A new tool to assess human fat grafts transplanted into nude mice using a nuclear magnetic resonance device
Abstract
Aim: Over the past two decades, there has been a dramatic increase in the research of the use of autologous fat grafting in clinical practice. Despite the many advantages this method possesses, the unpredictable fat resorption rates limit its use. The primary aim of this study was to develop an accurate, quick, non-invasive assessment tool, using the nuclear magnetic resonance (NMR) technique, which allows the injection of fat in small droplets rather than in large aliquots (the main drawback of our formerly described method) which allows assessment of fat retention in a more clinically relevant way.
Methods: A total of 7 nude mice were transplanted with human fat using the Coleman technique. Pre- and post-transplantation and then once weekly, mice were analyzed using an NMR scanner. At the end of the 7-week experimental period the mice were sacrificed.
Results: Seven weeks following transplantation 7 mice demonstrated a decrease of 40% of their average fat content compared to immediately post transplantation (standard deviation of 18%). All mice followed the same trend, and the low standard deviation throughout emphasizes the accuracy of NMR as a reliable assessment tool.
Conclusion: This preliminary study demonstrates that NMR is a reliable and accurate tool to assess fat content, and has allowed development of a clinically relevant animal model for human fat transplantation.
Keywords
Introduction
Autologous fat transplantation for soft-tissue augmentation has become increasingly popular in recent years. For more than a century fat grafting has been used for facial contouring, breast augmentation, breast reconstruction after mastectomies, post-traumatic deformities, congenital anomalies and burn injuries.[1-3]
Fat tissue is abundant, readily available, inexpensive, host compatible, associated with low morbidity and can be harvested easily and repeatedly. However, according to the literature, there is a varied overall survival rate of the fat graft in the range of 20-70%.[4-6]
Aspirated fat tissue used for autologous fat transplantation is devoid of blood microvessels which have been destroyed during aspiration and removed during processing prior to injection. Therefore, the fat tissue that is injected into a recipient is considered to be an ischemic fat cell mass. During the early period following transplantation, the fat transplant exists under hypoxic and hypo-nutritional conditions. Revascularization fail to be initiated in this early period, apoptosis ensues and results in late fat cell degeneration, low viability and ultimately fat resorption.[7]
In order to maximize the surface area and hence exposure to blood supply of the graft, surgeons now inject very small aliquots of fat grafts into multiple subcutaneous tunnels (Coleman’s technique).[8]
We previously developed a novel animal model in which human fat was grafted into the scalp area of nude mice.[9-13] This model allowed investigation of the mechanisms of fat absorbtion and exploration of the efficacy of new compounds, with potential to increase the vasculature and viability of fat grafts.[14-18] In this previous model, fat was grafted as a bolus to allow ease of collection and analysis.
However, a bolus has a relatively small surface area in contact with vascularized tissue, and therefore the center of the graft suffers from higher rates of ischemia, necrosis and resorption. Therefore, a new and more clinically relevant animal model needed to be developed.
In this manuscript, a new animal model is presented which is consistent with the clinically relevant Coleman fat grafting technique in which small droplets are transplanted, and a new assessment tool (NMR) is used [Figure 1]. Using this technique allowed measurement of the small fat droplet content in vivo, without the difficulties of manual collection of the dispersed small fat droplets at the end of the study.
Methods
Isolation and preparation of human fat tissue
Fat was harvested from the thigh of a 50-year-old woman undergoing suction-assisted lipectomy under general anesthesia. Prior to commencement of the procedure, the areas of aspiration were injected with a local anesthesia solution containing lidocaine (0.5%) and adrenaline in order to decrease bleeding during fat aspiration and relieve pain after the procedure. The fat was aspirated using a 14-gauge 3-hole blunt cannula, and then processed under sterile conditions for subsequent grafting into nude mice within 2 h of collection. Following aspiration, the fat-containing syringe underwent 2 rounds of centrifugation (1,200 rpm, 10 min at room temperature) and then was placed vertically for 10 min.[14-18]
Between centrifugations, the three different layers (oil, adipose and fluid) were separated. After the last centrifugation, the adipose layer was collected and loaded into 2 mL syringes. All procedures were performed under sterile conditions.[19]
The participant gave her written informed consent, and the study was reviewed and approved by the Helsinki committee of the Rambam Health Care Campus and the institutional review board of the Technion Animal Care and Use Committee.
Animals and study design
The study was composed of nine 7-week-old female CD-1 nude mice (Envigo, Jerusalem, Israel). Seven mice received transplants of human fat, while two mice served as controls without human fat transplantation.
The animals were housed in a specific pathogen-free room, with 1 animal per cage in a room with an artificial 12 h light/dark cycle at a constant temperature range (24 ± 2 ℃) and relative humidity (55 ± 10%). The mice were acclimatized for 5 days prior to the study, and fed standard chow and water ad libitum.
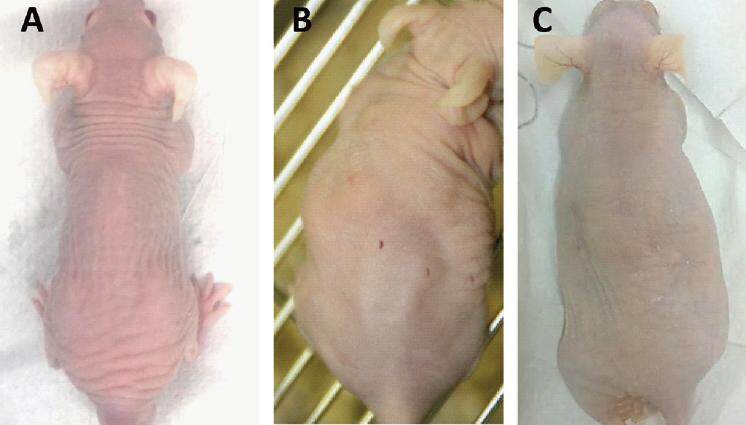
The recipient area of the mouse was disinfected with 70% ethanol. Fat was subsequently slowly injected as small droplets into multiple tunnels in the two lateral sides of 7 nude mice at 1 mL volume per side (for a total of 2 mL per mouse) [Figure 2].
Follow-up and data collection
The duration of the study was 7 weeks starting from the day of human fat transplantation. Before and immediately post-transplantation, and then once weekly following transplantation, the mice were weighed using a standard digital weighing machine and were analyzed using the minispec live mice analyzer (minispec, LF90. Bruker, USA). Seven weeks following human fat transplantation the mice were photo-documented [Figure 2B] and were sacrificed.
NMR analysis
The time-domain NMR (TD-NMR) provides a precise method for in vivo measurements of lean tissue, body fat and body fluid in live mice and rats.
The advantages of the TD-NMR make it an ideal device to measure fat and lean content in the following industries: food, textile, polymer, pharmaceutical and healthcare.
NMR uses a permanent magnetic field and radio frequency energy to examine sensitive nuclei, such as hydrogen and fluorine. The radio frequency signals generated by the nuclei are detected by the device. Each sample’s properties are translated by the minispec as different amplitudes and signal durations.[20] The use of the device is simple and not time consuming [Figure 1]. The miniaturization of the device makes it more accessible and cost-effective when compared to the NMR device used in the clinic.
Statistical analysis
Means and standard deviation were calculated. Differences between means were analyzed for statistical significance using one-way analysis of variance with the Tukey-Kramer multiple comparisons post test (SPSS version 17.0). P values ≤ 0.05 were considered significant.
Results
All 9 mice completed the 7-week study period. They appeared to be healthy and there was no evidence of cachexia during the entire study period. The photographs of the mice before and at the study endpoint clearly demonstrated the resorption of the fat droplets over the 7-week period [Figure 2].
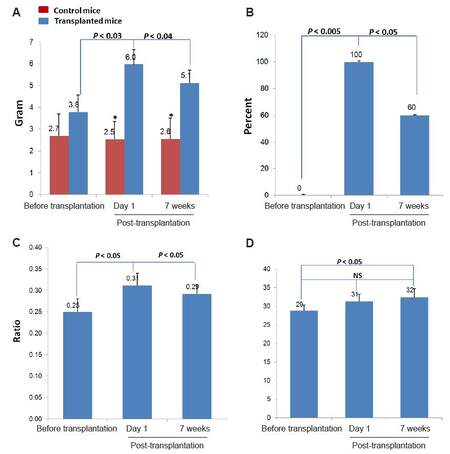
Seven mice were transplanted with human fat. Before fat transplantation, the average fat content, as analyzed by the NMR device, was 3.8 ± 0.8 gr per mouse. One day following transplantation, the fat content increased to 6.0 ± 0.7 gr [Figure 3A].
Figure 3. (A) Fat content as determined by nuclear magnetic resonance (NMR) analysis; (B) percent of fat absorption at the study end point; (C) fat/lean ratio (NMR analysis); (D) standard digital weight assessment of mice at different time points. NS: not significant (as compared to control mice before transplantation)
Seven weeks following transplantation, the average fat content had decreased by 0.9 ± 0.3 gr per mouse to 5.1 ± 0.6 gr (P < 0.04) [Figure 3A], representing 40 ± 18% percent fat resorption as compared to day 1 following transplantation [Figure 3B].
In the control group, 2 mice, not injected with human fat, were measured by the NMR device and demonstrated consistent fat content throughout the study period (2.5 ± 0.9 at day 1, and 2.6 ± 1.0, not significant) [Figure 3A].
The NMR device calculates both lean body mass and fat content. During the 7 weeks following fat transplantation, a decrease was also observed in the fat to lean ratio, as compared to the ratio 1 day following transplantation (0.29 ± 0.02 and 0.31 ± 0.03, respectively, P < 0.05) [Figure 3C]. The decrease of calculated fat content and fat/lean ratio represents an increase of the calculated lean content. Indeed, lean content increased from 15.0 ± 3.0 gr before transplantation to 17.6 ± 1.8 gr following the 7-week experimental period.
The total weight of the mice (as analyzed by a standard digital weighing scale), increased throughout the study from 28.8 ± 1.6 gr before fat transplantation, to 31.3 ± 1.9 gr 1 day following fat transplantation, to 32.4 ± 2.3 gr at the end of the study [Figure 3D]. This elevation was a result of fluid intake and increased lean body mass. The standard digital weighing scale measures total body weight which includes water content, lean body mass, and fat mass. The advantage of the NMR analyzer is in its ability to characterize the water content, lean body mass, and fat mass into separate measurements.
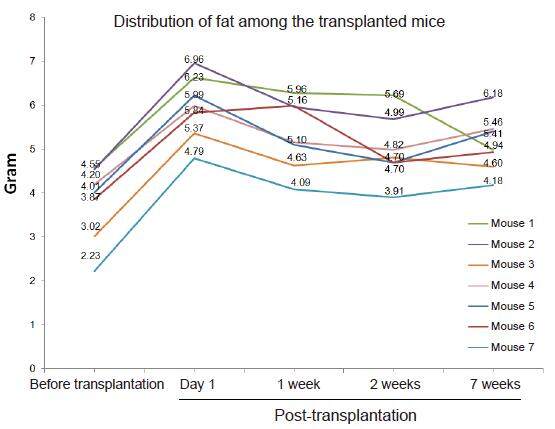
The distribution of the human fat within the tested mice is demonstrated in Figure 4. Although prior to transplantation all mice demonstrated variable fat content with the most lean mouse having 2.2 gr of fat and the most obese mouse having 4.6 gr of fat content (average of 3.7 gr with standard deviation of 0.8), 1 day following fat transplantation the average transplanted fat content (calculated as fat content 1 day post transplantation minus fat content 1 day before transplantation) was much less variable (2.2 gr with standard deviation of 0.2).
Figure 4. A graph demonstrating the change of fat composition at different time points as analyzed by nuclear magnetic resonance technology
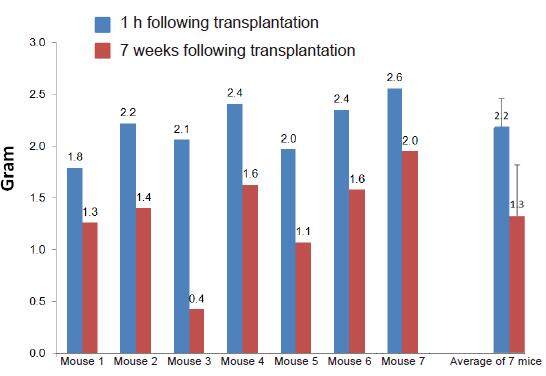
Figure 5 demonstrates a 40% decrease in fat graft weight with an average weight of 2.2 ± 0.3 gr following transplantation and 1.3 ± 0.1 gr 7 weeks following transplantation, as was observed by the NMR analysis. Most mice demonstrated a consistent decrease in fat graft weight (34 ± 7%) with only 1 mouse demonstrating an 80% decrease in fat weight 7 weeks following transplantation.
Discussion
Autologous fat transplantation was first described a century ago, but only in the past decade has it gained attention as an ideal filler for soft tissue reconstruction.[21] Because of the unpredictable outcomes associated with autologous fat transplantation, clinical scientists and physicians have experimented with various methods to find ways to increase the viability of injected fat while reducing its rate of reabsorption.[22] Researchers have evaluated how the fat is harvested, prepared, and injected, and if the fat can be treated with different compounds in order to improve survival. This includes compounds to increase angiogenesis such as erythropoietin, and growth factors including insulin growth factor, fibroblast growth factor, platelet derived growth factor and vascular derived growth factor.[23,24] Other relatively new methods include cell-assisted lipotransfer with adipose-derived stromal cells,[25] and the use of an enriched, serum free cell culture medium (Cariel) as a supplement to the injected fat.[18] Although most of these new methods have slightly improved the take of the transplanted fat, none have shown truly satisfactory and cost-effective results.
The majority of published results on long-term fat survival following transplantation and outcomes are based on subjective analysis of photographs or anecdotal reports.[26,27] In human studies, there is a relative lack of evidence with only a small number of objective studies using three-dimensional imaging, ultrasonography, computed tomography (CT), and magnetic resonance imaging.[28-30]
Small animal models have facilitated more accurate evaluations of fat graft survival and “take”. However, histologic assessment of fat grafts in animals is expensive, time consuming and not amenable to the examination of large volumes of tissue and accurate assessment of fat grafted by use of the Coleman technique. Further, it requires sacrifice of the animal in order for tissues to be harvested for processing, embedding, sectioning, and staining with subsequent analytical evaluation. Traditional CT or micro CT examination is expensive, time consuming (30+ min per animal), requires anaesthetic, and radiates the animal with roughly 30 Gy. Its use is limited as early fat necrosis cannot be easily distinguished from viable fat.
Currently, there are two types of instruments available for non-invasive and sequential ex vivo analysis of small animals: dual energy X ray absorptiometry (DXA) and small animal NMR. The NMR has several advantages over the DXA including the speed and cost of measurements (< 2 min) and the ability to ethically study non-anesthetized animals.[31,32]
NMR is a physical phenomenon which exploits the magnetic properties of certain nuclei to provide detailed structural, dynamic and energetic information of molecular compounds.[33] Although NMR spectroscopy is a powerful technique, application has been limited due to the high cost and complexity of the device. Recently, miniaturized low-field benchtop TD-NMR instruments have been developed. While the newly developed device lacks some degree of sensitivity and resolution, it is capable of powerful relaxation time analysis and has gained popularity due to its simplicity and cost-effectiveness.
However, validation studies of NMR instruments are currently limited and have not been used in fat graft assessment. Taicher et al.[34] examined the precision and accuracy of the NMR (EchoMRI) compared to DXA and chemical carcass analysis. They found higher precision of NMR (vs. DXA) for measurements of fat content in lean and obese mice. In diet-induced obese mice, the coefficient of variation for fat mass by NMR and DXA were 0.34% and 9.59%, respectively.
In order to overcome the inaccuracy of small droplet fat grafting collection and analysis, the use of a whole body composition analyzer based on NMR technology was studied (Brukersminispec, BCA-Analyzer). It is a precise method for measurement of lean tissue, fat and fluid in living mice.
A useful animal model should resemble the human disease, condition or technique, with as much accuracy as possible, yet be readily available and provide reproducible results. A number of such models have been developed primarily with immune deficient rodents.
During the last twenty years our lab has used the nude mouse model for human fat transplantation.[14-18] The athymic nude mouse model was selected because of its limited ability to reject xenografts. This model, which has been previously used for studying fat injection, enables the observation of the take of human fat in an animal model without a confounding immune response leading to graft rejection.
In our prior work human fat was injected as a bolus with a volume of 1 mL and a diameter of approximately 1 cm × 1 cm. The disadvantage of this relatively large fat bolus is necrosis of the adipocytes in the center of the bolus as a result of poor revascularization. Clinicians now inject human fat as small droplets < 0.1 mL, to maximize surface area and thus vascularization.
Indeed, many studies using the Coleman technique have confirmed that small droplets of fat provide better “take” than larger ones, secondary to improved vascularization, nutrient diffusion and less ultimate resorption.[35-37]
The main disadvantage of our previous model was the inability to injected small droplets of human fat due to the difficulty of assessing the fat graft “take” using traditional methods.
Use of the NMR device demonstrated a 53% fat resorption following the seventh week experimental period. This data is in accordance with the clinical and pre-clinical data demonstrating a rate of 20-70% of fat resorption following transplantation.
The accuracy of the NMR device and its suitability for our purposes has been emphasized by the measurements of 2 control mice without human fat transplantation, demonstrating a small and non-significant increase in fat content during the 7 weeks follow-up. In addition, the average delta of 2.2 gr of fat content before and 1 day following fat transplantation demonstrates the accuracy of the NMR device and its relevance to our model.
The conclusion from this study is that the NMR device may serve as a tool for the assessment of the Coleman small droplet fat grafting technique. Until now it was technically impossible to collect small droplets of transplanted fat for volume/weight analysis at the end of the study. Therefore, the fat graft was previously injected as a bolus, which was not clinically applicable. Another advantage of the NMR device is its ability to measures grafted fat without the need to sacrifice the animal, allowing the researcher to measure the fat at different time points during the study.
The advantages of this new model are the ability to analyze small fat droplets in vivo without the necessity for manual excision of the fat. This enables use of the more clinically relevant small fat droplet transplantation technique. This newly improved mouse model will allow researchers and clinicians to test new compounds for minimizing fat resorption.
Authors’ contributions
Data analysis and interpretation: A. Keren
Manuscript preparation: S. Filson
Technical assistance: N. Smirnov-Shalom
Manuscript’s review: A. Gilhar
Concept design, manuscript preparation and data interpretation: Y. Ullmann
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
The participant gave her written informed consent.
Ethics approval
The study was reviewed and approved by the Helsinki committee of the Rambam Health Care Campus and the institutional review board of the Technion Animal Care and Use Committee.
REFERENCES
1. Choi M, Small K, Levovitz C, Lee C, Fadl A, Karp NS. The volumetric analysis of fat graft survival in breast reconstruction. Plast Reconstr Surg 2013;131:185-91.
2. Clauser LC, Tieghi R, Galiè M, Carinci F. Structural fat grafting: facial volumetric restoration in complex reconstructive surgery. J Craniofac Surg 2011;22:1695-701.
3. Coleman SR. Structural fat grafting: more than a permanent filler. Plast Reconstr Surg 2006;118:S108-20.
4. Herold C, Ueberreiter K, Busche MN, Vogt PM. Autologous fat transplantation: volumetric tools for estimation of volume survival. A systematic review. Aesthetic Plast Surg 2013;37:380-7.
5. Ross RJ, Shayan R, Mutimer KL, Ashton MW. Autologous fat grafting: current state of the art and critical review. Ann Plast Surg 2014;73:352-7.
6. Wetterau M, Szpalski C, Hazen A, Warren SM. Autologous fat grafting and facial reconstruction. J Craniofac Surg 2012;231:315-8.
7. Nishimura T, Hashimoto H, Nakanishi I, Furukawa M. Microvascular angiogenesis and apoptosis in the survival of free fat grafts. Laryngoscope 2000;110:1333-8.
8. Coleman SR. Long-term survival of fat transplants: controlled demonstrations. Aesthetic Plast Surg 1995;19:421-5.
9. Ullmann Y, Shoshani O, Fodor A, Ramon Y, Carmi N, Eldor L, Gilhar A. Searching for the favorable donor site for fat injection: in vivo study using the nude mice model. Dermatol Surg 2005;31:1304-7.
10. Shoshani O, Berger J, Fodor L, Ramon Y, Shupak A, Kehat I, Gilhar A, Ullmann Y. The effect of lidocaine and adrenaline on the viability of injected adipose tissue -- an experimental study in nude mice. J Drugs Dermatol 2005;4:311-6.
11. Ramon Y, Shoshani O, Peled IJ, Gilhar A, Carmi N, Fodor L, Risin Y, Ullmann Y. Enhancing the take of injected adipose tissue by a simple method for concentrating fat cells. Plast Reconstr Surg 2005;115:197-201.
12. Ullmann Y, Shoshani O, Fodor L, Ramon Y, Carmi N, Shupak A, Lincoln R, Gilhar A. Long-term fat preservation. J Drugs Dermatol 2004;3:266-9.
13. Shoshani O, Ullmann Y, Shupak A, Ramon Y, Gilhar A, Kehat I, Peled IJ. The role of frozen storage in preserving adipose tissue obtained by suction-assisted lipectomy for repeated fat injection procedures. Dermatol Surg 2001;27:645-7.
14. Filson SA, Keren A, Goldstein N, Ullmann Y. The opposite expected effect of p38 inhibitors on fat graft survival. Plast Reconstr Surg Glob Open 2016;4:e806.
15. Hamed S, Ben-Nun O, Egozi D, Keren A, Malyarova N, Kruchevsky D, Gilhar A, Ullmann Y. Treating fat grafts with human endothelial progenitor cells promotes their vascularization and improves their survival in diabetes mellitus. Plast Reconstr Surg 2012;130:801-11.
16. Shoshani O, Livne E, Armoni M, Shupak A, Berger J, Ramon Y, Fodor L, Gilhar A, Peled IJ, Ullmann Y. The effect of interleukin-8 on the viability of injected adipose tissue in nude mice. Plast Reconstr Surg 2005;115:853-9.
17. Shoshani O, Shupak A, Ullmann Y, Ramon Y, Gilhar A, Kehat I, Peled IJ. The effect of hyperbaric oxygenation on the viability of human fat injected into nude mice. Plast Reconstr Surg 2000;106:1390-6.
18. Ullmann Y, Hyams M, Ramon Y, Beach D, Peled IJ, Lindenbaum ES. Enhancing the survival of aspirated human fat injected into nude mice. Plast Reconstr Surg 1998;101:1940-4.
19. Kurita M, Matsumoto D, Shigeura T, Sato K, Gonda K, Harii K, Yoshimura K. Influences of centrifugation on cells and tissues in liposuction aspirates: optimized centrifugation for lipotransfer and cell isolation. Plast Reconstr Surg 2008;121:1033-41.
20. Pykett IL, Newhouse JH, Buonanno FS, Brady TJ, Goldman MR, Kistler JP, Pohost GM. Principles of nuclear magnetic resonance imaging. Radiology 1982;143:157-68.
21. Jatana KR, Smith SP. The scientific basis for lipotransfer: is it the ideal filler? Facial Plast Surg Clin North Am 2008;16:443-8.
22. Zielins ER, Brett EA, Longaker MT, Wan DC. Autologous fat grafting: the science behind the surgery. Aesthet Surg J 2016;36:488-96.
23. Hamed S, Egozi D, Kruchevsky D, Teot L, Gilhar A, Ullmann Y. Erythropoietin improves the survival of fat tissue after its transplantation in nude mice. PLoS One 2010;5:e13986.
24. Chang L, Wang J, Zheng D, Zhang B, Fan Q, Zhu C, Yu L. Improvement of the survival of autologous free-fat transplants in rats using vascular endothelial growth factor 165-transfected bone mesenchymal stem cells. Ann Plast Surg 2014;72:355-62.
25. Banyard DA, Salibian AA, Widgerow AD, Evans GR. Implications for human adipose-derived stem cells in plastic surgery. J Cell Mol Med 2015;19:21-30.
26. Pu LL. Towards more rationalized approach to autologous fat grafting. J Plast Reconstr Aesthet Surg 2012;65:413-9.
27. Thanik VD, Chang CC, Lerman OZ, Allen RJ Jr, Nguyen PD, Saadeh PB, Warren SM, Levine JP, Coleman SR, Hazen A. A murine model for studying diffusely injected human fat. Plast Reconstr Surg 2009;124:74-81.
28. Fontdevila J, Serra-Renom JM, Raigosa M, Berenguer J, Guisantes E, Prades E, Benito-Ruiz J, Martinez E. Assessing the long-term viability of facial fat grafts: an objective measure using computed tomography. Aesthet Surg 2008;28:380-6.
29. Horl HW, Feller AM, Biemer E. Technique for liposuction fat reimplantation and long-term volume evaluation by magnetic resonance imaging. Ann Plast Surg 1991;26:248-58.
30. Meier JD, Glasgold RA, Glasgold MJ. Autologous fat grafting: long-term evidence of its efficacy in midfacial rejuvenation. Arch Facial Plast Surg 2009;11:24-8.
31. Wang PC, Rosebrough RW, Elsasser TH, Schmidt WF. Assessment of body composition of poultry by nuclear magnetic resonance imaging and spectroscopy. Poult Sci 1991;70:2494-500.
32. Napolitano AW, Wright A, Finer N, De Bruin TW, Bullmore ET, Nunez DJ. Validation of a quantitative magnetic resonance method for measuring human body composition. Obesity (Silver Spring) 2008;16:191-8.
34. Taicher GZ, Tinsley FC, Reiderman A, Heiman ML. Quantitative magnetic resonance (QMR) method for bone and whole-body-composition analysis. Anal Bioanal Chem 2003;377:990-1002.
35. Coleman SR. Facial recontouring with lipostructure. Clin Plast Surg 1997;242:347-67.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Keren A, Filson S, Smirnov-Shalom N, Gilhar A, Ullmann Y. A new tool to assess human fat grafts transplanted into nude mice using a nuclear magnetic resonance device. Plast Aesthet Res 2017;4:97-103. http://dx.doi.org/10.20517/2347-9264.2017.14
AMA Style
Keren A, Filson S, Smirnov-Shalom N, Gilhar A, Ullmann Y. A new tool to assess human fat grafts transplanted into nude mice using a nuclear magnetic resonance device. Plastic and Aesthetic Research. 2017; 4: 97-103. http://dx.doi.org/10.20517/2347-9264.2017.14
Chicago/Turabian Style
Keren, Aviad, Simon Filson, Nadia Smirnov-Shalom, Amos Gilhar, Yehuda Ullmann. 2017. "A new tool to assess human fat grafts transplanted into nude mice using a nuclear magnetic resonance device" Plastic and Aesthetic Research. 4: 97-103. http://dx.doi.org/10.20517/2347-9264.2017.14
ACS Style
Keren, A.; Filson S.; Smirnov-Shalom N.; Gilhar A.; Ullmann Y. A new tool to assess human fat grafts transplanted into nude mice using a nuclear magnetic resonance device. Plast. Aesthet. Res. 2017, 4, 97-103. http://dx.doi.org/10.20517/2347-9264.2017.14
About This Article
Copyright
Author Biographies

Data & Comments
Data
 Cite This Article 4 clicks
Cite This Article 4 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.