The tissue shrinkage phenomenon on surgical margins in oral and oropharyngeal squamous cell carcinoma
Abstract
Aim: One of the most important factors associated with recurrence rate and overall survival is the status of surgical margin of resection free of disease. However, sometimes, the margins measured intra-operatively at the time of surgery differ of those measured by the pathologist in the histopathologic analysis. Faced with this dilemma, a literature review of the best available evidence was conducted in an attempt to determine how the phenomenon of tissue shrinkage may influence on the surgical margin of resection in patients undergoing oral and oropharyngeal squamous cell carcinoma (SCC).
Methods: An electronic and manual search was conducted by one reviewer. A combination of controlled Medical Subjects Headings and keywords were used as search strategy. Inclusion and exclusion criteria were established.
Results: Finally, after an exhaustive selection process, four articles fulfilled the inclusion criteria and were analyzed. All articles reported a decrease of surgical margin after resection. The tumor site and tumor stage seem to influence in degree of margin shrinkage.
Conclusion: Tissue shrinkage on surgical margins of resection in oral SCC is a tangible phenomenon. There is a significant discrepancy between margins measured intraoperatively previous to resection and margins measured by pathologist after histologic processing. The highest percentage of retraction occurs at the time of resection. Margin shrinkage based on tumor site and tumor stage should be considered by any oncologic surgeon to ensure adequate margins of resection cleared of tumor.
Keywords
Introduction
According to the last report of World Health Organization in 2014, cancer of the oral cavity and pharynx constitute, in combination, the seventh most common cancer in the world and the ninth most common cause of death by cancer. Its annual incidence is estimated about 529,000 new cases/year.[1] Two-thirds of theses cases are described in developing countries.[2] The squamous cell carcinoma (SCC), with a high morbidity and mortality, constitutes the most common entity in the upper aerodigestive tract in approximately 90% of cases. Its survival rate at 5 years, for most countries, is around 50%.[2] Numerous clinical and histopathologic factors have been considered as prognostic at the SCC.[3,4] Among them, one of the most relevant, with respect to the overall survival and recurrence rate, is the status of surgical margin of resection free of disease.[5-8] However, although one goal in the oncologic surgery is the complete removal of the tumor with an appropriate margin of security and less aesthetic and functional impact; in the head and neck region, due to its three-dimensional characteristics and the presence of noble structures, the obtaining of appropriate limits of resection constitutes, on occasions, a real challenge for the surgeon.[9,10]
Nevertheless, since 1978 when Looser et al.[5] defined the term “positive surgical margin”, diverse concepts such as “close margins”, “involved margins” or “clear margins”, among others, have been introduced in the literature without a general consensus.[10-13] In fact, even today, there are no universal guidelines that permit different pathologists to adopt the same histologic criteria regarding to surgical margin.[5,14-17] This lack of agreement on what should constitute an “adequate” or “safe” margin of resection[8,18] have led to each pathology department to classify surgical margins according to its own experience or internal guidelines, thereby hampering the comparison of the results obtained in the different studies and its extrapolation to the clinical practice.[14] A recent systematic review concluded that a histopathologic margin of at least 5 mm is the minimum acceptable margin size that should be achieved in any oral SCC.[19] Currently, following the surgical standard, it has been established that a macroscopic surgical margin of 1 to 2 cm obtained intra-operatively is enough extent to obtain a free-tumor margin (5 mm at present) in the oral cavity and oropharynx.[3,19-21] Nevertheless, sometimes it happens that, despite surgical margins measured by the surgeon intra-operatively seem appropriate, a notable discrepancy is observed when are analyzed by the pathologist under the microscope. Faced with this dilemma, a review of the best available evidence in the literature regarding to the tissue shrinkage phenomenon observed on surgical margins of resection in patients with oral and oropharyngeal SCC was carried out.
Methods
An electronic literature search was conducted by one reviewer (D.G.B) in Pubmed (Medline) database, up to January 2016. No language or date restrictions were applied. The population, intervention, control and outcomes[22] (PICO) question that guided this review was as follows: is the effect of tissue shrinkage phenomenon a factor to take into account in the surgical treatment of oral and oropharyngeal squamous cell carcinoma?
Search strategy
A combination of controlled terms Medical Subjects Headings (MeSH) and keywords were used as strategy of search. The search terms used, where “[mh]” represented the MeSH terms and “[tiab]” represented title and/or abstract, were: “carcinoma, squamous cell” [MeSH Terms] OR “mouth neoplasms” [MeSH Terms] OR “oropharynx neoplasm” [MeSH Terms] OR “oral cancer” [Title/Abstract] OR “oral neoplasm” [Title/Abstract] OR “oral tumour” [Title/Abstract] OR “oropharynx cancer” [Title/Abstract] OR “oropharynx tumours” [Title/Abstract] OR “squamous cell carcinoma” [Title/Abstract]) AND (“tissues” [MeSH Terms] OR “tissues” [Title/Abstract] OR “tissue” [Title/Abstract] OR “margin” [Title/Abstract] OR “surgical margin” [Title/Abstract] OR “resection margin” [Title/Abstract]) AND (shrinkage [All Fields] OR “retraction” [Title/Abstract] OR “shrink” [Title/Abstract].
In addition, to identify supplementary articles, the related citations function of Pubmed was used. Likewise, a manual search based on an equivalent search strategy to that used in the Pubmed was performed. Some of the most relevant head and neck, oral and maxillofacial and plastic surgery-related journals were consulted, including: International Journal of Oral and Maxilofacial Surgery; Journal of Oral and Maxillofacial Surgery; Journal of Craniomaxillofacial Surgery; Journal of Maxillofacial Surgery; British Journal of Oral and Maxillofacial Surgery; Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology and Endodontology; Head and Neck; Oral Oncology and Journal of Plastic, Reconstructive and Aesthetic Surgery.
Eligibility criteria
Articles were included in this systematic review if they met the following inclusion criteria: prospective and retrospective studies, cohort, cross-sectional and case-control studies or human clinical trials that discussed: (1) the phenomenon of retraction or tissue shrinkage on surgical margins of resection; (2) the difference between measurements taken in the operation room and those reported by the pathologist on the surgical specimen; or (3) the influence of histopathologic processing on resection margins of surgical pieces in patients underwent surgery for oral or oropharyngeal SCC.
On the contrary, single case reports, animal or in vitro studies, literature reviews, letters, editorial, correspondence or those studies in which phenomenon of shrinkage was not centered on surgical margin were excluded.
Screening process and data extractions
The screening process was conducted by one reviewer (D.G.B). The titles and abstracts were firstly analyzed. The second step consisted of a selection of those articles related with the PICO question. All articles selected were carefully analyzed according to eligibility criteria for future data extraction. From each study included in the review the following information was extracted: first author, year of publication, study design, demographic data (age-range), number of subjects, tumor type, tumor site, tumor stage, margin identification, resection margin, surgical instrument used for resection, measuring instrument, pre-resection measurement, measuring time pre-post resection, mean shrinkage pre-post resection, measurement by stage and measurement by site.
Results
Literature search
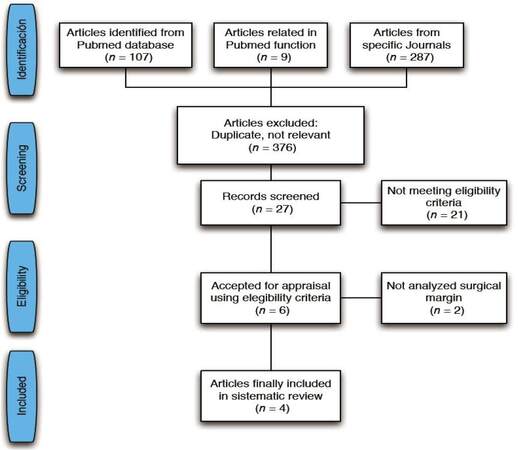
The initial search strategy yielded a total of 107 articles from Pubmed database, 9 articles obtained from related citations of Pubmed and 287 articles from hand- searching. In the first step, titles and abstracts of articles obtained in the strategy search were reviewed for elimination of irrelevant articles (376 articles). In the second step, those articles related with PICO question were analyzed for eligibility (27 articles). Subsequently, a comprehensive and careful reading of the full-text of selected articles was carried out (6 articles). Finally, only 4 articles were included for final data extraction
[Figure 1]. Due the heterogeneity of analyzed studies a qualitative synthesis of the data was performed.
Study characteristics
The characteristics of the studies included in this review are showed in Tables 1 and 2. One prospective study, one retrospective study and other two articles without a clear definition of the type of study design were included. The number of participants in the studies ranged from 35 to 95, and the average age was 59.32 years.
Summary of main characteristics of reviewed studies
| Authors (year) | Study design | Demographic data (No. of subjects; gender; age range; mean age) | Tumor type | Location tumor and No. of patients | Tumor stage and No. of patients |
|---|---|---|---|---|---|
| El-Fole et al.[23] (2014) | Prospective | 61 patients; 39 M (63.9%), 22 F (36.1%); 35-69 years; mean 51.6 years | Oral SCC | Tongue: 20; mucosa alveolar margin mandible: 13; buccal mucosa: 15; retromolar trigone: 6; floor of the mouth: 3; mucosa alveolar margin maxilla: 4 | T1: 4; T2: 47; T3: 5; T4: 5 |
| Egemen et al.[24] (2014) | NR | 21 patients; 14 M (66.6%), 7 F (33.3%); 47-92 years; mean 71.1 years | Lip SCC | Lower lip: 15; Upper lip: 5; Commisure: 1 | T1: 8; T2: 10; T3: 3 |
| Cheng et al.[20] (2008) | NR | 41 patients; 21 M (51%), 20 F (49%); 35-95 years; mean 67 years | Oral SCC | Group 1: buccal mucosa, mandibular alveolar ridge and retromolar trigone - 21; Group 2: maxilar alveolar ridge and hard palate - 6; Group 3: oral tongue - 14 | T1: 11; T2: 16; T3: 1; T4: 11; 2 patients excluded |
| Mistry et al.[16] (2005) | Retrospective | 27 patients; 18 M (66.6%), 9 F (33.3%); 36-61 years; mean 47.6 years | Oral SCC | Oral tongue - 16; buccal mucosa - 11 | T1: 11; T2: 11; T3: 3; T4: 2 |
Summary of the measurements made in the analyzed studies
| Authors (year) | Margin identification; surgical instrument; resection margin; measuring instrument | Surgical measurement | Measuring time, pre-post resection | Mean shrinkage | Shrinkage by site | Shrinkage by stage |
|---|---|---|---|---|---|---|
| El-Fole et al.[23] (2014) | Margin ink/sutures; electocautery; 1 cm; metric ruler or caliper | In situ/pre-resection; immediately post-resection | NR | 47.6% buccal mucosa; 33.3% tongue; 9.5% mandibular alveolus; 4.8% floor of mouth; 4.8% retromolar trigon | 66.7% buccal mucosa; 35% tongue; 33.3% floor of mouth; 15.4% mandibular alveolus; 16.7% retromolar trigon | NR |
| Egemen et al.[24] (2014) | Margin ink; not indicated; 1 cm; metric ruler/water flooding | In situ/pre-resection; immediately post-resection | At 24 h and 48 h of fixation (volume, tumor length and distance); After 48 h of fixation - standardized fashion | 41-47% volume decrease; 21.8% | NR | NR |
| Cheng et al.[20] (2008) | Margin ink; not indicated; 1 cm; metric ruler | In situ/pre-resection | NR | 59.02% | 71.90% Group 1; 53.33% Group 2; 41.14% Group 3 | T1/T -51.48%; T3/T -75% |
| Mistry et al.[16] (2005) | Margin ink/sutures; electrocautery; 1 cm; caliper | In situ/pre-resection | Half an hour post-resection | 22.7 % | 23.5% tongue; 21.2% buccal mucosa | T1-T2-25.6%; T3-T4-9.2% |
Three articles studied the discrepancy between “in situ” margins and “histopathologic” margins in patients underwent surgery for oral SCC, while one study analyzed the effect of tissue shrinkage on surgical margins of resection in patients operated for lip SCC. In all analyzed studies, the appropriate surgical margin of resection was defined as that margin located 1 cm from the tumor border; likewise, the resection margin was identified by mean of marking ink in all examined articles and only 2 of them reported the use of sutures as an additional means of margins identification. Only 2 studies indicated the instrument utilized for resection (electrocautery). The extent of the tumor was, in all cases, determined by visual inspection and palpation. With regards to the measuring instruments used to assess the discrepancy between “in situ” margins and “histologic” margin, 3 studies reported the use of the metric ruler as measuring instrument, 1 study described the caliper as the instrument utilized in the measurements and 1 study informed the use of both instruments for measuring of the margins. All articles reported a measurement of surgical margin prior to the surgical resection, but only 2 of them indicated the time until the measurement recorded by the pathologist (one study at 24-48 h postresection and another study at half an hour after resection).
A statically significant discrepancy between margins measured at the time of the surgery and margins measured after histopathologic processing was observed in all studies analyzed. Thus, Mistry et al.[16] reported a mean shrinkage from the pre-resection to the post-resection measurement of 3.18 mm (22.7%) (P < 0.011). In their study, Cheng et al.[20] informed a mean discrepancy between the in situ margins and the histopathologic margins for all patients of 59.02% (P < 0.001). However, El-Fol et al.[23] described a mean discrepancy between intraoperative margins and histopathologic margins exclusively analyzing all close and positive margins. In this study, the mean discrepancy for buccal mucosa was 47.6%, 33.3% for the tongue, 9.5% for the mandibular alveolus, and 4.8% for both, retromolar trigon and floor of the mouth. Finally, the study of Egemen et al.[24] based on the surgical margins of the resected lip specimens, reported a mean decrease of up to 41-47.5% in the length and of 21.8% in volume between measurements performed before the resection and those obtained in the histopatologic study.
Shrinkage depending on the tumor site
The degree of shrinkage based on the different tumor sites was analyzed by three studies. Mistry et al.[16] published a study on 27 patients with oral SCC of the tongue and buccal mucosa of the oral cavity where examined the distances pre-resection and post-resection. They reported a greater discrepancy of the tongue margins (23.5%) that the buccal mucosa margin (21.2%) and a mean loss of 22.7%, however, these results were not statistically significant. In the study of Cheng et al.[20] on 41 patients with diagnosis of oral SCC the amount of margin discrepancy between margins measured intraoperatively and those measured microscopically was quantified. The patients were grouped by locations obtaining the following statistically significant result: mean discrepancy for group 1 (buccal mucosa, mandibular alveolar ridge and retromolar trigone) 71.90%, 53.33% for group 2 (maxillary alveolar ridge and palate) and 42.14% for group 3 (oral tongue), with a P value corresponding to 0.0133. Likewise, El-Fol et al.[23] measured the difference between the “in situ” margins and “histopathologic” margins of 61 patients that underwent resective surgery for oral SCC. A significant difference in the measurement of resection margin according to the anatomical site was obtained with a mean of discrepancy of 66.7% for buccal mucosa, a 35% for the tongue, a 33.3% for the floor mouth, a 16.7% for the retromolar trigone and a 15.4% for the mandibular alveolus.
Shrinkage depending on the tumor stage
The percentage of discrepancy in the different studies analyzed according to the tumor stage was described in only two studies.
The study of Mistry et al.[16] compared the mean shrinkage of patients with lower stage tumors (T1 and T2) with the mean shrinkage in patients with higher stage tumors (T3 and T4). The difference between the two groups was statistically significant (P < 0.011), with a mean of 3.59 mm (25.6%) for T1/T2 tumors vs. 1.4 mm (9.2%) for T3/T4 tumors, respectively. However, these results were different to the study presented by Cheng et al.[20] where the mean of discrepancy for T1/T2 tumor was 51.48%, and 75% for T3/T4 tumors (P = 0.0264).
Discussion
One of the most important prognostic factors respect to overall survival and local recurrence rates is the status of surgical margins of resection.[5,25] Indeed, the main goal of the resective surgery of the head and neck is the complete removal of the tumor with suitable margins of resection free of disease.[23] However, even at the present day, there has not been consensus between researches on what constitutes tumor involvement at the resection margin (including mucosal dysplasia or carcinoma in situ) and what constitutes an “adequate” margin of resection.[7,8,18] Though controversial, it seems reasonable to accept, based on studies, that 5 mm of healthy tissue around the tumor should be the minimum acceptable margin size for a clear surgical margin in any oral SCC.[7,19]
Nevertheless, it sometimes happens that, the surgeon feels frustration when noticing that an appropriate surgical margin in the operation room presents a considerably decrease in size when is observed by the pathologist. In such cases, it is not surprising that a surgical margin that seems appropriate intra-operatively can be reported as positive or affected in the final histopathologic analysis. Diverse explanations have been considered in the literature. Thus, the invasive character of oral SCC can lead to occult microscopic margins, finger extensions or islands of tumor that extend beyond the clinically visible and palpable tumor, obtaining a margin that is closer than previously expected.[20,23] Moreover, it should be kept in mind that malignant molecular changes may be present even when there are histopathologic normal margins.[26] Nevertheless, it seems clear that the discrepancy observed between clinical and pathological margins is most often associated to shrinkage phenomenon after resection.[23]
The aim of this literature review was to identify studies that discussed the tissue shrinkage phenomenon on surgical margins of resection in patients underwent surgery for oral and oropharynx SCC. Only four articles were finally included in this review according to our search strategy (one prospective, one retrospective and two articles not defined). All of them reported a discrepancy between surgical margins measured intra-operatively and those margins of resection measured by the pathologist after processing of the surgical piece. These findings are consistent with those reported by others authors that observed the phenomenon of margin shrinkage at other places of the body.
Thus, in a study by Silverman et al.[27] on 199 cutaneous malignant melanoma reported of a shrinkage of a 15 to 25% on margins of surgical specimens depending on the patient’s age. Likewise, Weese et al.[28] observed in ten patients who underwent colonic resection that resected rectal margin could shrink up to 50% or more after processing histologic of surgical piece. Siu et al.[29] noted in a study on esophagus carcinoma that exist a different degree of shrinkage of the entire specimen from its surgical resection to its final pathological study. The surgical specimen shrank a 40% following resection and another 10% after formalin fixation.
However, the first reference regarding to the study of tissue shrinkage on surgical margins of resection in oral cavity and oropharynx is attributed to Johnson et al.[9] in 1997. In their experimental study on ten mongrel dogs they reported that a shrink of up to 30-50% may be expected in the specimens of oral cavity and oropharynx and the maximum shrinkage occurs immediately after the resection. These results are similar to those of the articles analyzed in this review of human study. In fact, a conclusion shared by all the authors is that specimens of oral SCC are significantly reduced after surgical resection.[16,20,23,24]
Thus, Mistry et al.[16] published a study on 27 patients with oral SCC of the tongue and buccal mucosa where analyzed the distances pre-resection and post-resection and reported a mean shrinkage of 3.18 mm (22.7%). However, El-Fol et al.[23] described a mean discrepancy between intraoperative margins and histopathologic margins exclusively analyzing all close and positive margins. In this study, the mean discrepancy for buccal mucosa was 47.6%, 33.3% for the tongue, 9.5% for the mandibular alveolus, and 4.8% for both, retromolar trigone and floor of the mouth.
In the study of Cheng et al.,[20] the mean discrepancy between the in situ margins and the histopathologic margins for all patients was reported statistically significant at 59.02% (P < 0.001). One conclusion of the authors for these findings is that the specimens of the oral cavity retract significantly after resection and subsequently after pathologic processing.
Likewise, Egemen et al.[24] in their study on surgical margins of the resected lip specimens, observed a mean decrease of up to 41-47.5% in length and of 21.8% in volume between measurements obtained before the resection and those reported in the histopathologic study. Moreover, they noted that the most significant step for shrinkage phenomenon was the excision step followed by the formalin fixation step and also noted that the duration of the fixation did not affect the shrinkage rate of surgical margins; however, the volume of the specimen was decreased in higher proportion at 48 h compared with 24 h of fixation.
An interesting fact to consider is whether the tumor site influences on the degree of tissue shrinkage. Certain intrinsic factors, such as tissue composition, have been studied as responsible of different shrinkage percentages at different locations in the given specimen. It has been observed that intra-tumoral shrinkage is less as is compared to the shrinkage at surgical margins. Even if different surgical margins of a single specimen are from the same location of the oral cavity, shrinkage of each margin may vary. The reason, according to some authors, could be diverse: presence of varying number of tumor cells underneath surgical margin, cohesiveness of tumor cells, degree of keratinization, degree of inflammation, variable susceptibility of invasion or inclusive heterogeneous biology among other.[20,30]
In this regard, Mistry et al.[16] reported a greater discrepancy of the tongue margins (23.5%) that the buccal mucosa margin (21.2%), however, these results were not statistically significant. They concluded that factors like age, gender or site of tumor do no significantly affect the quantum of shrinkage. On the other hand, in the study of Cheng et al.[20] where the patients were grouped by locations, the mean of discrepancy obtained for group 1 (buccal mucosa, mandibular alveolar ridge and retromolar trigone) was 71.90%, 53.33% for group 2 (maxillary alveolar ridge and palate) and 42.14% for group 3 (oral tongue), with a P value corresponding to 0.0133. Likewise, El-Fol et al.[23] found a significant difference in the measurement of resection margin according to the anatomical site. In this study the mean of discrepancy observed for buccal mucosa was of 66.7%, 35% for the tongue, a 33.3% for the floor mouth, a 16.7% for the retromolar trigon and a 15.4% for the mandibular alveolus. Given these results, it seems appropriate to believe that knowing shrinkage of each margin according to its location instead of shrinkage of the specimen, is what should guide treatment guidelines.[30]
When comparing margins discrepancies based on staging, Cheng et al.[20] described a mean discrepancies in T1/T2 tumors of 51.48% and in T3/T4 tumors of 75% with a P value of 0.0264. These findings differed from those presented by Mistry et al.[16] who observed a mean shrinkage of 3.59 mm (25.6%) for T1/T2 tumors and 1.4 mm (9.2%) for T3/T4 tumors with a difference statistically significant (P < 0.011). In this study, the authors hypothesized that late stage tumors may show a smaller discrepancy due to tumor related destruction of contractile elements surrounding cancer. However, Cheng et al.[20] tried to explain this discrepancy alluding that late stage tumors result in a greater microscopic invasiveness and that the small number of cases presented by Mistry et al.[16] could be considered an artifact. As we can see in view of the results obtained, the degree of discrepancy between the pre and post surgical resection margins based on tumor staging does not obtain extrapolated conclusions mainly because only two studies, whose tumor sites clearly differ from each other, were analyzed.
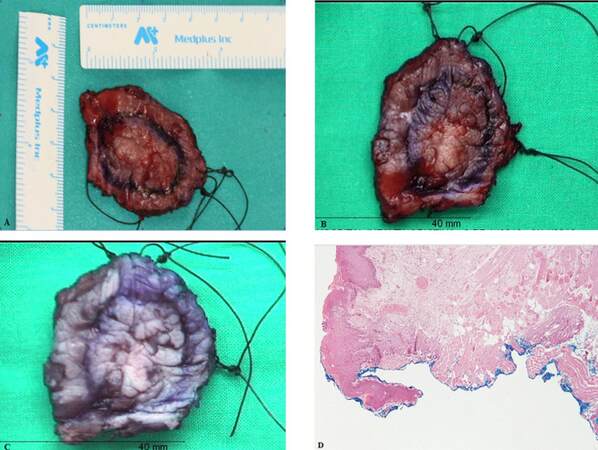
The results obtained in this review related to the phenomenon of tissue shrinkage on the surgical margin of resection in patients with oral SCC, coincide with the results observed by the author and his team. In our study, pending on its recent publication, patients diagnosed of oral and oropharyngeal squamous cell carcinoma and that underwent surgery with reconstruction by means of microsurgical techniques, the analysis of surgical resection margins was performed in several stages: pre-resection, immediately post-resection in the operating room, in fresh in the pathology department, after fixation by the pathologist and under microscope [Figure 2]. In anticipation of the results, only indicate that a large discrepancy between the margins intraoperatively measured and the margins microscopically analyzed was observed, with an average of 4.46 mm. The step in which a smaller reduction of surgical margins was observed coincide with that reported in this review, the step from formalin fixation to the microscope analysis, with an overall mean of 0.68 mm.
Figure 2. Brief summary of the measurements taken during the different phases of the study performed by the authors' team. (A) intraoperative measurement in fresh; (B) measurement in fresh by the pathologist; (C) measurement after fixation with formaldehyde; (D) measurement under microscope (C) in a patient with oral squamous cell carcinoma - T1N0M0
Study limitations
The author of this paper recognizes the limitations inherent to this review. The study is limited to one database (Pubmed) and only four articles were included in the final study according to our search strategy. The scarce number of patients observed by staging or by site in some articles, the variety of study designs, the absence of measurement post-resection immediately in the operation room in two of the studies, together to the fact that not all articles explain the surgical instruments used for tumor resection or how long was from resection of surgical specimen to the measurement by the pathologist after histopathologic processing, make that studies be quite heterogeneous as to obtain any type of quantitative results. Therefore, the conclusions achieved have to be taken with caution before its application to clinical practice. Maybe, future studies involving a largest number of patients by stage or locations can provide more reliable information for application to clinical routine.
In conclusion, tissue shrinkage on surgical margins of resection in oral SCC is a tangible phenomenon. The highest percentage of retraction occurs at the time of resection. Tumor staging should be established intra-operatively and no following histopathologic processing when the tissue shrinkage phenomenon is already established. The surgeon should take the tissue shrinkage phenomenon into account when affording surgical resection, while his/her actuation must be based on tumor site and stage in order to provide adequate definitive tumor margins.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Stewart BW, Wild CP. World Cancer Report 2014. World Health Organization: Geneva; 2014.
2. Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol 2009;45:309-16.
3. Massano J, Regateiro FS, Januário G, Ferreira A. Oral squamous cell carcinoma: review of prognostic and predictive factors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006;102:67-76.
4. Spiro RH, Guillamondegui O Jr, Paulino AF, Huvos AG. Pattern of invasion and margin assessment in patients with oral tongue cancer. Head Neck 1999;21:408-13.
5. Looser K, Shah J, Strong E. The significance of "positive" margins in surgically resected epidermoid carcinomas. Head Neck Surg 1978;1:107-11.
6. Yahalom R, Dobriyan A, Vered M, Talmi YP, Teicher S, Bedrin L. A prospective study of surgical margin status in oral squamous cell carcinoma: a preliminary report. J Surg Oncol 2008;98:572-8.
7. Kurita H, Nakanishi Y, Nishizawa R, Xiao T, Kamata T, Koike T, Kobayashi H. Impact of different surgical margin conditions on local recurrence of oral squamous cell carcinoma. Oral Oncol 2010;46:814-7.
8. Hinni ML, Ferlito A, Brandwein-Gensler MS, Takes RP, Silver CE, Westra WH, Seethala RR, Rodrigo JP, Corry J, Bradford CR, Hunt JL, Strojan P, Devaney KO, Gnepp DR, Hartl DM, Kowalski LP, Rinaldo A. Surgical margins in head and neck cancer: a contemporary review. Head Neck 2013;35:1362-70.
9. Johnson RE, Sigman JD, Funk GF, Robinson RA, Hoffman HT. Quantification of surgical margin shrinkage in the oral cavity. Head Neck 1997;19:281-6.
10. McMahon J, O'Brien CJ, Pathak I, Hamill R, McNeil E, Hammersley N, Gardiner S, Junor E. Influence of condition of surgical margins on local recurrence and disease-specific survival in oral and oropharyngeal cancer. Br J Oral Maxillofac Surg 2003;41:224-31.
11. Nason RW, Binahmed A, Pathak KA, Abdoh AA, Sándor GKB. What is the adequate margin of surgical resection in oral cancer? Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009;107:625-9.
12. Woolgar JA, Triantafyllou A. A histopathologic appraisal of surgical margins in oral and oropharyngeal cancer resection specimens. Oral Oncol 2005;41:1034-43.
13. Brandwein-Gensler M, Teixeira MS, Lewis CM, Lee B, Rolnitzky L, Hille JJ, Genden E, Urken ML, Wang BY. Oral squamous cell carcinoma: histologic risk assessment, but not margin status, is strongly predictive of local disease-free and overall survival. Am J Surg Pathol 2005;29:167-78.
14. Tirelli G, Zacchigna S, Biasotto M, Piovesana M. Open questions and novel concepts in oral cancer surgery. Eur Arch Otorhinolaryngol 2015; Epub ahead of print.
15. Chiou WY, Lin HY, Hsu FC, Lee MS, Ho HC, Su YC, Lee CC, Hsieh CH, Wang YC, Hung SK. Buccal mucosa carcinoma: surgical margin less than 3 mm, not 5 mm, predicts locoregional recurrence. Radiat Oncol 2010;5:79.
16. Mistry RC, Qureshi SS, Kumaran C. Post-resection mucosal margin shrinkage in oral cancer: quantification and significance. J Surg Oncol 2005;91:131-3.
17. Loree TR, Strong EW. Significance of positive margins in oral cavity squamous carcinoma. Am J Surg 1990;160:410-4.
18. Weinstock YE, Alava I, Dierks EJ. Pitfalls in determining head and neck surgical margins. Oral Maxillofac Surg Clin North Am 2014;26:151-62.
19. Anderson CR, Sisson K, Moncrieff M. A meta-analysis of margin size and local recurrence in oral squamous cell carcinoma. Oral Oncol 2015;51:464-9.
20. Cheng A, Cox D, Schmidt BL. Oral squamous cell carcinoma margin discrepancy after resection and pathologic processing. J Oral Maxillofac Surg 2008;66:523-9.
21. Upile T, Fisher C, Jerjes W, El Maaytah M, Searle A, Archer D, Michaels L, Rhys-Evans P, Hopper C, Howard D, Wright A. The uncertainty of the surgical margin in the treatment of head and neck cancer. Oral Oncol 2007;43:321-6.
22. Schardt C, Adams MB, Owens T, Keitz S, Fontelo P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform Decis Mak 2007;7:16.
23. El-Fol HA, Noman SA, Beheiri MG, Khalil AM, Kamel MM. Significance of post-resection tissue shrinkage on surgical margins of oral squamous cell carcinoma. J Craniomaxillofacial Surg 2015;43:475-82.
24. Egemen O, Bingöl D, Orman Ç, Sayilgan AT, Özkaya Ö, Akan M. Quantification of the surgical margin shrinkage in lip cancer: determining the relation between the surgical and histopathologic margins. J Craniofac Surg 2014;25:2152-5.
25. Ravasz LA, Slootweg PJ, Hordijk GJ, Smit F, van der Tweel I. The status of the resection margin as a prognostic factor in the treatment of head and neck carcinoma. J Craniomaxillofac Surg 1991;19:314-8.
26. van Houten VM, Leemans CR, Kummer JA, Dijkstra J, Kuik DJ, van den Brekel MW, Snow GB, Brakenhoff RH. Molecular diagnosis of surgical margins and local recurrence in head and neck cancer patients: a prospective study. Clin Cancer Res 2004;10:3614-20.
27. Silverman MK, Golomb FM, Kopf AW, Grin-Jorgensen CM, Vossaert KA, Doyle JP, Levenstein MJ. Verification of a formula for determination of preexcision surgical margins from fixed-tissue melanoma specimens. J Am Acad Dermatol 1992;27:214-9.
28. Weese JL, O'Grady MG, Ottery FD. How long is the five centimeter margin? Surg Gynecol Obstet 1986;163:101-3.
29. Siu KF, Cheung HC, Wong J. Shrinkage of the esophagus after resection for carcinoma. Ann Surg 1986;203:173-6.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
González-Ballester D. The tissue shrinkage phenomenon on surgical margins in oral and oropharyngeal squamous cell carcinoma. Plast Aesthet Res 2016;3:150-7. http://dx.doi.org/10.20517/2347-9264.2016.23
AMA Style
González-Ballester D. The tissue shrinkage phenomenon on surgical margins in oral and oropharyngeal squamous cell carcinoma. Plastic and Aesthetic Research. 2016; 3: 150-7. http://dx.doi.org/10.20517/2347-9264.2016.23
Chicago/Turabian Style
González-Ballester, David. 2016. "The tissue shrinkage phenomenon on surgical margins in oral and oropharyngeal squamous cell carcinoma" Plastic and Aesthetic Research. 3: 150-7. http://dx.doi.org/10.20517/2347-9264.2016.23
ACS Style
González-Ballester, D. The tissue shrinkage phenomenon on surgical margins in oral and oropharyngeal squamous cell carcinoma. Plast. Aesthet. Res. 2016, 3, 150-7. http://dx.doi.org/10.20517/2347-9264.2016.23
About This Article
Special Issue
Copyright
Author Biographies

Data & Comments
Data
 Cite This Article 8 clicks
Cite This Article 8 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.