Modulation of Wnt/β-catenin signaling affects the directional differentiation of hair follicle stem cells
Abstract
Aim: The differentiation of hair follicle stem cells (HFSCs) into hair follicle cells has potential clinical applications for cutaneous burns. However, the mechanisms regulating the differentiation of HFSCs into hair follicular papilla or epidermal cells are currently not clear. This study investigated the role of the Wnt/β-catenin pathway and its crosstalk with other signaling components during this differentiation process.
Methods: Lithium chloride (LiCl, 10 mmol/L) and keratinocyte growth factor (KGF, 10 µg/L) were used to induce HFSC differentiation, validated by immunofluorescence analysis. The mRNA expression of β-catenin, adenomatous polyposis coli, glycogen synthase kinase-3β (GSK-3β), axin, and lymphoid enhancer factor-1 after 3, 5, 7, and 9 days were measured to evaluate the role of the Wnt/β-catenin pathway.
Results: During LiCl-induced HFSC differentiation into hair follicle cells, the Wnt/β-catenin signaling pathway was activated and the expression of GSK-3β, a vital component of the degradation compound, was inhibited. This led to increased cytoplasmic β-catenin expression, nuclear translocation, and subsequent target gene transcription. By contrast, KGF induced the differentiation of HFSCs into epidermal cells and did not affect the expression of β-catenin. This data indicates that LiCl and KGF distinctly regulate the differentiation of HFSCs into hair follicle and epidermal cells, respectively. Furthermore, the Wnt/β-catenin signaling pathway is predominantly involved in hair follicle differentiation.
Conclusion: these results demonstrate that LiCl can be used to differentiate HFSCs into hair follicle cells in vitro, which has important therapeutic applications for treating patients with cutaneous damage.
Keywords
Introduction
The discovery of hair follicle stem cells (HFSCs) and the development of methods to culture these cells in vitro have led to the possibility of developing new tissue engineering treatments for cutaneous damage caused by burns.[1] HFSCs are highly proliferative multipotent stem cells with multi-lineage potential. They can differentiate into a range of distinct cell types including epidermal cells, hair follicle cells, and sebaceous cells.[2] Both the proliferation rate and the differentiation capacity of HFSCs are influenced by autologous genes and external signals. The Wnt/β-catenin signaling pathway plays a crucial role in the development of follicles and hair.[3,4] During both embryonic development and tumorigenesis Wnt proteins bind to their receptors and activate Wnt/β-catenin signaling, leading to β-catenin accumulation in the cytoplasm and subsequent nuclear translocation. β-catenin binding to lymphoid enhancer factor-1 (LEF1) activates target genes and accelerates the directional differentiation of HFSCs.[3,4] Other factors, including chemicals (e.g. LiCl) and growth factors, influence cytoplasmic β-catenin levels in HFSCs, leading to HFSC differentiation.[1] However, details of the mechanism require further investigation. The purpose of this study was to investigate how keratinocyte growth factor (KGF) and LiCl influence the function of the Wnt/β-catenin signaling pathway and its interaction with other signaling components during human HFSC differentiation into dermal papilla and epidermal cells.
Methods
Tissue harvest and cell isolation
Fresh scalp specimens were obtained under aseptic condition and HFSCs were isolated using previously established methods.[1] Briefly, scalp skin from patients undergoing surgery was washed repeatedly with phosphate buffered solution (PBS) containing 5% penicillin and streptomycin (North China Pharmaceutical Corporation, China). After removing subcutaneous adipose tissue and impurities, the skin was cut into 4 mm × 2 mm pieces, placed in a centrifuge tube containing 0.25% Dispase (Shanghai Biological Technical Company, China), and digested overnight at 4 °C. The hair follicles were then gently pulled out in the direction of hair growth. The dissociated adipose tissue was then washed with PBS and hair follicles were added to a 60-mm petri dish containing 10% fetal bovine serum (FBS) in Dulbecco’s Modified Eagle Media (DMEM; Hyclone, USA). Bulge areas were isolated from the hair follicles by incubation with 2.5 g/L trypsin and 0.04% EDTA for 10 min at 37 °C, followed by centrifugation and resuspension in K-SFM medium (Shanghai Biological Technical Company, Shanghai, China) at a density of 5 × 107 cells/L. Cells were incubated at 37 °C under 5% CO2 in a humidified incubator. The culture medium was replaced every 3-4 days, and HFSCs were cultured and passaged three times prior to being used for all experiments.
Measuring the proliferation rate of HFSCs
To measure their rates of proliferation, HFSCs were seeded at the following four different densities: 1 × 103, 1 × 104, 1 × 105, and 1 × 106 cells/mL of 24-well plate. Growth curves of the HFSCs were obtained by seeding 1 × 105 logarithmically growing cells in 1mL medium into each well of a 24-well culture plate. A total of 36 wells were used. Hoechst 33258 DNA quantitation was performed every day after 7 days in culture.
Immunofluorescence assay
Immunofluorescence analysis was performed as previously described.[5] Briefly, sterile glass coverslips were placed into 60 mm dishes, and approximately 2 × 104 cells/mL of 24-well plate were seeded into each dish. After 7 days in culture, adherent cells were stained for pan-cytokeratin, K19-Cy3, and β1-integrin and analyzed by immunofluorescence microscopy.
Treatment with LiCl and KGF
To investigate the effects of specific compounds on HFSC differentiation, HFSCs were treated with LiCl (0.5, 1.5, 10, or 25 mmol/L) or KGF (10, 25, 50, or 100 µg/L). After 7 days, Hoechst 33258 DNA quantitation was performed to compare the effects of different incubation times on cell proliferation.
Reverse transcriptase-polymerase chain reaction analysis of mRNA expression
The reverse transcriptase-polymerase chain reaction (RT-PCR)[6,7] was used to measure the expression of β-catenin, adenomatous polyposis coli (APC), axin, glycogen synthase kinase-3β (GSK-3β), and LEF1 mRNA at various time points following treatment with 10 mmol/L LiCl. HFSCs were resuspended in K-SFM medium (without fetal calf serum) and seeded into a 100-mm culture dish at a concentration of 1 × 105 cells/mL. LiCl was then added to the medium at a concentration of 10 mmol/L. RT-PCR was used to measure the expression of β-catenin, APC, axin, GSK-3β, and LEF1 mRNA after 3, 5, 7, and 9 days in culture. An untreated group was included as a control. Similarly, following treatment with KGF, β-catenin, APC, axin, GSK-3β, and LEF1 mRNA expression was measured on days 3, 5, 7, and 9. An untreated control group was included.
Isolation of total RNA: total RNA was isolated with TRIZOL using Superscript III (Invitrogen, CA, USA), according to the manufacturer’s protocol.
Reverse transcription: template RNA (1 µg) and 1 µg Oligo (dT) 15 were added to RNase-free water (to a final volume of 5 µL). After mixing well, the reaction was incubated at 70 °C for 5 min, chilled on ice for 5 min, and then 13.5 µL RNase-free water, 4 µL 5 × RT Buffer, 1 µL dNTPs, 1 µL RNase inhibitor and 1 µL MMLV RNase were added. The reaction was incubated at 36 °C for 10 min, 42 °C for 60 min, and then inactivated at 70 °C for 10 min, cooled on ice, and stored.
Primer design and synthesis: primer design was based on the target DNA sequences. To avoid contamination of genomic DNA, primer pairs were designed to amplify across an intron [Table 1]. Primers were synthesized by Shanghai Sango Biotech Co. Ltd. (Shanghai, China).
Primer sequences and amplified fragments
| Primer pairs | Sequences (5ʹ to 3ʹ) | Denaturation temperature (°C) | Fragment size (bp) |
|---|---|---|---|
| β-catenin forward | TACCTCCCAAGTCCTGTATGAG | 56.0 | 180 |
| β-catenin reverse | TGAGCAGCATCAAACTGTGTAG | ||
| APC forward | TTAAAGCAAGTTGAGGCACTG | 55.5 | 270 |
| APC reverse | ACCGGCTTCCATAAGAACGGA | ||
| Axin forward | GCGGGACAGATTGATTCACTT | 57.0 | 190 |
| Axin reverse | TGTGGACACCAGTTCTCCCT | ||
| GSK-3β forward | ATTTCCAGGGGATAGTGGTGT | 56.0 | 154 |
| GSK-3β reverse | TCCTGACGAATCCTTAGTCCAAG | ||
| LEF1 forward | AATGAGAGCGAATGTCGTTGC | 56.5 | 137 |
| LEF1 reverse | GCTGTCTTTCTTTCCGTGCTA | ||
| GAPDH forward | TGTTGCCATCAATGACCCCTT | 57.0 | 202 |
| GAPDH reverse | CTCCACGACGTACTCAGCG |
RT-PCR reagents: RT-PCR reagents contained 1 µL template RNA, 1.5 µL of 25 mmol/L MgC12, 4 µL 5 × PCR buffer, 1 µL dNTPs, l µL each primer, 0.5 µL Taq DNA polymerase, and 9.5 µL RNase-free water. RT-PCR were subjected to an initial denaturation for 3 min at 94 °C, and then 35 cycles of 94 °C for 45 s, 56 °C for 30 s, and 72 °C for 1 min. A final extension for 5 min at 72 °C was performed before the reaction was stored at 4 °C. Reaction products were analyzed by electrophoresis. Primers and annealing temperatures are shown in Table 1.
Statistical analysis
All data were analyzed using SPSS10.0. Statistical significance was set at P < 0.05; P < 0.01 indicates that the difference between experimental groups was significant; P > 0.05 indicates no difference between experimental groups.
Results
HFSC growth characteristics
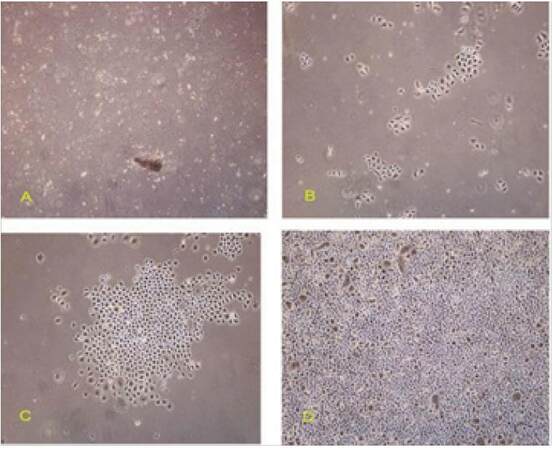
Primary cells derived from the hair follicle bulge were spheroid, small, and evenly sized. Live cells were optically transparent with a clear boundary and clusters of undissociated cells could be seen [Figure 1]. Trypan blue staining showed that 95% of cells were alive. At the initial stage of culture, cell adhesion and activity were weak. After culture in serum-free K-SFM containing epidermal growth factor (EGF) for 1-3 days, a few human HFSCs were observed to adhere to culture vessel, although most remained in suspension [Figure 1A and B]. After 4-6 days in culture, a few single cells or cell clusters had attached to the culture dish; these had a cobblestone-like appearance, an orderly arrangement, and were highly refractive. Cell numbers increased with increasing culture time [Figure 1C and D]. After 2 weeks in culture, a small number of aging cells were observed.
Dynamics of cell growth
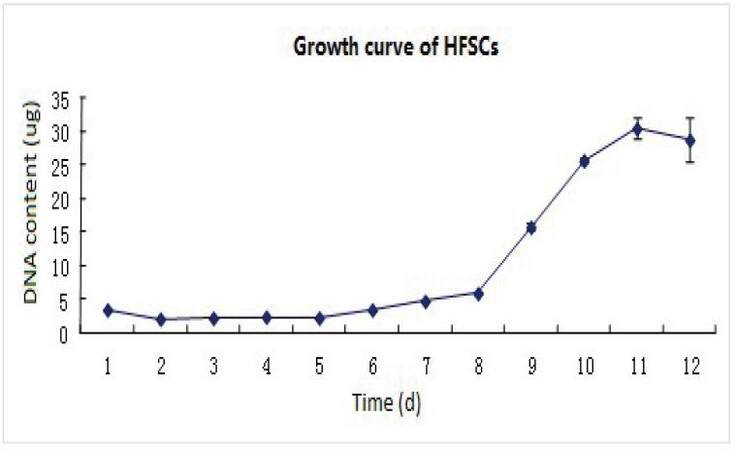
Growth curves [Figures 1 and 2] show that the cell growth rate was slow on days 1-3. On days 4-6, cells grew faster and were in the logarithmic phase. After this point, cell growth became slower and aging cells appeared.
Figure 2. Growth curves of primary culture cells from scalp follicular bulge. HFSCs: hair follicle stem cells
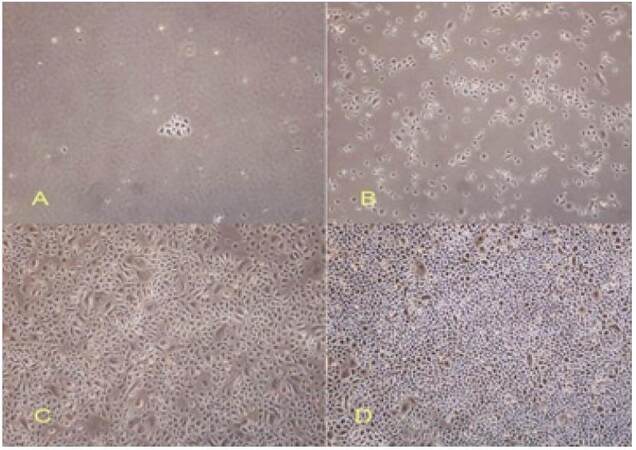
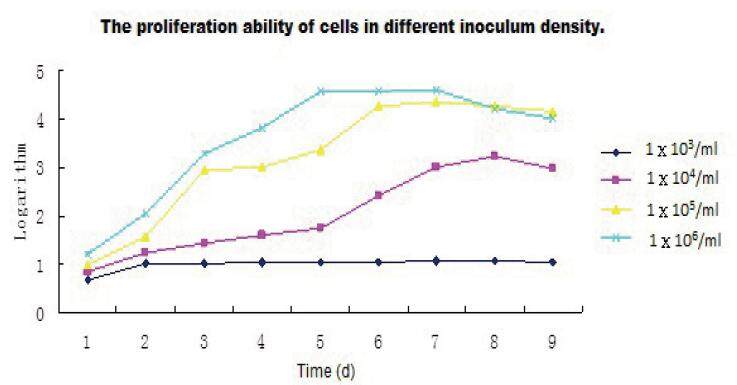
Cell proliferation varied according to seeding density [Figures 3 and 4]. At seeding densities of 1 × 103 and 1 × 104, a few cells showed clonal growth but cells proliferated slowly and most died after a short time in culture. When the seeding density was increased to 1 × 105 cells/mL, cells proliferated well and showed adherent growth at early time points. Cells seeded at a density of 1 × 106 cells/mL rapidly formed clusters and underwent contact inhibition following rapid nutrient depletion. These cells showed signs of aging.
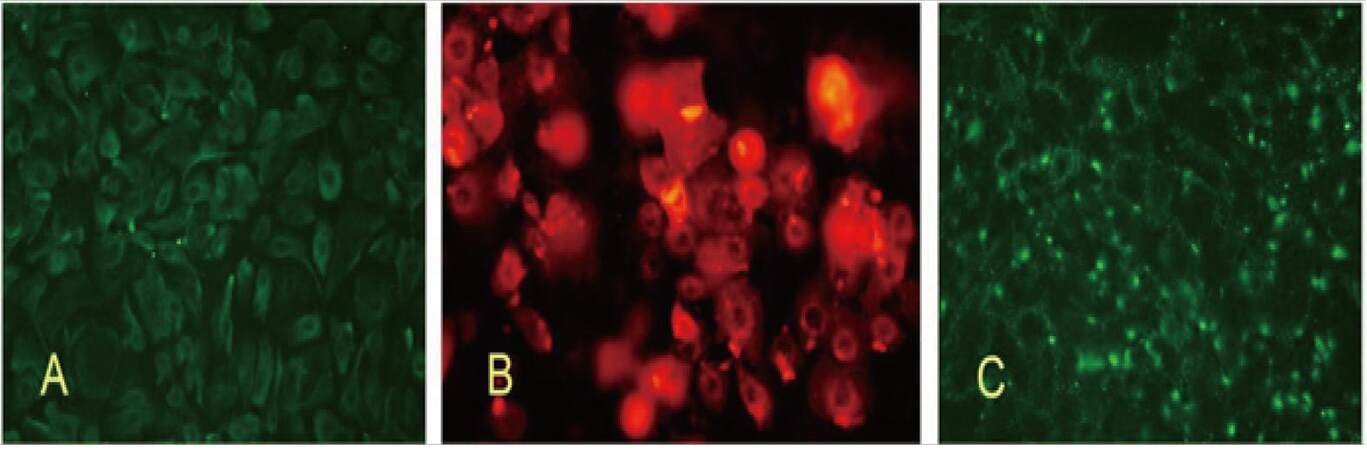
Identification of HFSCs and characteristics of HFSC protein expression
Pan-cytokeratin expression: adherent pan-cytokeratin-labeled HFSCs showed that the cytoplasm of these cells were positive for pan-cytokeratin [Figure 5A]. Cytokeratin is a defining feature of HFSCs.[5,8]
K19-Cy3 expression: isolated keratin 19 (K19)-labeled HFSC clusters were positive for cytoplasmic K19 [Figure 5B]. K19 is a specific cytoplasmic marker for HFSCs.[5,9]
β1-integrin expression: HFSCs showed a high rate of β1-integrin expression, as shown in Figure 5C. Positively stained cells grew in clusters. Previous studies have confirmed that β1-integrin is a membrane marker for HFSCs.[5,8]
Figure 3. The effect of seeding density on cell growth. Cells were seeded at densities of 1 × 103 cells/mL (A), 1 × 104 cells/mL (B), 1 × 105 cells/mL (C), or 1 × 106 cells/mL (D). Cell proliferation and cells morphology were assessed after 3 days in subculture. Magnification, X40
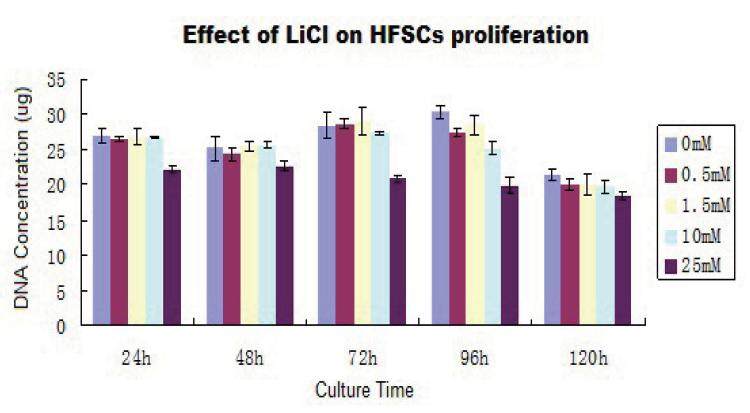
The influence of LiCl on HFSC proliferation
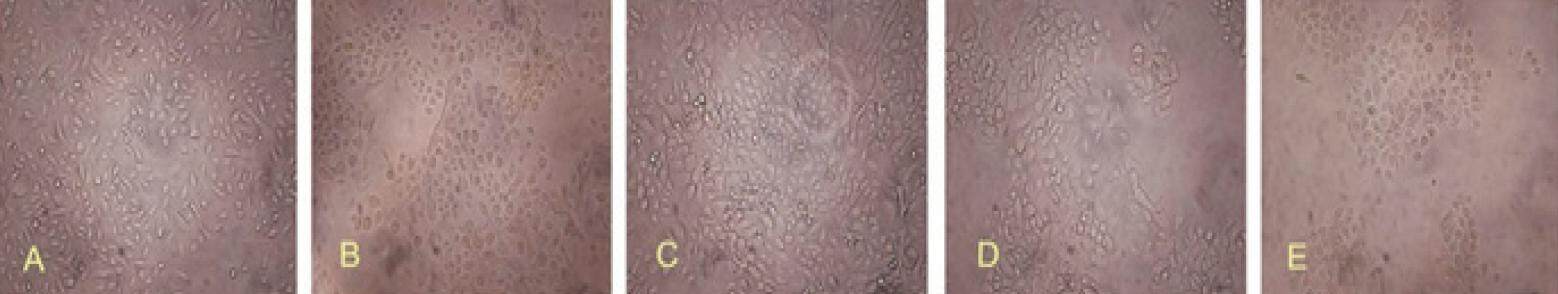
There were no significant differences between the proliferation rates of HFSCs treated with lower doses of LiCl and that of the control group; there was an inverse correlation between dose and proliferation rate at higher concentrations of LiCl than 10 mmol/L [Figures 6 and 7]. LiCl concentrations less than 10 mmol/L had no effect on the proliferation rate; moreover, treatment with 0-10 mmol/L LiCl did not markedly affect cell proliferation, especially for culture times greater than 72 h (P > 0.05). However, the proliferation rate decreased at a LiCl concentration of 25 mmol/L; there was a clear difference between treated and control cells at each time point (P < 0.05).
Figure 6. Effect of different LiCl concentrations on hair follicle stem cells proliferation. Cells were assessed 3 days after treatment of LiCl at different concentrations. (A) after treatment with 0 mmol/L LiCl, cells have grown well and display a cobblestone-like appearance; (B) after treatment with 0.5 mmol/L LiCl, cells have become rounded, or occasionally an elongated spindle shape, and show a tendency to aggregate; (C) after treatment with 1.5 mmol/L LiCl, in general, cells have become larger. Occasionally, cells have developed protuberances, which may be related to cell migration; (D) after treatment with 10 mmol/L LiCl, cell morphology has changed. Cells have become highly aggregated and cell density is reduced; (E) after treatment with 25 mmol/L LiCl, cell density has significantly decreased. Both the cytoplasm and nuclei show various morphological changes. The proliferation rate has also decreased. Magnification, X100
After 24 h of culture, DNA quantitation in the 25 mmol/L group compared with 0 mmol/L group, P < 0.05, as compared with other groups, P > 0.05; after 48 h of culture, DNA quantitation in 25 mmol/L group compared with 0 mmol/L group, P < 0.05, as compared with other groups, P > 0.05; after 72 h of culture, DNA quantitation in 25 mmol/L group compared with 0 mmol/L group, P < 0.01, as compared with other groups P > 0.05; after 96 h of culture, DNA quantitation in 25 mmol/L group compared with 0 mmol/L group, P < 0.01, as compared with other groups, P < 0.05, also representing a significant difference; after 120 h of culture, DNA quantitation in 25 mmol/L group compared with 0 mmol/L group P < 0.05, as compared with other groups P > 0.05.
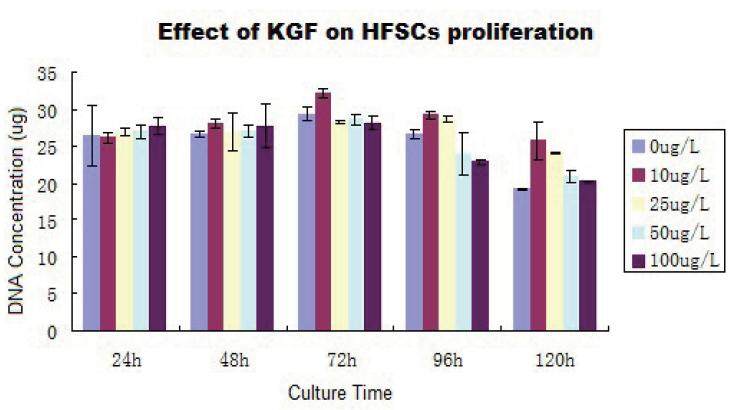
Effect of KGF on HFSC proliferation
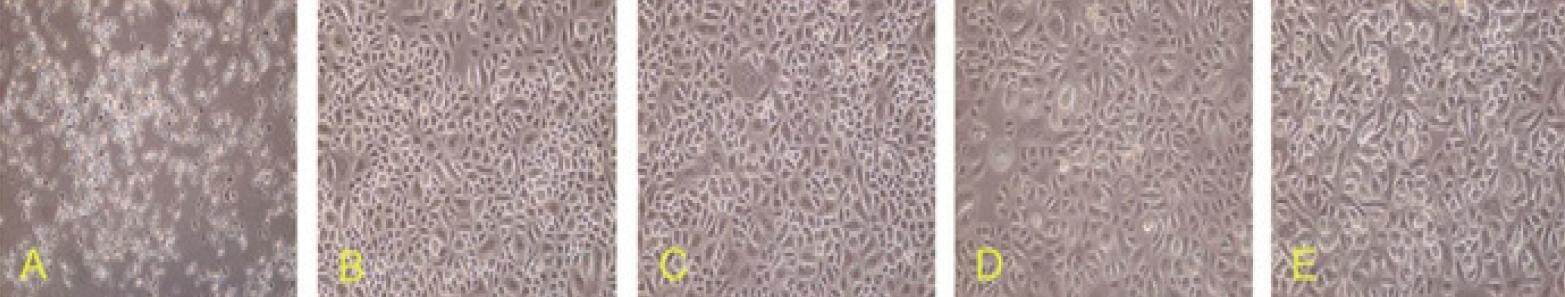
The proliferation rate of HFSCs increased following KGF treatment, with a direct correlation between KGF dose and HFSC proliferation rate being observed [Figures 8 and 9]. HFSCs proliferated well at KGF concentrations less than 50 µg/L. By contrast, at very high KGF concentrations HFSCs proliferated well at early time points, but died prematurely at later time points. At KGF concentrations of 10-25 µg/L, cells proliferated well and showed no signs of premature aging at later time points.
Figure 8. Effect of different keratinocyte growth factor (KGF) concentrations on hair follicle stem cells proliferation. Cells were assessed 3 days after treatment. (A) the KGF 0 µg/L group shows cells have grown well and display a cobblestone-like appearance; (B) the KGF 10 µg/L group shows cell density has increased. A small number of enlarged cells have appeared; (C) the KGF 25 µg/L group shows differentiated cells with enlarged nuclei and enriched cytoplasm can be seen; (D) the KGF 50 µg/L group shows the proportion of differentiated cells increased, and the cell density decreased; (E) the KGF 100 µg/L group shows the proportion of differentiated cells increased, cell aging was observed. Magnification, X100
Figure 9. Effect of keratinocyte growth factor (KGF) on hair follicle stem cells (HFSCs) proliferation
Statistical analyses showed that after 24 h of culture, there were no significant differences between the KGF-dose treatment groups and the control group.
After 48 h of culture, the KGF 10 µg/L treatment group was significantly different as compared with the control group (P < 0.05), but there was no difference between the control group and the other KGF-dose treatment groups. After 72 h of culture, the KGF 10 µg/L treatment group showed a significant increase in proliferation when compared with the control group (P < 0.01). However, there was no observed difference between the control group and the other KGF-dose treatment groups. After 96 h of culture, the 10, 25, and 100 µg/L KGF treatment groups showed a significant increase in proliferation when compared with the control group (P < 0.01). However, there was no significant difference between the control group and the 50 µg/L KGF treatment group. After 120 h of culture, all doses of KGF had significantly increased the proliferation of HFSCs compared with the control group (P < 0.05).
Effects of LiCl and KGF on HFSC differentiation and morphology
There were obvious differences in cell morpho-differentiation, adhesion, and proliferation between the 10 mmol/L LiCl and 10 µg/L KGF treatment groups and the control group [Figure 10A-D]. The LiCl 10 µg/L treatment group showed a significant increase in proliferation [Figure 10E-H]. By contrast, the proliferation rate of KGF-treated cells initially increased in a dose-dependent manner. However, cell proliferation decreased with aging and vacuoles formed within the cells at later time points [Figure 10I-L].
Figure 10. Untreated hair follicle stem cells (HFSCs). Cells morphological features of negative controls (HFSCs subculturing without intervention) after 3 (A), 5 (B), 7 (C), and 9 (D) days in culture. HFSC treated with 10 mmol/L LiCl. Cells morphological features shown after 3 (E), 5 (F), 7 (G), and 9 (H) days in culture. HFSCs treated with 10 μg/L KGF. Cells morphological features shown after 3 (I), 5 (J), 7 (K), and 9 (L) days in culture. Magnification, X40
Effects of LiCl and KGF on the mRNA expression of components of the Wnt/β-catenin signaling pathway
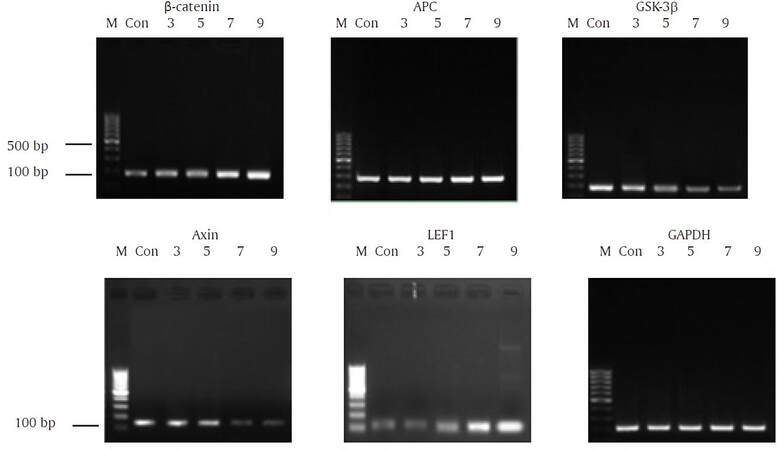
Following treatment with 10 mmol/L LiCl, the mRNA expression of β-catenin was increased when compared with control cells. There was also an increased expression of APC and LEF1 mRNA, but a gradual reduction in axin and GSK-3β mRNA expression [Figure 11].
Figure 11. Effect of 10 mmol/L LiCl on β-catenin, APC, GSK-3β, axin, and LEF1 mRNA expression. APC: adenomatous polyposis coli; GSK-3: glycogen synthase kinase-3β; LEF1: lymphoid enhancer factor-1
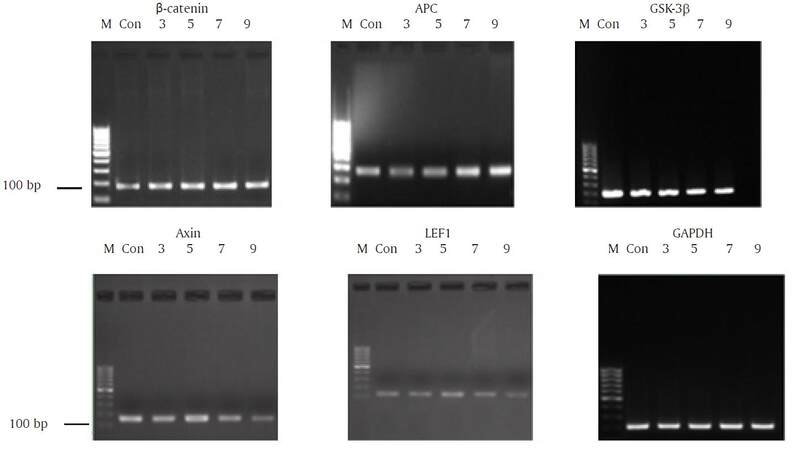
Following treatment with 10 µg/L KGF, the expression of β-catenin mRNA did not change markedly compared with control cells; however, GSK-3β and LEF1 mRNA expression gradually declined [Figure 12].
Discussion
HFSC differentiation is regulated by multiple factors,[1] which interact to form an intricate regulatory system.[1,2] The main intrinsic regulatory factors affecting HFSC proliferation and differentiation are Wnt, β-catenin and the Notch and exit domain A (EDA) signaling pathways.[1,10-12] Extrinsic signals primarily include mitogens, intercellular interactions, cell growth factors [e.g. basic fibroblast growth factor (bFGF) and EGF], and some chemical substances (e.g. LiCl).[13-15] These signals constitute the external microenvironment of proliferation and differentiation of HFSCs. KGF is a member of the FGF superfamily, which regulates the proliferation and differentiation of various cell types. KGF expression in matrix cells is sharply upregulated when skin is injured.[13] KGF also facilitates HFSC proliferation. In this study, HFSCs were isolated from the hair follicle bulge and the effects of different concentrations of LiCl and KGF on HFSC proliferation and differentiation were explored.
LiCl, a crucial Wnt signaling agonist, inhibits the activity of GSK-3β.[11] GSK-3 phosphorylation at serine-9 prevents GSK-3β from degrading β-catenin, thus elevating β-catenin expression.[14] This study found that low LiCl concentrations (0-10 mmol/L) not only induce β-catenin expression but also slightly increase HFSCs proliferation. However, proliferation decreased as the LiCl concentration was increased. LiCl treatment also led to a reduction in cell adhesion and caused HFSCs to grow in dispersed clusters. This is a result of LiCl inhibiting intracellular E-cadherin expression, which weakens cell adhesion and facilitates cell migration.[15] This phenomenon may result from E-cadherin transposition caused by β-catenin up-regulation. These data suggest that changes in β-catenin expression can change cell adhesion, which has an important role in promoting cell proliferation.
In the current study, β-catenin expression directly correlated with the β-catenin-axin-APC-GSK-3β complex, but inversely correlated with GSK-3β and axin levels. Axin, a critical negative regulator of the Wnt signaling pathway, is a key component of the β-catenin degradation complex. It has several protein-protein interaction domains and functions as scaffolding protein in various protein complexes. Axin brings GSK-3β in close proximity to β-catenin and regulates the degradation of phosphorylated β-catenin. This study demonstrated that LiCl suppresses the expression of GSK-3β mRNA and thus inhibits the formation of the axin complex, which indirectly elevates the level of β-catenin.
As a downstream transcription factor in the Wnt signaling pathway, LEF1activates the transcription of specific genes. According to recent reports,[7,8,16] endogenous LEF1 synergizes with Wnt signaling. It is widely accepted that β-catenin activates downstream target genes through a complex formation with LEF1. Cofactor ALY, enhancer TCRα, and LEF1 cooperatively form a higher-order nucleoprotein complex that enhances the synergistic activation of β-catenin/LEF1 and thus transcriptional activity.[16] This study found that LiCl treatment increased the levels of β-catenin and LEF1 mRNA along with the course induce. It is likely that increased protein levels of β-catenin complex with LEF1. These results are identical with those of the above-mentioned mechanisms of the Wnt signaling pathway, and indicate that activations of the β-catenin and LEF1 signaling pathways are involved in HFSC’s differentiation.
Numerous studies have shown that β-catenin functions by forming a complex with LEF1 in the Wnt signaling pathway.[17-20] Zhu et al.[17] transfected keratinocytes with retrovirus and found that mutant LEF1 expressed in the basal lamina of transgenic mice cannot interact with β-catenin. Thus, hair formation is prevented and adult basal epidermal cells are transformed into pluripotent embryonic-like ectoderm. LEF1 is an essential transcriptional factor for the normal development of hair follicles. LEF1 expression stimulates the stem cell to develop into hair, facilitates differentiation of both hair matrix cells and hair follicles, and participates in the formation of the root sheath.
In an attempt to improve our understanding of the role of β-catenin in HFSC proliferation and differentiation, this study investigated whether KGF treatment stimulates these processes via the Wnt signaling pathway. The results indicate that adding KGF to serum-free culture medium containing EGF stimulated HFSC proliferation. Although this effect was not apparent in the first 48 h of culture, for incubation times more than 48 h, there was a marked difference in DNA content between the 10 µg/L KGF treatment group and control cells. The reduction in DNA content indicates that KGF maintains cell proliferation and postpones keratinocyte differentiation.
Richardson et al.[6] observed repression of hair follicle induction by KGF and EGF in cultured mice embryo skin. They found that Wnt signaling plays a role in the initial formation of hair follicles, and that the latter stages of follicle formation are dependent on the Shh signal. Formation of the hair follicle is usually caused by activation of Wnt signaling and upregulation of β-catenin mRNA. The Shh activating signal also elevates Gli1 mRNA. Nuclear LEF1 expression occurs at the initial stages and increases after 24 h. However, epidermal cells induced by KGF or EGF contain high nuclear LEF1 expression at all time points. KGF and EGF noticeably inhibited several important signaling molecules produced by hair follicles, including Wnt (LEF1, DKK4), Shh single pathway, and delayed HFSC differentiation. Recently, Andreadis et al.[21] using a skin equivalent model system, demonstrated that KGF significantly facilitates the proliferation of epidermal cells inoculated in a cellular dermal matrix, which is associated with induced expression of α5β1 integrins and delayed keratin-10 expression. In the present study, HFSCs were observed to exhibit a low proliferation rate in the first 72 h after KGF treatment, especially in the 10 µg/L treatment group. When the culture time was longer than 72 h, DNA measurements showed that KGF treatment postponed apoptosis, but that high KGF concentrations accelerated cell aging. This may be related to excessive nutrient consumption in the in vitro culture medium. In cells treated with 10 µg/L KGF β-catenin expression increased slightly and GSK-3β expression decreased on day 7, as HFSCs underwent differentiation. LEF1 levels, however, declined at an early stage. How KGF switches the Wnt/β-catenin pathway from an activated state to an inhibited state is unclear. However, this data suggest that KGF concentration inversely correlates with LEF1 expression and represses HFSC differentiation and the formation of hair follicles. This result supports a direct association of KGF with the Wnt signaling pathway through reducing LEF1 levels. These data are consistent with the results of Richardson et al.[6] However, some of the links that mediate KGF regulation of the Wnt signaling pathway are not yet clear and require further investigation.
Financial support and sponsorship
This study is supported by the National Natural Science Foundation of China (No.30772099) and Beijing Municipal Natural Science Foundation (No.7112111).
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Yang B, Xiang MJ. Regulation of Wnt signaling pathway involved in the differentiation of hair follicle stem cells induced by lithium chloride. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu 2011;15:9202-6.
2. Taylor G, Lehrer MS, Jensen PJ, Sun TT, Lavker RM. Involvement of follicular stem cells in forming not only the follicle but also the epidermise. Cell 2000;102:451-61.
3. Waikel RL, Kawachi Y, Waikel PA, Wang XJ, Roop DR. Deregulated expression of c-Myc depletes epidermal stem cells. Nat Genet 2001;28:165-8.
4. DasGupta R, Kaykas A, Moon RT, Perrimon N. Functional genomic analysis of the Wnt-wingless signaling pathway. Science 2005;308:826-33.
5. Michel M, Török N, Godbout MJ, Lussier M, Gaudreau P, Royal A, Germain L. Keratin 19 as a biochemical marker of skin stem cells in vivo and in vitro: keratin 19 expressing cells are differentially localized in function of anatomic sites, and their number varies with donor age and culture stage. J Cell Sci 1996;109:1017-28.
6. Richardson GD, Bazzi H, Fantauzzo KA, Waters JM, Crawford H, Hynd P, Christiano AM, Jahoda CA. KGF and EGF signalling block hair follicle induction and promote interfollicular epidermal fate in developing mouse skin. Development 2009;136:2153-64.
7. Mullis KB, Faloona FA. Specific synthesis of DNA in vitro via a polymerase-catalyzed chain reaction. Methods Enzymol 1987;155:335-50.
8. Watt FM. Epidermal stem cells: markers, patterning and the control of stem cell fate. Philos Trans R Soc Lond B Biol Sci 1998;353:831-7.
9. Commo S, Gaillard O, Bernard BA. The human hair follicle contains two distinct K19 positive compartments in the outer root sheath: a unifying hypothesis for stem cell reservoir. Differentiation 2000;66:157-64.
11. Lapébie P, Gazave E, Ereskovsky A, Derelle R, Bézac C, Renard E, Houliston E, Borchiellini C. Wnt/β-catenin signaling and epithelial patterning in the homoscleromorph sponge oscarella. PLoS One 2009;4:1-6.
12. Lowell S, Jones P, Le Roux I, Dunne J, Watt FM. Stimulation of human epidermal differentiation by delta-notch signaling at the boundaries of stem-cell clusters. Curr Biol 2000;10:491-500.
13. Marchese C, Chedid M, Dirsch OR, Csaky KG, Santanelli F, Latini C, LaRochelle WJ, Torrisi MR, Aaronson SA. Modulation of keratinocyte growth factor and its receptor in reepithelializing human skin. J Exp Med 1995;182:1369-76.
14. Wexler EM, Geschwind DH, Palmer TD. Lithium regulates adult hippocampal progenitor development through canonical Wnt pathway activation. Mol Psychiatry 2008;13:285-92.
15. Rao AS, Kremenevskaja N, Resch J, Brabant G. Lithium stimulates proliferation in cultured thyroeytes by activating Wnt/beta-catenin signalling. Eur J Endocrina1 2005;153:929-38.
16. Hsu SC, Galceran J, Grosschedl R. Modulation of transcriptional regulation by LEF-1 in response to Wnt-1 signaling and association with beta-Catenin. Mol Cell Biol 1998;18:4807-18.
17. Zhu AJ, Watt FM. Beta-catenin signaling modulates proliferative potential of human epidermal keratinocytes independently of intercellular adhesion. Development 1999;126:2285-98.
18. DasGupta R, Rhee H, Fuchs E. A developmental conundrum: astabilized form of beta - catenin lacking the transcriptional activation domain triggers features of hair cell fate in epidermal cells and epidermal cell fate in hair follicle cells. J Cell Biol 2002;158:331-44.
19. Nguyen H, Rendl M, Fuchs E. Tcf3 governs stem cell features and represses cell fate determination in skin. Cell 2006;10:171-83.
20. Fathke C, Wilson L, Shah K, Kim B, Hocking A, Moon R, Isik F. Wnt signaling induces epithelial differentiation during cutaneous wound healing. BMC Cell Biol 2006;7:4.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Yang B, Wu XY, Ni J, Li BH, Deng LH, Xiang MJ. Modulation of Wnt/β-catenin signaling affects the directional differentiation of hair follicle stem cells. Plast Aesthet Res 2016;3:39-46. http://dx.doi.org/10.20517/2347-9264.2015.75
AMA Style
Yang B, Wu XY, Ni J, Li BH, Deng LH, Xiang MJ. Modulation of Wnt/β-catenin signaling affects the directional differentiation of hair follicle stem cells. Plastic and Aesthetic Research. 2016; 3: 39-46. http://dx.doi.org/10.20517/2347-9264.2015.75
Chicago/Turabian Style
Yang, Bin, Xiao-Ying Wu, Jian Ni, Bing-Hang Li, Li-Huan Deng, Meng-Juan Xiang. 2016. "Modulation of Wnt/β-catenin signaling affects the directional differentiation of hair follicle stem cells" Plastic and Aesthetic Research. 3: 39-46. http://dx.doi.org/10.20517/2347-9264.2015.75
ACS Style
Yang, B.; Wu X.Y.; Ni J.; Li B.H.; Deng L.H.; Xiang M.J. Modulation of Wnt/β-catenin signaling affects the directional differentiation of hair follicle stem cells. Plast. Aesthet. Res. 2016, 3, 39-46. http://dx.doi.org/10.20517/2347-9264.2015.75
About This Article
Copyright
Author Biographies

Data & Comments
Data

 Cite This Article 0 clicks
Cite This Article 0 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.