies are supported by further investigation in a proper in vivo model, there is no firm evidence for a physiological differentiation of ASCs into keratinocytes that would allow a translation into the clinical context. ASCs also appear to have an impact on fibroblasts although the evidence is scarce. In vitro studies indicated an increase in fibroblast proliferation by direct cell-to-cell contact with ASCs but also by soluble factors released by ASCs[25]. The authors observed up-regulation of fibronectin, collagen I, collagen III as well as a down-regulation of MMP-1. The same authors supported their in vitro findings by additional in vivo experiments, where they found accelerated wound healing in mice by ASC treatment. However, those in vivo observations were of pure descriptive nature and did not look into the molecular or cellular effect (e.g., ASC-fibroblast interaction) in more detail.
Proper vascularization is of paramount importance to wound healing. Mounting evidence portrays the pro-angiogenic effect of ASCs, predominantly by release of prominent vasculogenic factors such as VEGF-A and bFGF, and differentiation into endothelial cells[26].
Eventually, through differentiation into myofibroblasts and paracrine modulation of myofibroblast function ASCs dynamically influence wound maturation during the remodeling phase[27].
Experimental data on the effect of ASCs in cutaneous wound repair
Particularly in non-healing wounds that experience a depletion of ASCs/impairment of ASC function, the transplantation of “fresh” ASCs from distant donor sites by fat grafting or stem cell therapy appears to be invaluable[28]. But also physiological cutaneous wounds appear to benefit from ASC treatment.
In vitro experiments with simple co-cultures or ASC-derived supernatants have shown that human lipoaspirates and ASCs enhance keratinocyte proliferation, stratification and migration[29,30]. Comparable experimental settings show enhanced fibroblast proliferation with beneficial effects on collagen synthesis and ECM remodeling[31].
Plenty of in vivo studies investigated the role of ASCs in either normal or pathological wound healing in more detail. In normal wound healing, where ASCs were transferred into wounds by local injection, seeded on scaffolds (fibrin, collagen, acellular dermal matrix, etc.), as cell sheets or even systemically, the vast majority suggests a beneficial ASC effect on wound healing[21]. However, due to the limited clinical relevance of ASC treatment in physiologically healing wounds and considering the increase of patients with wounds not responding to conventional treatment paradigms, animal models focusing on pathological wound healing attract more attention.
As type 2 diabetes is undoubtedly the greatest trigger for delayed wound repair, it also covers the largest body of evidence. Maharlooei et al.[32] for instance locally injected excisional wounds in diabetic rats with ASCs and found wound healing rates almost comparable to those of normal rats. Similar observations were made in many other diabetic mice and rat models that saw ASC-induced keratinocyte and fibroblast regeneration as well as improved wound vascularization. Among these, the study of Nambu et al.[33] specifically used ASCs isolated from diabetic mice whereas all other authors used ASCs from either healthy human patients or healthy animals. Importantly, the authors found that even ASCs from diabetic mice significantly accelerated healing of excisional wounds. In the same way, ASCs appear to promote wound healing under ischemia. Steinberg et al.[34] reported enhanced wound granulation in rabbit ears under ischemic conditions (wounding after ligating two of three main arteries of the ear) upon ASC treatment and comparable observations were made in other animal models. Chronic wounds are frequently found in irradiated area and thus are subject to intense investigation. ASCs administered directly into the irradiated wound, intramuscularly e.g., into the irradiated limbs or even intravenously led to markedly increased cutaneous wound repair in rats and mice, respectively[35-37].
Taken together, existing experimental data mostly support the positive effect of ASCs on cutaneous wound repair while there is no consensus on the exact protocol of ASC transfer to the wound side.
Clinical application of ASCs in cutaneous wound repair
For cutaneous wound repair in the clinical setting, ASCs may be transplanted to the wound by different ways: fat grafts, fat grafts enriched by cell fractions/SVF/ASCs or pure ASCs via stem cell therapy. While fat grafts and enriched fat grafts surely contain ASCs to a certain extent, only the transplantation of purely in vitro expanded and characterized ASCs as mostly done in animal studies permits to draw definite conclusions of the genuine ASC effect on cutaneous wound repair. However, only few clinical studies follow exact ASC isolation protocols due to regulatory issues and use SVF/cell fraction enriched fat grafts instead in different critical wounds (diabetic foot ulcers, radiation injury, peripheral artery disease (PAD), venous leg ulcers and burn scars).
In 2010, Akita et al.[38] reported healing of radiation wounds without adverse effects by applying ASCs seeded on an artificial dermis (Terudermis®). At a closer look, however, ASCs were isolated by Celution® rather than according to precise ASC isolation protocols with an unclear number of patients. Bura et al.[39] performed a phase 1 study on the effect of in vitro expanded ASCs on ulcers due to critical limb ischemia. The authors found increased oxygen pressure in the affected limbs, improved ulcer healing with no adverse effects. Although the authors used proper ASC isolation protocols, the small number of merely seven patients with no control group did not allow a statistical evaluation of the results. In 2015, Liu et al.[40] summarized existing data on mesenchymal stem cells including ASCs on PAD in a total of 527 patients divided in 13 clinical studies. Stem cells were applied intramuscularly, intravenously or intraarterially. Although no difference in the all-cause mortality was found, a significant improvement of ulcer healing, amputation rate and the ankle-brachial-index were observed. In a review of the feasibility of ASCs in venous leg ulcers, Zollino et al.[41] concluded that ASCs induce new well-vascularized tissue formation at the transplanted site with only minor complications. Conde-Green et al.[42] reviewed twelve studies that addressed the clinical application fat grafts and fat grafts enriched by progenitor cells via various techniques in burn scars. Despite the fact that improvements in scar size/texture, enhancement of angiogenesesis, alleviation of inflammation/pain and improved function were observed, no clear and statistical significant conclusions could be made and ASC isolation protocols varied. On the contrary, Gal et al.[43] examined the use of fat grafts in pediatric burn scars in a prospective, randomized, double-blinded, placebo-controlled pilot study in eight patients and found no evidence for scar improvement. Of course, the number of patients in Meng et al.[44]’s study is low, but its scientifically sound study design, when compared to most other hitherto published clinical trials, adds to the strength of the work. Although several preclinical studies analyzed the value of ASCs in diabetic wounds, most of the clinical trials are still ongoing with no definite results provided by now.
In summary, most of the studies indicate a positive effect of ASCs/fat grafting/cell enriched fat grafts on various cutaneous wound conditions with no obvious complications, all authors of the afore-mentioned studies unanimously complain the lack of randomized high quality studies with a sufficient number of patients.
Conclusion
Accruing experimental data indicate that ASCs are conductive to normal and pathological cutaneous wound repair by release of soluble factors, differentiation into various cell lines and facilitating angiogenesis. However, due to a lack of high quality clinical studies, no conclusive statement is yet possible on the true benefits of ASCs in the clinical setting.
Declarations
Authors’ contributionsConception, design, wrote, revised, final manuscript version: Kim BS, Debye B, Beier JP
Performed literature review: Kim BS
Assisted in literature review: Debye B
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Neuber GA. Fettransplantation. Chir Kongr Verhandl Dtsch Gesellsch Chir 1893;22:66.
2. Czerny V. Plastischer Ersatz der Brustdruse durch ein Lipom. Chir Kong Verhandl 1895;2:216.
3. Ross RJ, Shayan R, Mutimer KL, Ashton MW. Autologous fat grafting: current state of the art and critical review. Ann Plast Surg 2014;73:352-7.
4. Marchand F. Ueber die Veranderungen des Fettgewebes nach der Transplantation. Beitr Pathol Anat Allg Pathol 1919;66:32.
5. Neuhof H. The Transplantation of Tissues. New York: D Appleton & Co; 1923.
6. Julien P, Despres JP, Angel A. Scanning electron microscopy of very small fat cells and mature fat cells in human obesity. J Lipid Res 1989;30:293-9.
7. Sterodimas A, Boriani F, Magarakis E, Nicaretta B, Pereira LH, Illouz YG. Thirtyfour years of liposuction: past, present and future. Eur Rev Med Pharmacol Sci 2012;16:393-406.
9. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 2001;7:211-28.
10. Salibian AA, Widgerow AD, Abrouk M, Evans GR. Stem cells in plastic surgery: a review of current clinical and translational applications. Arch Plast Surg 2013;40:666-75.
11. Liao HT, Chen CT. Osteogenic potential: comparison between bone marrow and adipose-derived mesenchymal stem cells. World J Stem Cells 2014;6:288-95.
12. Gimble JM, Bunnell BA, Frazier T, Rowan B, Shah F, Thomas-Porch C, Wu X. Adipose-derived stromal/stem cells: a primer. Organogenesis 2013;9:3-10.
13. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006;8:315-7.
14. Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 2013;15:641-8.
15. Mojallal A, Auxenfans C, Lequeux C, Braye F, Damour O. Influence of negative pressure when harvesting adipose tissue on cell yield of the stromal-vascular fraction. Biomed Mater Eng 2008;18:193-7.
16. Lin K, Matsubara Y, Masuda Y, Togashi K, Ohno T, Tamura T, Toyoshima Y, Sugimachi K, Toyoda M, Marc H, Douglas A. Characterization of adipose tissue-derived cells isolated with the Celution system. Cytotherapy 2008;10:417-26.
18. Aboutaleb Kadkhodaeian H, Tiraihi T, Ahmadieh H, Ziaei Ardakani H, Daftarian N, Taheri T. Survival and migration of adipose-derived stem cells transplanted in the injured retina. Exp Clin Transplant 2018;16:204-11.
19. Pallua N, Serin M, Wolter TP. Characterisation of angiogenetic growth factor production in adipose tissue-derived mesenchymal cells. J Plast Surg Hand Surg 2014;48:412-6.
20. Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm-Grove CJ, Bovenkerk JE, Pell CL, Johnstone BH, Considine RV, March KL. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004;109:1292-8.
21. Toyserkani NM, Christensen ML, Sheikh SP, Sorensen JA. Adipose-derived stem cells: new treatment for wound healing? Ann Plast Surg 2015;75:117-23.
22. Owczarczyk-Saczonek A, Wocior A, Placek W, Maksymowicz W, Wojtkiewicz J. The use of adipose-derived stem cells in selected skin diseases (Vitiligo, Alopecia, and Nonhealing Wounds). Stem Cells Int 2017;2017:4740709.
23. Chavez-Munoz C, Nguyen KT, Xu W, Hong SJ, Mustoe TA, Galiano RD. Transdifferentiation of adipose-derived stem cells into keratinocyte-like cells: engineering a stratified epidermis. PLoS One 2013;8:e80587.
24. Hu R, Ling W, Xu W, Han D. Fibroblast-like cells differentiated from adipose-derived mesenchymal stem cells for vocal fold wound healing. PLoS One 2014;9:e92676.
25. Kim WS, Park BS, Sung JH, Yang JM, Park SB, Kwak SJ, Park JS. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci 2007;48:15-24.
26. Zhao L, Johnson T, Liu D. Therapeutic angiogenesis of adipose-derived stem cells for ischemic diseases. Stem Cell Res Ther 2017;8:125.
27. Garcia-Honduvilla N, Cifuentes A, Ortega MA, Delgado A, Gonzalez S, Bujan J, Alvarez-Mon M. High sensitivity of human adipose stem cells to differentiate into myofibroblasts in the presence of C. aspersa egg extract. Stem Cells Int 2017;2017:9142493.
28. Kim BS, Tilstam PV, Springenberg-Jung K, Boecker AH, Schmitz C, Heinrichs D, Hwang SS, Stromps JP, Ganse B, Kopp R, Knobe M, Bernhagen J, Pallua N, Bucala R. Characterization of adipose tissue macrophages and adipose-derived stem cells in critical wounds. Peer J 2017;5:e2824.
29. Kim BS, Gaul C, Paul NE, Dewor M, Stromps JP, Hwang SS, Nourbakhsh M, Bernhagen J, Rennekampff HO, Pallua N. The effect of lipoaspirates on human keratinocytes. Aesthet Surg J 2016;36:941-51.
30. Moon KM, Park YH, Lee JS, Chae YB, Kim MM, Kim DS, Kim BW, Nam SW, Lee JH. The effect of secretory factors of adipose-derived stem cells on human keratinocytes. Int J Mol Sci 2012;13:1239-57.
31. Haubner F, Muschter D, Pohl F, Schreml S, Prantl L, Gassner HG. A co-culture model of fibroblasts and adipose tissue-derived stem cells reveals new insights into impaired wound healing after radiotherapy. Int J Mol Sci 2015;16:25947-58.
32. Maharlooei MK, Bagheri M, Solhjou Z, Jahromi BM, Akrami M, Rohani L, Monabati A, Noorafshan A, Omrani GR. Adipose tissue derived mesenchymal stem cell (AD-MSC) promotes skin wound healing in diabetic rats. Diabetes Res Clin Pract 2011;93:228-34.
33. Nambu M, Kishimoto S, Nakamura S, Mizuno H, Yanagibayashi S, Yamamoto N, Azuma R, Kiyosawa T, Ishihara M, Kanatani Y. Accelerated wound healing in healing-impaired db/db mice by autologous adipose tissue-derived stromal cells combined with atelocollagen matrix. Ann Plast Surg 2009;62:317-21.
34. Steinberg JP, Hong SJ, Geringer MR, Galiano RD, Mustoe TA. Equivalent effects of topically-delivered adipose-derived stem cells and dermal fibroblasts in the ischemic rabbit ear model for chronic wounds. Aesthet Surg J 2012;32:504-19.
35. Tsumano T, Kawai K, Ishise H, Nishimoto S, Fukuda K, Fujiwara T, Kakibuchi M. A new mouse model of impaired wound healing after irradiation. J Plast Surg Hand Surg 2013;47:83-8.
36. Huang JI, Zuk PA, Jones NF, Zhu M, Lorenz HP, Hedrick MH, Benhaim P. Chondrogenic potential of multipotential cells from human adipose tissue. Plast Reconstr Surg 2004;113:585-94.
37. Ebrahimian TG, Pouzoulet F, Squiban C, Buard V, Andre M, Cousin B, Gourmelon P, Benderitter M, Casteilla L, Tamarat R. Cell therapy based on adipose tissue-derived stromal cells promotes physiological and pathological wound healing. Arterioscler Thromb Vasc Biol 2009;29:503-10.
38. Akita S, Akino K, Hirano A, Ohtsuru A, Yamashita S. Noncultured autologous adipose-derived stem cells therapy for chronic radiation injury. Stem Cells Int 2010;2010:532704.
39. Bura A, Planat-Benard V, Bourin P, Silvestre JS, Gross F, Grolleau JL, Saint-Lebese B, Peyrafitte JA, Fleury S, Gadelorge M, Taurand M, Dupuis-Coronas S, Leobon B, Casteilla L. Phase I trial: the use of autologous cultured adipose-derived stroma/stem cells to treat patients with non-revascularizable critical limb ischemia. Cytotherapy 2014;16:245-57.
40. Liu Y, Xu Y, Fang F, Zhang J, Guo L, Weng Z. Therapeutic efficacy of stem cell-based therapy in peripheral arterial disease: a meta-analysis. PLoS One 2015;10:e0125032.
41. Zollino I, Zuolo M, Gianesini S, Pedriali M, Sibilla MG, Tessari M, Carinci F, Occhionorelli S, Zamboni P. Autologous adipose-derived stem cells: basic science, technique, and rationale for application in ulcer and wound healing. Phlebology 2017;32:160-71.
42. Conde-Green A, Marano AA, Lee ES, Reisler T, Price LA, Milner SM, Granick MS. Fat grafting and adipose-derived regenerative cells in burn wound healing and scarring: a systematic review of the literature. Plast Reconstr Surg 2016;137:302-12.
43. Gal S, Ramirez JI, Maguina P. Autologous fat grafting does not improve burn scar appearance: a prospective, randomized, double-blinded, placebo-controlled, pilot study. Burns 2017;43:486-9.
44. Meng FZ, D, Li, W. Adipose-derived stem cells as a potential weapon for diabetic foot ulcers. Int J Clin Exp Med 2017;10:15967-73.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Kim BS, Debye B, Beier JP. Adipose-derived stem cells in cutaneous wound repair. Plast Aesthet Res 2018;5:31. http://dx.doi.org/10.20517/2347-9264.2018.26
AMA Style
Kim BS, Debye B, Beier JP. Adipose-derived stem cells in cutaneous wound repair. Plastic and Aesthetic Research. 2018; 5: 31. http://dx.doi.org/10.20517/2347-9264.2018.26
Chicago/Turabian Style
Kim, Bong-Sung, Berthold Debye, Justus P. Beier. 2018. "Adipose-derived stem cells in cutaneous wound repair" Plastic and Aesthetic Research. 5: 31. http://dx.doi.org/10.20517/2347-9264.2018.26
ACS Style
Kim, B.S.; Debye B.; Beier JP. Adipose-derived stem cells in cutaneous wound repair. Plast. Aesthet. Res. 2018, 5, 31. http://dx.doi.org/10.20517/2347-9264.2018.26
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 1 clicks
Cite This Article 1 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.