Collagen membrane for reconstruction of soft tissue defects after surgery of oral cancer and precancer: a brief review
Abstract
There are some surgeries after which a temporary cover for raw wounds is required to ensure healing. Some of those circumstances are loss of tissue due to burns, trauma, amputation, chronic ulcer, leprosy, and skin graft sites. Although the body initiates regeneration mechanisms, however the time taken for complete healing of wounds is unpredictable. Also, there is a tendency for long standing wounds to undergo infection and scarring. Oral mucosa is no exception to scarring and infection of wounds and there has always been a search for new materials that can be used for coverage of oral defects. Xenogenous collagen is one such grafting material. Over the years collagen implant solutions for a number of clinical applications include general surgery, burn surgery, neurosurgery, plastic and reconstructive surgery, oral surgery, and peripheral nerve and tendon surgery. This paper aims to focus on collagen as an effective option of wound closure in plastic and reconstructive surgery of the head and neck, especially after loss of soft tissue following resection of oral malignancies.
Keywords
Introduction
Oral mucosa is predisposed to a variety of pathologies such as leukoplakia, oral submucous fibrosis, oral squamous cell carcinoma; the treatment of which may lead to significant soft tissue loss that may not be amenable to primary closure. According to Ashley’s principles of plastic surgery, covering the raw wound is necessary to prevent infection, tissue contracture and scarring.[1] A variety of approaches have been used in the past, including split and full-thickness skin grafts, oral mucosa free grafts, oral connective tissue grafts, the latest being tissue engineered grafts.[2] For larger defects, pedicled local and distant flaps or free microvascular grafts have been tried with variable success rates.[2]
Non-vascularized free graft materials like split thickness skin graft, human amniotic membrane, palatal mucosa, buccal mucosa and collagen are easy to obtain, but each material has its own limitations.[1,3-5] One of these materials is collagen, which is in extensive use as temporary dressing material in a lot of surgical fields. This paper aims to highlight the various types of collagen, its manipulation and applications in the field of oral cancer and precancer.
Collagen as a biomaterial in plastic and reconstructive surgery
Collagen is the major insoluble protein (fibrous protein) in the extra-cellular matrix and in connective tissues. More than 80% of the skin is composed of collagen. It is also the main component of the ligaments and tendons. In the early 1970’s, John F. Burke and Ioannas Yannas developed a bio-compatible collagen matrix to improve wound healing. It is commonly used in the management of burns,[6] diabetic foot ulcer,[7,8] toxic epidermal necrolysis,[9] chronic wounds, etc.[10-12] This versatile material also finds its application in the field of plastic surgery, oral and maxillofacial surgery and dentistry for various purposes e.g. as an interpositional graft material during palatoplasty;[13] for guided bone regeneration during maxillary sinus lift;[14] for inducing bone formation along with/without certain medicaments like gusuibu;[15,16] bone augmentation of posterior atrophic mandibular ridge for placement of dental implants;[17] as a reconstructive material for orbital floor fractures;[18] in treatment of localized gingival recession;[19] as a scaffold in tissue engineering to generate dental pulp;[20] for coverage of small intraoral soft tissue defects of the oral cavity[21] and much more. Collagen also has use as a medium for culturing cells such as osteoblasts.[22]
Biological dressings are the logical best candidate for the management of wounds since they create the most physiological interface between the wound surface and the environment.[23] Collagen is a biological skin substitute, i.e. natural, easily available, ready to use, non-immunogenic, and non-pyrogenic. Today, a variety of collagen sheets are available with or without carriers, e.g. gels, pastes, polymers, oxidized regenerated cellulose, and ethylene diamine tetraacetic acid. This collagen may be derived from bovine, porcine, equine, or avian sources, which is purified to make it nonantigenic. Collagen dressings are made of either type I (native) or denatured collagen and they can come in a variety of pore sizes and surface areas. Many collagen dressings contain an antimicrobial agent to control pathogens within the wound.[24]
According to Lazovic et al.[7] the physical properties of collagen sheet can be divided into two categories, i.e. biological, physiological. The biological properties include its non-inflammatory nature, low antigenicity, no toxicity and minimal biodegradation. It facilitates migration of fibroblasts and microvascular cells and helps in the synthesis of neodermal collagen matrices. Collagen sheet al so helps in minimizing scarring. The physiological properties of collagen are its non-permeability to bacterial migration, modulation of fluid flux from the wound, elasticity, softness and suppleness, good tear strength, good suturing characteristics and enough strength to be peeled off the wound. Collagen sheet has been found to be well tolerated in clinical trials.[7] There have been no reports of clinically significant immunological or histological responses to the implementation of collagen sheet that could cause its rejection.[7]
Research shows that some collagen-based dressings can produce a significant increase in the fibroblast production; can promote fibroblast permeation by the virtue of their hydrophilic nature; can cause increased deposition of oriented and organized collagen fibers by attracting fibroblasts and causing a directed migration of cells; can help in the uptake and bioavailability of fibronectin; can cause preservation of leukocytes, macrophages, fibroblasts, and epithelial cells; and assist in the maintenance of the chemical and thermostatic microenvironment of the wound.[24]
Collagen makes large areas of ulcerated skin pain free. The use of collagen dressing has been found to inhibit the action of metalloproteinases, hence it is useful in treating chronic wounds.[25] Collagen is a biomaterial that encourages wound healing through deposition and organisation of freshly formed fibres and granulation tissue in the wound bed, thus creating a good environment for wound healing.[26] Cutaneous lesions can bleed due to shearing of the gauze dressings. Application of collagen sheet to such wounds, not only promote angiogenesis, but also aid in body's repair mechanisms.[7,27] Collagen sheets act as a mechanical support, reduce oedema and loss of fluids from the wound site, facilitate the of migration of fibroblasts into the wound, thus enhancing the metabolic activity of the granulation tissue.[27-29] This fact is of particular importance in treating painful and bleeding ulcers of the mucosal surfaces of the body such as buccal cavity, nasal cavity, conjunctiva, urethra, vagina, and anal canal.
Collagen for coverage of soft tissue defects of oral mucosa secondary to resection of precancerous and cancerous lesions
Cancer is one of the main causes of death in all societies, its relative position varying with age and sex.[30] Globally, oral cancer is the sixth most common cause of cancer related- death, although many people are unaware of its existence.[30] In the International Classification of Diseases [9th revision-World Health Organization (WHO)], oral cancer is classified under the rubrics 140 (lip), 141 (tongue), 143 (gingiva), 144 (floor of the mouth), and 145 (other parts of the mouth). Oral precancer is distinguished by WHO into “precancerous lesions” (e.g. leukoplakia, erythroplakia) and “precancerous conditions” (e.g. oral sub mucous fibrosis, lichen planus).[31] The treatment of these suspicious precancerous lesions involve wide excision followed by grafting of the surgical sites. Depending on the status of metastasis of established malignancies, resection of affected area and radical neck dissection followed by adjuvant radiotherapy with or without chemotherapy may be required. In the surgery of oral submucous fibrosis (OSMF), bilateral fibrotic bands are excised with or without bilateral coronoidectomy or temporalis myotomy.
In all such cases, wounds left uncovered are prone to infection, contraction, and scarring with other clinical complications. Raw wounds of the oral cavity, like any other wounds, heal by epithelialization and granulation. However, in the oral cavity the healing of raw wounds presents some special problems. The environment is always moist with contamination from salivary secretion and food ingestion. This, compounded by poor oral hygiene and constant movements of the cheek and tongue and masticatory forces, may interfere with graft adherence and acceptance. The risk of infection in the oral cavity is also quite high, which may result in scarring and contraction. The oral cavity is highly sensitive to any residual scarring, which may undergo ulceration and could be a constant source of irritation to patients wearing dentures. Hence, a need arises to use a biologic cover to prevent these complications.[32]
Free split-skin graft and free mucosal graft have been used to cover raw wounds in the oral cavity. The use of these grafts required a separate surgical procedure with associated technical difficulties. The color and texture of skin do not conform totally to the oral cavity. Also seen is the growth of adnexal structures such as hair and sweat glands. In elderly persons the skin is atrophic and inelastic, making it unsuitable. Mucosal grafts offer the best solution because they come nearest to fulfilling the requirements of an ideal graft material, which include the ability to replace lost structures and the ability to induce the formation of such tissues. Donor sites for mucous graft are limited, and there is always morbidity associated with donor-site healing. The oral environment and its constant movements are impediments to graft acceptance.[32] Other reconstructive options which have been used in the past include nasolabial flaps, transposition of the buccal pad of fat, dorsal tongue flap, radial forearm flaps, flaps of the temporalis fascia/muscle or both, palatal island flaps to cover surgical defects, each having their own advantages and shortcomings.[33,34] Use of island palatal flap has limitation such as its involvement with fibrosis and second molar tooth extraction is required for flap cover without tension. Bilateral palatal flaps leave a large raw area on palatal bones. Sometimes the defect created may be large and local flaps may not be able to cover the entire defect. A nasolabial flap is too short to cover the defect and causes visible scaring on the face and requires division at second stage. Tongue flaps have been used to cover the buccal defects but were found to be bulky and needed additional surgery for detachment. Bilateral tongue flaps can cause severe dysphagia and disarticulation and carry the risk of postoperative aspiration. Pindborg et al.[1] found an incidence of 38% tongue involvement in OSMF, which precludes its use. Bilateral free radial artery forearm free flaps require micro vascular expertise, the flaps are hairy and 40% of patients require secondary de-bulking procedures. Extraction of third molar tooth is required to avoid flap inclination between teeth.
Chemically, bovine collagen is very similar to the human form. This is crucial, as the human immune system will reject everything that deviates too much from its own proteins. For these reasons, collagen sheets are well qualified for use as an effective wound cover.[7] Bovine collagen contain mostly type I and III collagen, packed in a neutral glass vial of sterile preservative mixture of isopropyl alcohol and water sterilized with ethylene oxide and are available in different sizes for clinical application. The freeze-dried form of collagen is also available so that there is no need to treat the membrane in normal saline before application. Meshed collagen membrane in wet form, porous collagen dressing and collagen film dressings are also available.[6] Wet collagen membrane comes in varying in dimensions of 10 cm × 10 cm, 10 cm × 25 cm and 25 cm × 25 cm with thickness of 0.6 mm.[35]
Collagen covers sensitive nerve endings, thereby diminishing degree of pain in raw wounds. Initially collagen adheres due to fibrin collagen interaction and later by fibrovascular ingrowth into the collagen. All collagen membranes, with time, slowly undergo collagenolysis and get eventually sloughed off.[1]
The advantages of collagen sheet as a wound dressing material in surgery precancerous and cancerous lesions of oral soft tissues include the easy availability of collagen sheet, convenience of application, good tolerance of oral tissue, no adverse effects of the use of this membrane, obviation of second surgery to obtain graft or detachment of the pedicle, there is no morbidity associated with the use of grafts, and there are no problems associated with donor site healing.[34,36] No threat of human immunodeficiency virus or hepatitis infections is associated with collagen, as the bovine material is obtained from countries free of bovine spongiform encephalopathy and has a long shelf-life under normal storage conditions.[7]
OSMF is an insidious chronic disabling disease involving oral mucosa, oropharynx and rarely larynx characterized by juxtaepithelial inflammatory reaction followed by progressive fibrosis of the lamina propria and deeper connective tissues with concomitant muscle degeneration. Although vesicle formation is an early sign, patient’s usual complaint will barely be burning sensation and inability to have hot and spicy food. In the later stages, it shows a tendency for progressive fibrosis, leading to gradual reduction in mouth opening which hinders the function.[37,38] Management of trismus in OSMF is extremely challenging because of the nature of the disease, making the oral mucosa prone to contraction causing a significant reduction in the interincisal opening that was achieved with surgery. Nataraj et al.[1] performed a study in which collagen was used to cover surgical defects of OSMF in 15 cases and in other 15 cases, buccal pad of fat was used for the same. They found that the use of collagen membrane following excision of fibrotic bands in the management of oral submucous fibrosis, though statistically not significant gave better results with respect to post operative mouth opening as seen with a 6-month follow up. Pradhan et al.[33] also in a similar study found a significant difference in the postoperative mouth opening, an insignificant difference for post surgical morbidity and higher grades of surgical convenience in using collagen sheet as a wound dressing material as compared to buccal pad of fat. Reddy et al.[39] found good results in cases of OSMF when they impregnated dexamethasone in the collagen graft after excision of fibrous bands.
Manipulation of collagen
Though it has not been mentioned in literature, we have observed that most surgeons find it difficult to handle the wet collagen sheet in the oral cavity once it is taken out from its sterile packing. Even after washing away the preservative medium by immersing the material in sterile solution for 5-10 min, the tendency of the collagen to coil in itself does not go away. In our opinion, it can be attributed to its minimal thickness, elasticity and cohesiveness. So, the technique of using a “tie-over” bolster dressing (as used with skin grafts)[40] can be tried to secure collagen membrane to the recipient site. However, if the surgeon does not desire to keep the gauze or sponge dressing tied to the collagen graft, we suggest an easy technique that not only reduces the difficulty in manipulating collagen, but also provides perfect adaptation of the graft to the recipient site in oral cavity.
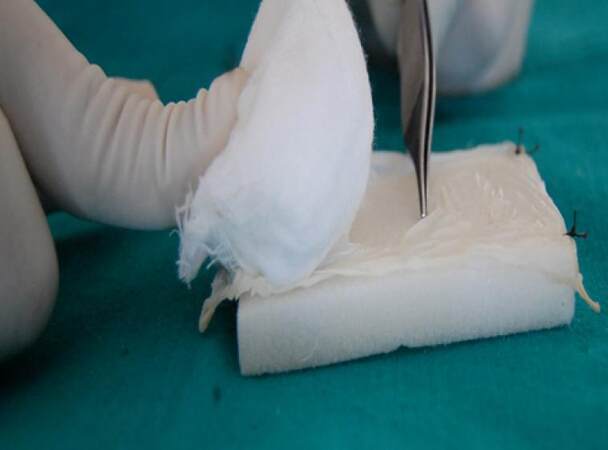
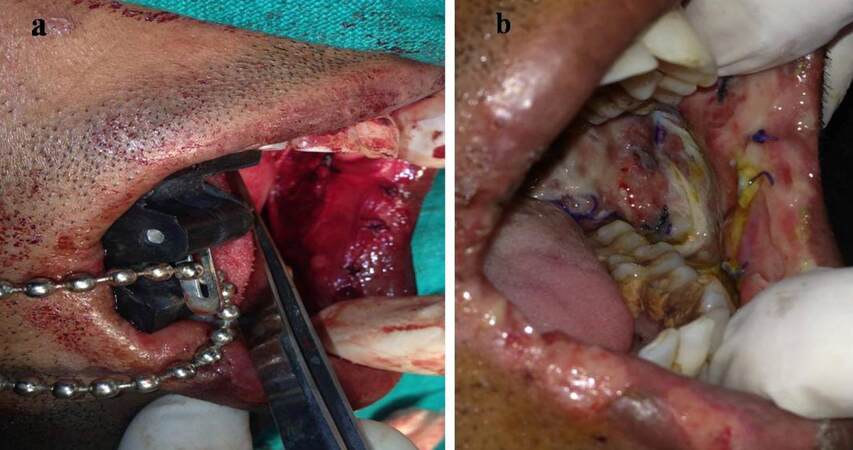
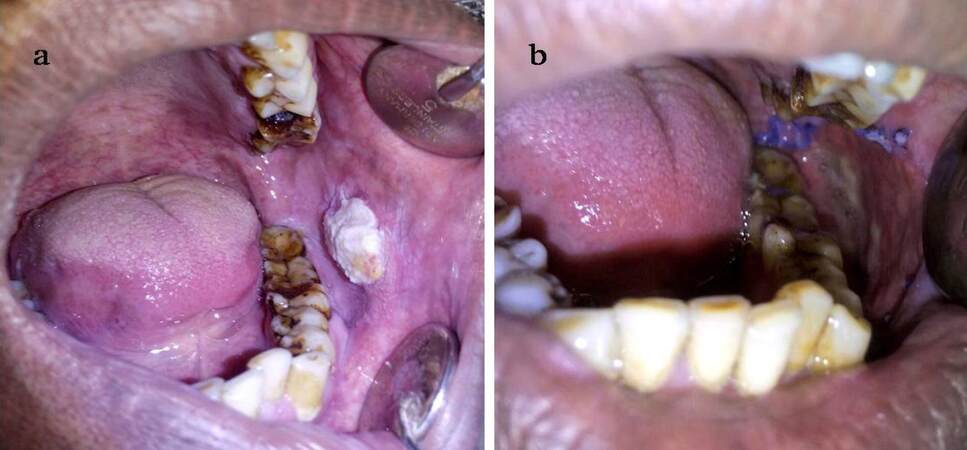
The method involves spreading the wet collagen sheet over a thick moistened gauze ball [Figure 1] after removing the preservative from collagen by immersing in in saline for 10 min. The size of graft and gauze depends on the size of the surgical defect. This gauze along with the graft is then taken to the surgical site and placed there with collagen facing the recipient site. With the gauze still in place, the accessible portion of collagen sheet underneath the gauze can be sutured to the wound margin [Figure 2]; choice of the suture depends on the surgeon. Next, the gauze can be slightly lifted over the portion of graft situated adjacent to the sutured collagen and another couple of stay sutures can be placed as required [Figure 3]. For example, if a buccal mucosa defect has to be grafted, the first suture can be placed anteriorly and lifting the gauze pad can proceed from anterior to posterior region. Thereafter, using this same technique, the whole circumference of the wound can be covered with the graft by sutures [Figure 4]. It is important to note that the gauze should be removed only before the last suture remains to be given to secure the graft in proper adaptation. After the gauze is removed, a well-adapted collagen can be seen which is not amenable to the problem of mobility and rolling of the material during suturing; making the placement of additional sutures (if required) very easy. A dressing may or may not be placed over the graft, depending on the choice of surgeon. We prefer to snugly fit a thick, removable, moistened gauze dressing over the graft at least for two days, to avoid graft contamination and to prevent the collection of fluid between graft and recipient site that could predispose the site to infection, thus jeopardizing the successful take-up of graft. If the surgical defects are multiple or bilateral, we advocate placement of a Ryle’s tube for 3-4 days so that immediate oral intake after the surgery can be avoided, yet nutrition is maintained. Clinical appearance of collagen grafted in OSMF and oral squamous cell carcinoma can be seen in Figures 5 and 6 respectively.
Figure 2. Placing the first suture through the accessible portion of graft to the surgical site, while the gauze is stabilized over the graft with a finger rest or an instrument
Figure 3. The gauze is slowly mobilized/ rolled, but not removed completely from the graft surface so that more area of the graft is accessible for suturing without much warping of the graft. Simultaneously, an instrument tip can be used to stabilize the graft
Figure 4. The collagen graft in place after suturing; the gauze is removed just before placing the last suture
Figure 5. (a) Collagen membrane secured on buccal mucosa in a case of oral submucous fibrosis following excision of buccal fibrous bands; (b) photograph of the 7th postoperative day showing partial healing of buccal mucosa and partial sloughing of collagen
Conclusion
Oral and maxillofacial surgeons treat various pathologies in and around the oral cavity. The commonest protocol of treatment for all pathologies is the surgical excision, rendering postoperative wounds prone to infection and scarring, unless they are covered using some kind of a graft. Various materials like skin, buccal mucosa, buccal pad of fat, tongue, palatal mucosa and xenografts have been used in the past for this purpose, each having its own advantages and shortcomings. Collagen is one such biomaterial that can be safely and effectively used for coverage of oral defects following surgery of oral cancer and precancer owing to a number of benefits such as its free availability, no additional donor site morbidity, economic, non-allergenic, biocompatibility, good epithelization with less contraction, etc. It is of particular value in cancer surgeries if one wants to safeguard autogenous grafts for future use, should a second resective surgery be required.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Nataraj S, Guruprasad Y, Shetty JN. A comparative clinical evaluation of buccal fat pad and collagen in surgical management of oral submucous fibrosis. Arch Dent Sci 2011;2:17-24.
2. Bagheri SC, Bell RB, Khan HA. Current therapy in oral and maxillofacial surgery. Philadelphia: Elsevier Saunders; 2011.
3. Shevale VV, Kalra RD, Shevale VV, Shringarpure MD. Management of oral submucous fibrosis: a review. Indian J Dent Sci 2012;4:107-14.
4. Mokal NJ, Raje RS, Ranade SV, Prasad JS, Thatte RL. Release of oral submucous fibrosis and reconstruction using superficial temporal fascia flap and split skin graft- a new technique. Br J Plast Surg 2005;58:1055-60.
5. Lai DR, Chen HR, Lin LM, Huang YL, Tsai CC. Clinical evaluation of different treatment methods for oral submucous fibrosis. A 10-year experience with 150 cases. J Oral Pathol Med 1995;24:402-6.
6. Mariappan N. Collagen dressing for thermal burns. Sch J App Med Sci 2015;3:58-61.
7. Lazovic G, Colic M, Grubor M, Jovanovic M. The application of collagen sheet in open wound healing. Ann Burns Fire Disasters 2005;18:151-6.
8. Holmes C, Wrobel JS, MacEachern MP, Boles BR. Collagen-based wound dressings for the treatment of diabetes-related foot ulcers: a systematic review. Diabetes Metab Syndr Obes 2013;6:17-29.
9. Bhattacharya S, Tripathi HN, Gupta V, Nigam B, Khanna A. Collagen sheet dressings for cutaneous lesions of toxic epidermal necrolysis. Indian J Plast Surg 2011;44:474-7.
10. Singh O, Gupta SS, Soni M, Moses S, Shukla S, Mathur RK. Collagen dressing versus conventional dressings in burn and chronic wounds: a retrospective study. J Cutan Aesthet Surg 2011;4:12-6.
11. Shankar N, Eligar R, Ramesh H, Hebsur NI. A comparative study of collagen granule dressing versus conventional dressing in deep wounds. IOSR J Dent Med Sci 2015;14:23-8.
12. Rai R, Sudarshan SH, Dsouza R, Saldhana E, Aithala PS. Collagen dressing versus heparin dressing in burn wound management. J Evol Med Dent Sci 2013;2:9124-30.
13. Hudson JW, Pickett DO. A 5-year retrospective review of primary palatoplasty cases utilizing an acellular collagen interpositional graft. J Oral Maxillofac Surg 2015;73:1393.e1-3.
14. Li X, Chen SL, Zhu SX, Zha GQ. Guided bone regeneration using collagen membranes for sinus augmentation. Br J Oral Maxillofac Surg 2012;50:69-73.
15. Wong RW, Rabie AB. Effect of gusuibu graft on bone formation. J Oral Maxillofac Surg 2006;64:770-7.
16. Ferreira AM, Gentile P, Chiono V, Ciardelli G. Collagen for bone tissue regeneration. Acta Biomater 2012;8:3191-200.
17. Block MS, Kelley B. Horizontal posterior ridge augmentation: the use of a collagen membrane over a bovine particulate graft: technique note. J Oral Maxillofac Surg 2013;71:1513-9.
18. Birkenfeld F, Flörke C, Behrens E, Rohnen M, Kern M, Gassling V, Wiltfang J. Mechanical properties of collagen membranes modified with pores - are they still sufficient for orbital floor reconstruction? Br J Oral Maxillofac Surg 2015;53:957-62.
19. Babu HM, Gujjari SK, Prasad D, Sehgal PK, Srinivasan A. Comparative evaluation of a bioabsorbable collagen membrane and connective tissue graft in the treatment of localized gingival recession: a clinical study. J Indian Soc Periodontol 2011;15:353-8.
20. Prescott RS, Alsanea R, Fayad MI, Johnson BR, Wenckus CS, Hao J, John AS, George A. In vivo generation of dental pulp-like tissue using human pulpal stem cells, a collagen scaffold and dentin matrix protein 1 following subcutaneous transplantation in mice. J Endod 2008;34:421-6.
21. Sowjanya NP, Rao NM, Satya Bhushan NVVS, Krishnan G. Versitality of the use of collagen membrane in oral cavity. J Clin Diagn Res 2016;10:ZC30-3.
22. Wiesmann HP, Nazer N, Klatt C, Szuwart T, Meyer U. Bone tissue engineering by primary osteoblast-like cells in a monolayer system and 3-dimensional collagen gel. J Oral Maxillofac Surg 2003;61:1455-62.
23. Chvapil M, Kronenthal L, Van Vinkle W Jr. Medical and surgical applications of collagen. Int Rev Connect Tissue Res 1973;6:1-61.
25. Veves A, Sheehan P, Pham HT. A randomized, controlled trial of promogran (a collagen/oxidized regenerated cellulose dressing) vs. standard treatment in the management of diabetic foot ulcers. Arch Surg 2002;137:822-7.
26. Nataraj C, Ritter G, Dumas S, Helfer FD, Brunelle J, Sander TW. Extra cellular wound matrices: Novel stabilization and sterilization method for collagen-based biologic wound dressings. Wounds 2007;19:148-56.
27. Park SN, Lee HJ, Lee KH, Suh H. Biological characterization of EDC-crosslinked collagen-hyaluronic acid matrix in dermal tissue restoration. Biomaterials 2003;24:1631-41.
28. Nagata H, Ueki H, Moriguchi T. Fibronectin: localization in normal human skin, granulation tissue, hypertrophic scar, mature scar, progressive systemic sclerotic skin, and other fibrosing dermatoses. Arch Dermatol 1985;121:995-9.
29. Motta G, Ratto GB, De Barbieri A, Corte G, Zardi L, Sacco A, Castagnola M. Can heterologous collagen enhance the granulation tissue growth? An experimental study. Ital J Surg Sci 1983;13:101-8.
30. Shah JP, Johnson NW, Batsakis JG. Oral cancer. London: Taylor and Francis Group; 2003.
31. Kramer IR, Lucas RB, Pindborg JJ, Sobin LH. World Health Organization Collaborating Center for oral precancerous lesions. Definition of leukoplakia and related lesions: an aid to studies on oral precancer. Oral Surg Oral Med Oral Pathol 1978;46:518-39.
32. Rastogi S, Modi M, Sathian B. The efficacy of collagen membrane as a biodegradable wound dressing material for surgical defects of oral mucosa: a prospective study. J Oral Maxillofac Surg 2009;67:1600-6.
33. Pradhan H, Gupta H, Sinha V, Gupta S, Shashikanth M. Two wound-covering materials in the surgical treatment of oral submucous fibrosis: a clinical comparison. J Oral Biol Craniofac Res 2012;2:10-4.
34. Borle RM, Nimonkar PV, Rajan R. Extended nasolabial flaps in the management of oral submucous fibrosis. Br J Oral Maxillofac Surg 2009;47:382-5.
35. Rahman A, Packiaraj I, Genmorgan, Alaguvelrajan, Kandasamy. Role of collagen membrane: a comprehensive review. J Adv Med Dent Sci Res 2015;3:95-7.
36. Paramhans D, Mathur RK, Newaskar V, Shukla S, Sudrania MK. Role of collagen membrane for reconstruction of buccal defects following fibrotic band excision and coronoidectomy in oral submucous fibrosis. MP Soc Otolaryngol Waent 2010;3.
37. Borle RM, Borle SR. Management of oral submucous fibrosis: a conservative approach. J Oral Maxillofac Surg 1991;49:788-91.
38. Pillai R, Balaram P, Reddiar KS. Pathogenesis of oral submucous fibrosis: Relationship to risk factors associated with oral cancer. Cancer 1992;69:2011-20.
39. Reddy YR, Srinath N, Nandakumar H, RajiniKanth M. Role of collagen impregnated with dexamethasone and placentrix in patients with oral submucous fibrosis. J Maxillofac Oral Surg 2012;11:166-70.
40. Reus WF, Mathes SJ. Plastic Surgery: Principles and Practice. In: Jurkeiwicz MJ, Krizek TJ, Mathes SJ, Ariyan S, editors. Wound closure. St. Louis: Mosby; 1990. pp. 20-2.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Vastani A, Chourasia N, Pahlajani V, Choubey S. Collagen membrane for reconstruction of soft tissue defects after surgery of oral cancer and precancer: a brief review. Plast Aesthet Res 2016;3:100-5. http://dx.doi.org/10.20517/2347-9264.2015.119
AMA Style
Vastani A, Chourasia N, Pahlajani V, Choubey S. Collagen membrane for reconstruction of soft tissue defects after surgery of oral cancer and precancer: a brief review. Plastic and Aesthetic Research. 2016; 3: 100-5. http://dx.doi.org/10.20517/2347-9264.2015.119
Chicago/Turabian Style
Vastani, Ankita, Nishant Chourasia, Vedant Pahlajani, Suryakant Choubey. 2016. "Collagen membrane for reconstruction of soft tissue defects after surgery of oral cancer and precancer: a brief review" Plastic and Aesthetic Research. 3: 100-5. http://dx.doi.org/10.20517/2347-9264.2015.119
ACS Style
Vastani, A.; Chourasia N.; Pahlajani V.; Choubey S. Collagen membrane for reconstruction of soft tissue defects after surgery of oral cancer and precancer: a brief review. Plast. Aesthet. Res. 2016, 3, 100-5. http://dx.doi.org/10.20517/2347-9264.2015.119
About This Article
Special Issue
Copyright
Author Biographies




Data & Comments
Data
 Cite This Article 10 clicks
Cite This Article 10 clicks














Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.