Early experiences with the use of Earfold™ for correction of prominent ears
Abstract
Aim: This paper describes the author’s personal experience with using the minimally-invasive Earfold™ implant.
Methods: The author inserted the Earfold™ implant into 19 patients (5 men, 14 women) between November 2016 and June 2018. Bilateral implantation was performed in 14 patients. In 5 cases, implantation was limited to one ear. The author’s main indication for treatment was a helical-mastoid distance of more than 20 mm. Additional antihelixplasty of the upper ear was performed in 4 patients (2 primarily and 2 after explantation). One patient underwent simultaneous treatment of protruding ear lobes.
Results: The overall satisfaction rate was high, with 16 patients (84%) being satisfied or very satisfied. The procedure proved to be rapid with little down-time in the recovery phase. The demand for, and acceptance of the procedure was high. Although surgical otoplasty was always discussed as an alternative, no patient who presented for consultation chose standard otoplasty surgery. Complications occurred in 6 patients and implants were removed in 5 patients, 1 of them completely.
Conclusion: The EarfoldTM procedure is an interesting, minimally-invasive alternative to surgical otoplasty which produces results which patients are pleased with. However, in this early series, the complication rate was high. A hybrid technique might reduce the complications observed.
Keywords
Introduction
The demand for non-surgical or minimally-invasive procedures in aesthetic surgery continues to increase. To date, the only minimally-invasive otoplasty procedure that has been described is the incisionless thread technique described by Fritsch[1] and modified by Haytoglu et al.[2]. The Earfold procedure, introduced by Kang and Kerstein[3], offers a new approach to minimally-invasive prominent ear correction and uses a metal clip to reshape the antihelical fold. In this article, the author reported his early experiences with this new implant.
Methods
The Earfold™ implant was inserted into the ears of 19 patients between November 2016 and February 2018.
Earfold clips (Earfold, Allergan, Clonshaugh Business and Technology Park, Coolock, Dublin, D17 E400, Ireland) are made from thin strips of nitinol (nickel-titanium alloy) which are heat-treated and coated with 24-carat gold. The ends of the implant are fitted with tiny teeth. Once deployed, the super-elastic properties of the material ensure that the implant returns to its predetermined form[4] forcing the cartilage of the antihelix to adopt a new shape.
According to the instructions for use (IFU), the specific indication for use of the implant is an absence or poor definition of the antihelical fold in a patient who perceives that their ears are prominent. The likely outcome is first demonstrated to the patient by using non-sterile Prefold™ positioners. These are identical in shape, size and elasticity to Earfold™ but do not have the tiny teeth and can therefore be easily placed onto the ear in the region of the antihelical fold - simulating the probable outcome for the patient. Assessment of patients with Prefold™ is an important first step to determining whether a patient is suitable for treatment with Earfold™. If it proves difficult or impossible to produce a satisfactory aesthetic outcome for the patient, and/or if the implant is unlikely to end up lying flush with the cartilage, the IFU advises that an alternative to Earfold™ should be used. Once the assessment with Prefold™ is complete, the outline of the positioner is marked on the skin. This then determines the specific site for implantation of the Earfold™ implant.
After meticulously disinfecting the skin with Octenisept (0.1 g octenidine hydrochloride, 2.0 g phenoxyethanol/100 g), the site was anesthetized with xylocaine 2% and epinephrine 1:200,000. The author used a vertical incision between the outer margin of the ear and the antihelical fold, and performed a subcutaneous and subperichondrial dissection of the previously marked area. The author then scored the cartilage to weaken its bending force using either a needle or scalpel.
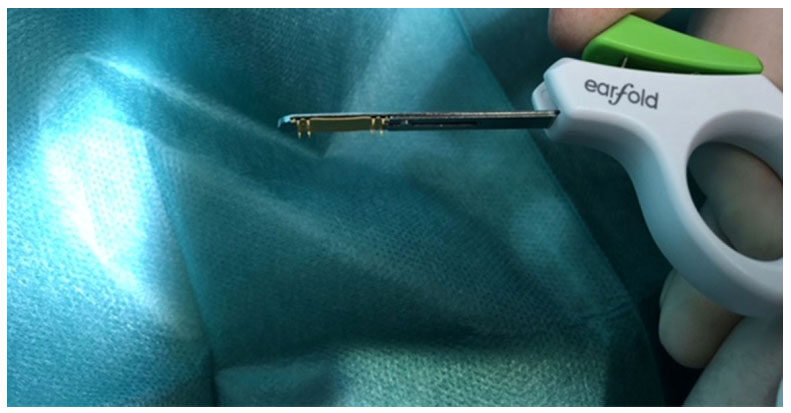
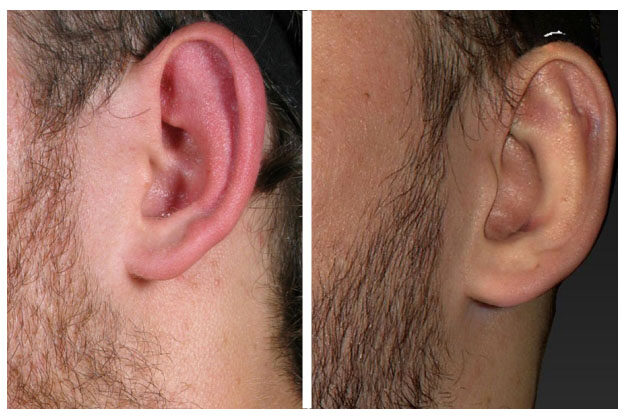
The implant was then released subcutaneously (and hopefully sub-perichondrially) flat against the anterior cartilage in the position of the previously marked area. Once deployed with the introducer [Figure 1], the Earfold™ implant returned to its predetermined shape and grasped the cartilage, forcing the antihelix to adopt a new shape, thereby correcting any associated prominence of the ear [Figure 2].
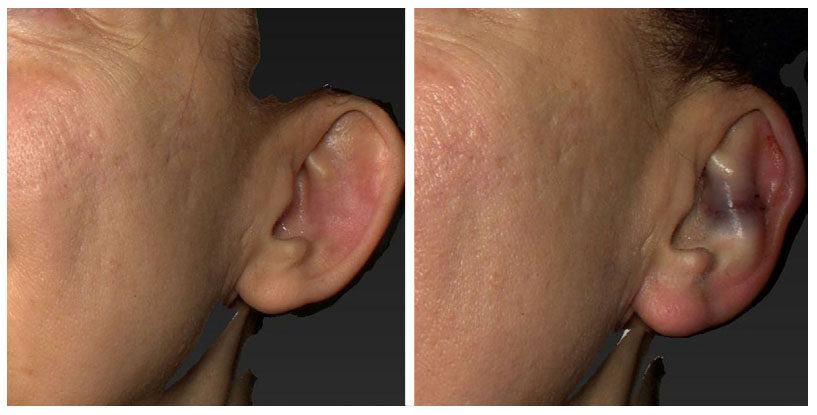
Figure 2. Single EarfoldTM implant in left ear [appearance before (left) and after (right) insertion]
The author then closed the skin with 6-0 Prolene or skin glue such as Dermabond (Ethicon Inc., Route 22, West Somerville, NJ, 08876, USA).
Post-operative pain and bruising was managed with a combination of ibuprofen 600 mg three times a day and bromelain 3 tablets a day. Antibiotic prophylactic treatment was commenced one day before the procedure with cefuroxime 500 mg twice a day. No dressing was used.
Post-operatively, the author advised patients to refrain from swimming or engaging in contact sports for the 3-4 weeks following surgery. The author also advised them to avoid any contact with water during this time. To further prevent accidental infection, any items in contact to the ear (cell phones, earplugs helmets, earrings) should not be used, or only with extreme caution and held away from the incisions. Patients with a history of recurrent otitis externa were also excluded from surgery or were followed up closely after the procedure.
Results
The author inserted Earfold™ into the ears of 19 patients between November 2016 and June 2018. Unilateral implantation was performed in 5 cases; 3 were treated with 1 implant, 2 with 2 implants in each ear. Bilateral implantation was performed in 14 patients; 2 patients had 2 implants inserted into each ear. The principal motive for all patients was to avoid standard otoplasty surgery. Many patients had additional reservations about the need for bulky and visible dressings after standard otoplasty - that might make it obvious that an otoplasty had been performed.
Fifty-one patients underwent pre-operative assessment with Prefold™: 42 women/girls and 9 men. In 6 patients, the author advised that they should not undergo the Earfold™ procedure because of a deep conchal bowl. Moreover, every patient was explicitly advised of the pros and the cons of Earfold™ compared to standard surgical otoplasty (especially using the Mustardé technique[5]). Importantly, Prefold™ positioners were used to simulate the likely outcome in every case, including those patients with a deep conchal bowl. Interestingly, for some of these patients, even when it was pointed out that they might have to accept an aesthetic compromise, this was mostly accepted.
Nineteen of the 51 patients who underwent a Prefold™ assessment decided to undergo the Earfold™ procedure. Interestingly, of those who declined to go ahead with treatment using Earfold™, none went on to opt for a standard surgical alternative suggesting that this is a group of patients who would not otherwise come forward for treatment of their prominent ears.
Implants were placed at the inferior part of the antihelical fold in 5 patients. In 19 cases, the implant was placed at the superior part of the antihelical fold (11 proximal to the bifurcation, 8 distal to the bifurcation). In addition, surgery to reduce the size of the anti-tragus was performed bilaterally in 1 patient. In further 4 patients, Earfold™ was used in combination with Mustardé sutures.
Building familiarity with the introducer in the weeks before surgery is performed is critical for eventual correct deployment of the implant. In 2 cases, the implants had to be discarded as the implants were noted to be sitting proud of the cartilage. In those cases, the cartilage had not been kept flat in relation to the cartilage during release of the implant.
The author did not use a validated method of assessment of patient satisfaction and the average duration of follow-up in this series was short (6 months). However, anecdotally, 16 out of 19 patients were satisfied or very satisfied with the aesthetic result. Three patients were dissatisfied with the outcome of treatment due to issues with persistent pain and sensitivity, which were resolved after removal of the implants and the performance of a Mustardé otoplasty.
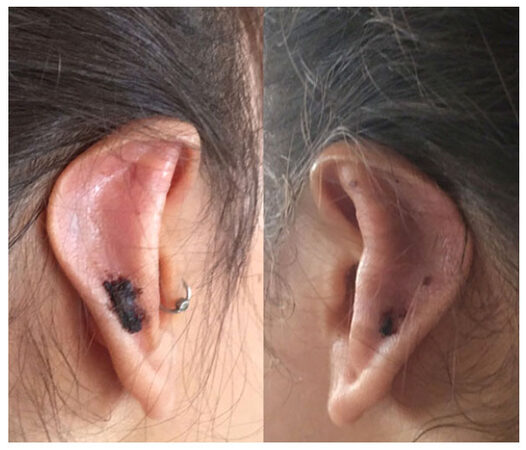
In total, the author removed 8 implants from 5 patients. Removal was necessary because of erosion of the skin over the implant in 2 patients [Figures 3 and 4] and because of continuing pain and inflammation around the implant in 2 cases. In one case, the positioning of the implant was aesthetically unsatisfying, inducing an excessive folding. Removals were performed within 2 to 15 months after implantation.
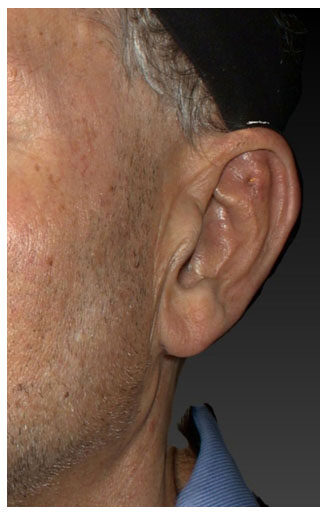
Figure 3. Skin erosion of the upper edge of an implant placed over the superior crus of the antihelical fold
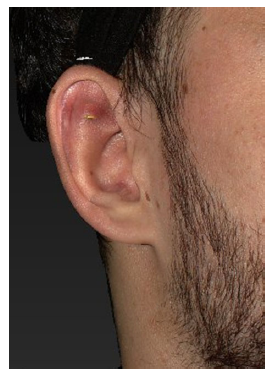
Figure 4. Skin erosion of the lower edge of an implant placed over the superior crus of the antihelical fold at 15 months after implantation
Careful examination of the cases with skin erosion [Figure 3] showed that the implant was not flush with the cartilage resulting in a sharp edge of the implant sitting proud and eventually eroding through the skin. Although every effort was made to ensure that the implants were completely flush with the cartilage at the time of deployment, it is entirely possible that minor mispositioning of the implant was not detected due to soft-tissue swelling at the time of surgery. Alternatively, the implants may have moved due to patient factors during the first 3-4 weeks after surgery (i.e., before encapsulation of the implant occurred preventing any further movement).
In the second case with skin erosion [Figure 4] the reason is unclear as the problem became evident 15 months after implantation. Primary insufficient positioning is unlikely, or the symptoms must have started earlier. In this case, the author did no scratching to the cartilage; the unweakened strength of the cartilage might have forced the implant out of its position.
Discussion
The author acknowledges that the limited number of patients treated in this series means that this report probably reflects a very preliminary impression of the potential of this new technique. However, as there are few independent reports of outcomes using the Earfold™ implant, the author believes that providing surgeons with additional information on his outcomes is important.
The procedure is promoted as being fast and minimally invasive with an acceptable rate of complications. The author accepts that the technique is fast and minimally invasive. However, the rate of complications in this series was higher than that reported in the “first-in-human” pilot study by Kang and Kerstein[3] who reported 7 cases of skin erosion (13% of patients) with complications affecting 20.5% of patients. The higher rate in the present series occurred despite the fact that the author applied the same care to the Earfold™ technique as to surgical otoplasty. Therefore, the author has concluded that the Earfold™ technique is different to standard otoplasty, with a different learning curve and different technical requirements - even for an experienced otoplasty surgeon.
Though the complication rate reported by Kang and Kerstein[3] is lower, it is not neglectable-especially compared to the rates of revision with the classic Mustardé technique of 2.9% reported by Olivier et al.[6]. This leads to the question if the complications encountered are purely surgery-related or if additional immanent factors might add to them. Possible mechanical irritation of the implant in combination with the bradytrophy of the cartilage might predispose to local infections.
The author observed complications in 6 out of 19 patients [Table 1]. Four patients developed pain and discomfort at the implantation site and went on to develop an infection forcing us to explant the clips in 2 cases; in 2 patients the clip induced an excessive folding. In total, 5 patients had to have their implants removed. Two patients only had 1 of 2 implants removed - from the superior crus. After removal of the implant, patients developed a recurrence of their prominence and so the author performed an upper antihelixplasty in 4 patients using the Mustardé technique, leading to an aesthetically and functionally satisfactory result without complications.
Complications (more than one event can occur in one patient)
| Patients (n = 19) | Perichondritis subcutaneous infection, neuralgia | Abscess | Perforation + neualgia | Neuralgia | Insufficient aesthetic result | Explantation, revision, reposition |
|---|---|---|---|---|---|---|
| Implant superior anthelixfold distal to bifurcation (n = 8) | 2 | 2 | 1 | 3 | 3 | 4 |
| Implant superior anthelixfold proximal to bifurcation (n = 11) | - | - | - | 2 | 1 | 1 |
| Implant inferior anthelixfold (n = 5) | - | - | - | - | - | - |
| Anthelixplasty (Mustardé) (n = 4) | - | - | - | - | - | - |
| Correction of protruded lobula (n = 1) | - | - | - | - | - | - |
In 3 of the 4 patients who developed an infection, the author did not provide prophylactic antibiotic cover. However, in view of the infections, the author now covers every case with oral antibiotics (cefuroxime), beginning one day prior to the procedure. The author suggests starting prophylactic antibiotic coverage the day before the procedure using 2× cefuroxime 500 mg/day and continuing this for 1 week. In the author’s opinion, mechanical irritation of the cartilage predisposes to infection. However, the author accepts that other surgeons might disagree with this opinion. For example, Kang and Kerstein[3] suggest that infection is more likely if non-absorbable sutures are used because of the additional trauma involved in their removal. Moreover, Kang and Kerstein[3] suggested that the use of earrings, mobile phones or direct trauma to the area during the first 3-4 weeks after implantation were also important aetiological factors for infection.
Skin erosions affected 2 patients and occurred exclusively for implants placed over the superior crus. Therefore, the author suggests that placement of implants at this site should be avoided altogether. Alternatively, the implant could be used in combination with Mustardé sutures for the upper pole - as a hybrid procedure. In the author’s opinion, a hybrid approach would neither significantly prolong the duration of the procedure nor have any major impact on down-time. If the Mustardé sutures were to be placed solely in the upper pole of the ear, then there would be no need to use an ear/head bandage - which is so disliked by patients [Figure 5].
Figure 5. Earfold™ - hybrid technique: Earfold™ implant inferior antihelical fold, Mustardé technique upper antihelical fold [before (left) and after implantation (right)]. In this case, there was additional reduction of the antitragus
One patient complained that their implants were too visible under the skin. The 24-carat gold coating of the Earfold™ implant is intended to make them less visible under the skin. However, the author has noted that even when the implants are flush with the cartilage, the contour of the implants is mostly detectable as a slightly raised area and patients should be warned of this before treatment.
The author encountered a problem of a “Spock-ear” in two cases [Figure 6]. The author has noted that this was the result of creating an antihelical fold that was too vertical in patient where the cartilage was relatively soft. Therefore, there was a degree of overcorrection of the prominence. From this and other experiences with using Earfold™, the author has concluded that although the technique seems simple in principle, it is critical to:
Figure 6. Excessive folding and "Spock"-ear deformity. This was not anticipated during the Prefold™ assessment and was probably the result of creating an antihelical fold which was too vertical in an ear with relatively weak cartilage. After removal of the upper implants, an aesthetically satisfying result was achieved
• Perform careful patient selection in advance of treatment - using Prefothat the implant is flat in relationd™;
• Ensure that the implant is flat in relation to the cartilage before and after deployment;
• Consider (weakening) of the cartilage - either through needle perforation or scoring with a scalpel - before placement of the implant, especially in patients with very thick and inelastic cartilage;
• Consider antibiotic coverage;
• Observe careful post-operative management.
All of these factors are of course detailed in the IFU for the implant and surgeons are strongly advised to read this material carefully before proceeding to use the implant.
Although the author did not perform a formal assessment of satisfaction using a validated assessment score (e.g., Ear-Q), anecdotally, patients reported a high level of satisfaction with treatment [Table 2]. This correlates with the experience of Kang and Kerstein[3] in their pilot study of Earfold™. In our series, a total of three patients were dissatisfied with the aesthetic result and insisted on explantation. These patients also complained of pain and sensitivity related to the clips, so it is difficult to know whether it was the pain and sensitivity or dissatisfaction with the appearance that motivated them to have their implants removed. In 1 patient, we removed only the upper implant, leading to a much more pleasing result. Total explantation due to aesthetic dissatisfaction was performed in 2 patients (4 implants).
Self-reported satisfaction with the aesthetic result (n = 19)
| Very dissatisfied | Dissatisfied | No change | Satisfied | Very satisfied |
|---|---|---|---|---|
| 1 | 2 | 3 | 13 |
In conclusion, the minimally invasive Earfold™ technique for antihelixplasty by implantation of nitinol clips is a fast and aesthetically satisfactory procedure (16 out of 19 patients satisfied). The author can confirm that the postoperative recovery is very short and in general only limited by a small degree of localised swelling at the implantation site. Importantly, no ear/head bandage or headband was necessary and the author’s patients were able to return to normal social interactions almost immediately - an important factor in deciding to proceed with treatment.
Although the complication rate in this series was high, the author accepts that this probably might partially reflect the learning curve with the technique which is very different to standard otoplasty. The actual study of Kang et al.[7] presents 403 patients with a complication rate requiring intervention of 9.7%. This is still high compared to revision rates of 2.9% in surgical otoplasty as reported by Olivier et al.[6] and it reflects that beside surgery-related complication, immanent factors might have to be considered like mechanical irritation. Further long term observations have to show that the safety and efficacy of an Earfold™ otoplasty is equivalent to that of surgical otoplasty. Surgeons should be familiar with the complication management and standard otoplasty manoeuvres if they decide to offer treatment with Earfold™. In the case of explantation they should be able to convert patients to standard otoplasty (e.g., Mustardé) without difficulty.
The overall aesthetic results were very satisfying and the procedure meets the demands of many patients for a fast and minimal invasive otoplasty. Despite the reported problems, the author believes therefore that this technique offers a promising alternative to surgical otoplasty and might eventually be combined with a modified surgical approach.
Indeed, it is the author’s opinion that Earfold™ is most effective and secure when used as part of a hybrid approach where the implant is placed at the middle third of the ear and used in combination with other surgical manoeuvres for correction of prominent ears (e.g., Mustardé sutures).
Further studies should be performed to see if there are ways to shorten the learning curve and reduce the complication rate. Moreover, increasing the range of angles of the clips would help expand the number of patients who may benefit from this new treatment option.
Declarations
Authors’ contributionsSchuster BW contributed solely to the paper.
Availability of data and materialsThe data presented is original and obtained in the author’s clinic. It can be made available if required.
Financial support and sponsorshipNone.
Conflicts of interestThere are no conflicts of interest.
Ethical approval and consent to participateAs a retrospective study, no ethical approval was necessary. All photographs have been anonymized, even though written consent was obtained from all patients.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
2. Haytoglu S, Haytoglu TG, Bayar Muluk N, Kuran G, Arikan OK. Comparison of two incisionless otoplasty techniques for prominent ears in children. Int J Pediatr Otorhinolaryngol 2015;79:504-10.
3. Kang NV, Kerstein RL. Treatment of prominent ears with an implantable clip system: a pilot study. Aesthet Surg J 2016;36:NP100-16.
5. Mustarde JC. Cosmetic surgery. II. The correction of prominent ears with buried mattress sutures. Mod Trends Plast Surg 1964;16:233-6.
6. Olivier B, Mohammad H, Christian A, Akram R. Retrospective study of the long-term results of otoplasty using a modified Mustardé (cartilage-sparing) technique. J Otolaryngol Head Neck Surg 2009;38:340-7.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Schuster BW. Early experiences with the use of Earfold™ for correction of prominent ears. Plast Aesthet Res 2018;5:22. http://dx.doi.org/10.20517/2347-9264.2018.13
AMA Style
Schuster BW. Early experiences with the use of Earfold™ for correction of prominent ears. Plastic and Aesthetic Research. 2018; 5: 22. http://dx.doi.org/10.20517/2347-9264.2018.13
Chicago/Turabian Style
Schuster, Bernd W.. 2018. "Early experiences with the use of Earfold™ for correction of prominent ears" Plastic and Aesthetic Research. 5: 22. http://dx.doi.org/10.20517/2347-9264.2018.13
ACS Style
Schuster, BW. Early experiences with the use of Earfold™ for correction of prominent ears. Plast. Aesthet. Res. 2018, 5, 22. http://dx.doi.org/10.20517/2347-9264.2018.13
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.