Larger breast implants warranted for post-mastectomy reconstruction
Abstract
Aim: Our goal was to ascertain if there was a role for larger breast implants in breast reconstruction.
Methods: Patients that underwent mastectomy and implant-based breast reconstruction were identified and reviewed.
Results: Of the total specimens, 92 (14.7%) weighed more than 800 g with a mean weight of 1140 g (range 803 to 2177 g). Of the patients with these larger specimens, 45 (48.9%) selected the largest available implants (800 mL implants) for their reconstruction.
Conclusion: There are patients undergoing mastectomy and implant-based breast reconstruction who are unable to have reconstruction to their native breast volume because of the current implant-volume restrictions.
Keywords
Introduction
The mastectomy rate in the United States has been steadily increasing, including the rate for contralateral prophylactic mastectomy and elective mastectomy[1-5]. Mastectomies permanently alter a patient’s body image and thereby impact self-esteem[6]. Patients who undergo breast reconstruction after mastectomy are more satisfied than those who undergo mastectomy without reconstruction[7-9]. However, the reconstructed breast can leave a patient feeling incompletely restored when the native breast size was larger than reconstructed breast volume[10]. Limited data exists that links patient satisfaction to the size of a reconstructed breast[10], especially for patients who have native breast volumes larger than 800 mL. These women are not able to match their native breast size with implant alone based reconstruction with the current implant-size limitations imposed by the US Food and Drug Administration (FDA) (maximum volume, 800 mL, Table 1). Of note, the FDA has recently approved human trials of larger breast implants (volumes to 1445 mL)[11,12]. We designed a retrospective study to determine how native breast volumes related to reconstructive implant volumes for patients who underwent mastectomy followed by reconstruction with a silicone gel-filled implant.
FDA-approved breast implants (January 1, 2016)
| Breast implants | Volume range (mL) |
|---|---|
| Saline-filled | |
| Ideal Implant Saline-Filled Breast Implant (Ideal Implant Inc.) | 210-755 |
| Allergan Medical RTV Saline-Filled Breast Implant (Allergan, Inc.) | 120-800 |
| Mentor and Spectrum (Mentor Worldwide) | 125-700 |
| Silicone Gel-filled | |
| Allergan Natrelle (Allergan, Inc.) | 80-800 |
| Allergan Natrelle 410 (Allergan, Inc.) | 140-740 |
| Mentor MemoryGel (Mentor Worldwide) | 100-800 |
| Mentor MemoryShape (Mentor Worldwide) | 120-775 |
| Sientra (Silimed Indústria de Implantes Ltda) | 80-700 |
Methods
This retrospective study was conducted at 2 tertiary care centers after institutional review board approval at both locations. We reviewed electronic health records of patients who underwent mastectomy followed by reconstruction with silicone-gel breast implants during a 5-year period from January 2009 to December 2014. All patients included in the study underwent mastectomy either for a diagnosis of breast cancer or a desire for prophylactic mastectomy. All patients had reconstruction with breast implants either at the time of mastectomy or by tissue expansion and subsequent implant reconstruction. Each patient had only one implant per reconstructed breast, no implant stacking was utilized. We collected data regarding mastectomy specimen weights and final implant volumes used in the breast reconstructions. The patients’ native breast volume was extrapolated from the recorded mastectomy specimen mass. Each breast was considered separately (a patient with a bilateral mastectomy and bilateral implant reconstruction was entered in the data twice, with unique data for each breast). Patients who underwent any form of autologous tissue reconstruction or had saline implants were excluded.
Results
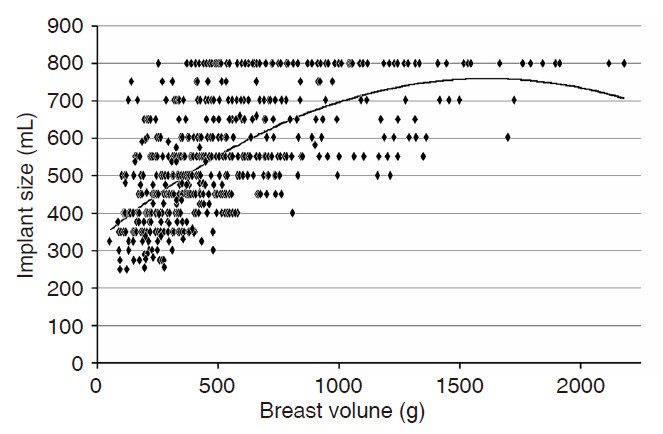
Weights were available for 627 mastectomy specimens for patients who underwent mastectomy and implant-based reconstruction during the 5-year period. The mean gross mastectomy specimen weight was 501.2 g (range 51 to 2177 g). The mean implant size used in the reconstructions was 533.9 mL. The mean patient body mass index (BMI) was 26.9 (range 16.6 to 52). Of the total specimens, 92 (14.7%) weighed more than 800 g (mean 1140 g and range 803 to 2177 g) and these patients had a mean BMI of 34.0 (21.3 to 52). Forty-five (48.9%) of the patients with these larger specimens selected 800 mL implants for their reconstructions. From our total patient population, 80 patients (12.7%) chose 800 mL implants for their reconstructions. The mean specimen weight for this group was 933 g (252 to 2177 g) and the mean BMI was 34.6 (20.3 to 49.5). The Figure 1 shows the trend for native breast mass (as a proxy for volume) vs. reconstruction implant size.
Discussion
Patients’ desires ultimately determine the goal of post-mastectomy breast reconstruction. Not all patients with large native breasts (mastectomy specimens > 800 g) selected 800 mL implants for their reconstruction. Alternatively, some women with smaller breasts (mastectomy specimens < 800 g) chose to increase the size of their breasts at the time of implant-based reconstruction by selecting 800 mL implants. Thus, at the time of breast reconstruction, some patients with large breasts wanted to have smaller reconstructed breasts, some with smaller breasts wanted larger reconstructed breasts, and many elected to maintain a similar breast volume following their mastectomy. Other factors to be considered in the decision regarding the size of implant used in reconstruction may include history of radiation or other comorbid conditions such as current smoking status and diabetes. Native breast shape and degree of ptosis must also be considered. These are especially important when trying to achieve symmetry in unilateral breast reconstruction. There are many reconstructive options available to patients, thus surgeons must aid patients in this decision making process.
A study by Huber et al.[10] reported that women who augmented their native breast volume at the time of reconstruction were more satisfied with their overall reconstructive outcome than those who did not, and no increase in complication rate occurred in those who augmented their breast volume. For woman with large breasts, low patient satisfaction may be related to the inability to match native breast volume with a similarly sized implant at reconstruction because of current implant-volume restrictions. Patient-reported outcomes would provide more insight as to what influences patients’ initial decisions, if they remain satisfied long-term with their choice, and if they would have chosen differently had a larger implant been available at the time of their reconstruction.
Women may have asymmetry between native breast volumes. Those who underwent unilateral mastectomy with implant based reconstruction likely desired their reconstructed breast to appear similar in size and volume to their native breast. This would affect their choice in implant size.
Our study is limited by the lack of patient-satisfaction data for our patient population. However, it was not designed to evaluate this aspect of breast reconstruction. To investigate this further, we would use an outcome measurement tool, such as the BREAST-Q questionnaire (Memorial Sloan Kettering Cancer Center). Future studies could investigate the relationship between patients’ preoperative decisions and postoperative satisfaction scores. For example, how many patients would have selected an implant with a volume > 800 mL if they had that option available to them at the time of their reconstruction? This will be especially informative when patients have the option to match their native breast volume to breast implant volumes as large as 1445 mL.
In addition to satisfaction data, future studies will investigate complication rates to ensure that larger implants are as safe and effective as those currently approved for use in reconstruction. Larger implants will have their own unique risks. Known complications that may be associated with breast implants include, but are not limited to, asymmetry, tissue atrophy/skin necrosis, extrusion, infection, hematoma, ptosis, and pain. The specific incidence of these, and other, complications associated with large volume implant use will need to be determined.
Furthermore, patients with class II or III obesity have an increased risk of surgical morbidity following breast reconstruction of any modality[13]. The risks of larger implant use in this population should be carefully considered. In our study, women with breast volumes greater than 800 g had a mean BMI of 34.0. BMIs of 30-34.99 are classified by the World Health Organization as obesity class I[14]. This patient cohort is not at increased risk of surgical morbidity following breast reconstruction[13].
There may be a role for implants larger than 800 mL for patients undergoing post-mastectomy breast reconstruction in the United States. The FDA has recently approved ATHENA, a clinical trial that will allow patients to select breast implants with larger volumes ranging from 800 to 1445 mL for breast reconstruction. Patient preferences and outcome goals will continue to guide reconstructive efforts. Future studies on satisfaction and complication rates will allow us to better counsel our patients and assist them in their decision making.
Declarations
AcknowledgmentsThe authors would like to thank Mark Todd, PA (Department of Laboratory Medicine and Pathology, Mayo Clinic AZ) and Dr. Robert Bernard (Associate Professor of Surgery at the University of Arizona College of Medicine and an Adjunct Assistant Professor of Plastic Surgery at Mayo Clinic AZ) for their invaluable assistance with this manuscript.
Authors’ contributionsAbstract and manuscript author, data collection: A.L. Howarth
Substantial data collection, manuscript review: A.M. Rodriguez
Abstract writing and data collection, manuscript review: V. Gargya
Substantial data collection, manuscript review: H.D. Lucas
Primary investigator, original idea, manuscript review: R.C. Mahabir
Financial support and sponsorshipNone.
Conflicts of interestDr. Mahabir serves on the Mentor Advisory Board.
Patient consentInstitutional review board approval was obtained at both locations of data collection prior to beginning this retrospective study.
Ethics approvalInstitutional review board approval was obtained at both locations of data collection prior to beginning this study.
Copyright© The Author(s) 2017.
REFERENCES
1. Dragun AE, Huang B, Tucker TC, Spanos WJ. Increasing mastectomy rates among all age groups for early stage breast cancer: a 10-year study of surgical choice. Breast J 2012;18:318-25.
2. Dragun AE, Pan J, Riley EC, Kruse B, Wilson MR, Rai S, Jain D. Increasing use of elective mastectomy and contralateral prophylactic surgery among breast conservation candidates: a 14-year report from a comprehensive cancer center. Am J Clin Oncol 2013;36:375-80.
3. Katipamula R, Degnim AC, Hoskin T, Boughey JC, Loprinzi C, Grant CS, Brandt KR, Pruthi S, Chute CG, Olson JE, Couch FJ, Ingle JN, Goetz MP. Trends in mastectomy rates at the Mayo Clinic Rochester: effect of surgical year and preoperative magnetic resonance imaging. J Clin Oncol 2009;27:4082-8.
4. Kummerow KL, Du L, Penson DF, Shyr Y, Hooks MA. Nationwide trends in mastectomy for early-stage breast cancer. JAMA Surg 2015;150:9-16.
5. Mahmood U, Hanlon AL, Koshy M, Buras R, Chumsri S, Tkaczuk KH, Cheston SB, Regine WF, Feigenberg SJ. Increasing national mastectomy rates for the treatment of early stage breast cancer. Ann Surg Oncol 2013;20:1436-43.
6. Rowland JH, Desmond KA, Meyerowitz BE, Belin TR, Wyatt GE, Ganz PA. Role of breast reconstructive surgery in physical and emotional outcomes among breast cancer survivors. J Natl Cancer Inst 2000;92:1422-9. Erratum in: J Natl Cancer Inst 2001;93:68
7. Atisha DM, Rushing CN, Samsa GP, Locklear TD, Cox CE, Shelley Hwang E, Zenn MR, Pusic AL, Abernethy AP. A national snapshot of satisfaction with breast cancer procedures. Ann Surg Oncol 2015;22:361-9.
8. Guyomard V, Leinster S, Wilkinson M. Systematic review of studies of patients' satisfaction with breast reconstruction after mastectomy. Breast 2007;16:547-67.
9. Ng SK, Hare RM, Kuang RJ, Smith KM, Brown BJ, Hunter-Smith DJ. Breast reconstruction post mastectomy: patient satisfaction and decision making. Ann Plast Surg 2016;76:640-4.
10. Huber KM, Zemina KL, Tugertimur B, Killebrew SR, Wilson AR, DallaRosa JV, Prabhakaran S, Dayicioglu D. Outcomes of breast reconstruction after mastectomy using tissue expander and implant reconstruction: bigger is better. Ann Plast Surg 2016;76:S316-9.
11. FDA Investigational Device Exemption (IDE) approval number G150173.
12. Mentor Receives FDA IDE Approval for its ATHENA Clinical Trial. Available from: http://www.prnewswire.com/news-releases/mentor-receives-fda-ide-approval-for-its-athena-clinical-trial-300220185.html [Last accessed on 27 Dec 2017].
13. Fischer JP, Wes AM, Kanchwala S, Kovach SJ. Effect of BMI on modality-specific outcomes in immediate breast reconstruction (IBR)-a propensity-matched analysis using the 2005-2011 ACS-NSQIP datasets. J Plast Surg Hand Surg 2014;48:297-304.
14. World Helath Organizaton BMI Classification. Available from: http://apps.who.int/bmi/index.jsp?introPage=intro_3.html [Last accessed on 27 Dec 2017].
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Howarth AL, Rodriguez AM, Gargya V, Lucas HD, Mahabir RC. Larger breast implants warranted for post-mastectomy reconstruction. Plast Aesthet Res 2017;4:215-8. http://dx.doi.org/10.20517/2347-9264.2017.80
AMA Style
Howarth AL, Rodriguez AM, Gargya V, Lucas HD, Mahabir RC. Larger breast implants warranted for post-mastectomy reconstruction. Plastic and Aesthetic Research. 2017; 4: 215-8. http://dx.doi.org/10.20517/2347-9264.2017.80
Chicago/Turabian Style
Howarth, Ashley L., Abigail M. Rodriguez, Vipul Gargya, Heather D. Lucas, Raman C. Mahabir. 2017. "Larger breast implants warranted for post-mastectomy reconstruction" Plastic and Aesthetic Research. 4: 215-8. http://dx.doi.org/10.20517/2347-9264.2017.80
ACS Style
Howarth, AL.; Rodriguez AM.; Gargya V.; Lucas HD.; Mahabir RC. Larger breast implants warranted for post-mastectomy reconstruction. Plast. Aesthet. Res. 2017, 4, 215-8. http://dx.doi.org/10.20517/2347-9264.2017.80
About This Article
Copyright
Data & Comments
Data

 Cite This Article 3 clicks
Cite This Article 3 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.