Recalcitrant cubital tunnel syndrome
Abstract
Ulnar nerve neuropathy at the elbow represents the second most frequent compression neuropathy of the upper extremity. Of the five different anatomical areas responsible for ulnar nerve compression at the elbow region, the epitrochlear-olecranon channel and Osborne’s arcade are the most common. An additional cause of nerve damage is a dynamic process in which the ulnar nerve dislocates anteriorly at the epitrochlear-olecranon level during elbow flexion, partially or completely, causing nerve friction and constriction leading to chronic neuropathic pain. Failure after primary surgery is generally secondary to procedural errors or technical omissions, frequently represented by incomplete nerve decompression, failure to recognize nerve instability after nerve decompression, loosening of the nerve anchor after superficial nerve transposition with consequent spontaneous nerve relocation in the epitrochlear-olecranon channel, perineural fibrosis and neurodesis, which creates new nerve compression. In association with the clinical evaluation, electromyography studies, magnetic resonance imaging and ultrasound are useful tools that may aid in the decision-making process when considering revision surgery. Superficial anterior transposition is the most commonly employed technique but also has a high failure rate, as opposed to anterior deep transposition that is the method of choice for many surgeons despite being more technically demanding. The results of revision surgery following recalcitrant ulnar nerve compression at the elbow are inferior to those obtained after primary surgery. Nonetheless, the clinical advantages remain relevant provided that the revision surgery is performed by an expert surgeon. To avoid misinterpretation, the patient is completely informed of the quality of results.
Keywords
Introduction
Ulnar nerve neuropathy at the elbow represents the second most frequent compression neuropathy of the upper extremity.[1] The ulnar nerve may be compressed at the elbow region in the following five different anatomical areas,[2] listed from proximal to distal: (1) arcade of Struthers, (2) the proximal epitrochlear region, (3) the epitrochlear-olecranon channel, (4) the fibrous arch between the humeral and ulnar portions of the flexor carpi ulnaris (FCU), better known as Osborne’s arcade, and (5) the vertical fibrous septum that originates from the ulna and separates the ulnar nerve and the ulnar part of the FCU from the pronator-flexor muscles innervated by the median nerve[3][Figure 1a and b]. The epitrochlear-olecranon channel and Osborne’s arcade or ligament are the most frequent areas of compression. An additional cause of nerve damage at the epitrochlear-olecranon level is nerve instability. This is a dynamic process in which the ulnar nerve dislocates anteriorly during elbow flexion, reaching the epicondylar crest (subluxation) or passing over it completely (luxation). Ligamentous laxity or the absence of stabilization mechanisms[4,5] causes a continuous snapping of the nerve over the epitrochlea, and in the case of complete anterior dislocation, the nerve kinks at the Osborne arcade, causing nerve friction and constriction leading to chronic neuropathic pain.[6] Various surgical procedures have been described for the treatment of cubital tunnel syndrome, including in situ decompressions,[7] medial epicondylectomy,[8] subcutaneous transposition,[9] intramuscular transposition,[10] and submuscular transposition.[11] However, as reported in the first description of this neuropathy by Panas[12] in 1878, surgical treatment frequently yields poor results and may worsen the initial clinical condition. In a recent study conducted by the American Association of Hand Surgery,[13] 61% of surgeons reported inferior results following surgical ulnar nerve decompression at the elbow compared with the results obtained following decompression after carpal tunnel release (44% satisfactory results vs. 88%, respectively).
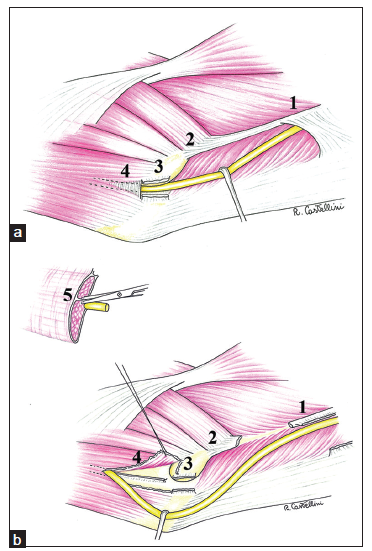
Figure 1. (a) There are five possible areas of ulnar nerve compression at the elbow level. (1): Arcade of Struthers; (2): proximal epitrochlear region; (3): epitrochlear-olecranon channel; and (4): fibrous arch between the humeral and ulnar parts of the flexor carpi ulnaris (FCU), better known as Osborne's arcade; (b) the figure shows the opening of Osborne's arcade (4) with the release of the septum between the ulnar part of the FCU and the flexor-pronator group (5). (1), (2) and (3) indicate the release of the other areas of decompression
According to the literature, no surgical technique may be defined as superior to the others.[14-16] The persistence of preoperative symptoms after surgery is defined as failure, whereas the reappearance of symptoms after a period of relief is defined as recurrence, and surgical revision is indicated in both cases.[15] Preoperative factors that may be associated with poor results after surgery depend on multiple elements, including an incorrect diagnosis,[17] advanced neuropathy with a neurological lesion and muscular atrophy,[18] coexisting pathologies such as double crush syndrome,[16] cervical spine radiculopathies,[19] thoracic outlet syndrome,[20] ulnar compression syndromes at the wrist, endocrine disorders such as diabetes mellitus or thyroid disease, and polyneuropathies, particularly if they are associated with muscular atrophy or decreased sensation.[13] Some of these conditions may mimic ulnar nerve syndrome at the wrist, which, in addition to the different symptoms noted by patients, may render recognition of ulnar neuropathy at the elbow difficult.
Causes of failure after primary surgery
Failure after surgery is mainly due to procedural errors or technical omissions, frequently represented by incomplete nerve decompression, failure to recognize nerve instability after the nerve has been decompressed, loosening of the nerve anchor after superficial nerve transposition with consequent spontaneous nerve relocation in the epitrochlear-olecranon channel, perineural fibrosis and neurodesis that creates a new site of nerve compression in areas different from those affected by the original compression, unintended injury to one or more sensory regional nerves or to the ulnar nerve itself, articular elbow instability due to unintended injury to the ulnar collateral ligament, or elbow stiffening in the flexed position.
It is widely accepted that when primary nerve decompression is performed, only one of the five possible sites of compression is generally found to actually be responsible for the nerve impingement,[2] and these sites are usually the epitrochlear-olecranon channel or the Osborne fibrous arcade. The surgeon's experience will generally determine the decision to proceed with a wide nerve decompression or to perform a limited procedure.[15]
The creation of a new nerve compression site may be realized when the anteriorly transposed nerve has not been widely released before the transposition. In fact, regardless of the method employed, when the nerve is anteriorly transposed, a new and nonanatomical path is created. It is therefore mandatory that the nerve lies in a soft and loose tissue bed such that no compression is endured by the nerve, which can occur when the medial intramuscular septum is not released or when the nerve kinks between the ulnar part of the FCU and the flexor and pronator muscle groups.[13,14,16,21] To avoid such compression, when transposition is performed, it is advisable to widely release the nerve by opening the cubital channel and Osborne’s arcade, removing the medial muscular septum, and opening both the arcade of Struthers and the septum between the ulnar stump of the FCU and the flexor-pronators.[13] When in situ nerve decompression is completed, dynamic nerve instability during elbow flexion may occur (nerve subluxation or luxation), and the omission of nerve stability evaluation is considered to be a technical error. According to the literature,[22] more than 50% of failures after simple decompression are due to the misdiagnosis of nerve instability. In cases of nerve instability, anterior nerve transposition, either deep or superficial, should be considered. Notably, nontraumatic nerve debridement and release, including that for a long tract (10-15 cm), does not damage the nerve or cause its devascularization, as was previously believed.[13,15,23]
Perineural fibrosis prevents the nerve from gliding during elbow excursion and may be related to patient predisposition or to improper intraoperative manipulation of the nerve.[14] Fibrosis following simple decompression can cause adherence of the nerve to the epitrochlear-olecranon channel and can additionally cause the channel’s closure due to the scarring at Osborne’s ligament. Fibrosis after anterior transposition may occur independently of the technique employed and usually occurs at the site of a technical error or omission. Following superficial nerve transpositions, in particular, fibrosis preferentially localizes to the anterior soft tissue area and the epitrochlear region. According to the literature, superficial transposition presents the highest percentage of failure, suggesting that this technique has some intrinsic limitations represented by the position of the nerve under the skin, in a relatively hypovascular tissue susceptible to trauma.[13,15] During anterior transposition, unintended injury to the subcutaneous antebrachial nerves may occur, leading to the formation of painful neuromas. During harvest of the ulnar nerve, in 61% of cases, 1 to 3 sensory nerves can be found proximal to the epicondyle (at a mean level of 1.8 cm from the epicondyle) or distally in 100% of cases (at a mean level of 3.1 cm from the epicondyle).[24,13] An unintended nerve lesion may produce one or more painful neuromas, creating a hyperalgesic or hyperesthetic area in the medial part of the elbow, jeopardizing achieving satisfactory results from the decompression. Clinical studies have reported a nerve lesion rate of up to 90%, which is thought to occur secondary to the difficulty in locating and protecting these nerves during dissection.[25,26] In contrast, lesions of the main trunk of the ulnar nerve are rare. To allow anterior nerve transposition, it is generally necessary to sacrifice the first motor fascicle to the FCU, which does not impair muscular function.[27] Medial elbow instability is quite uncommon but may occur following damage to the collateral ulnar ligament, particularly during medial epicondylectomy, or as a consequence of an excessively aggressive anterior submuscular transposition.[28] The ulnar collateral ligament is located just below the flexor-pronator group and originates, according to O’Driscoll et al.,[28] from the medial epicondyle. Elbow stiffness presents as a flexion contracture due to prolonged immobilization, inappropriate postoperative rehabilitation, or excessive fibrosis formation in the soft tissues. The extension lag is generally from 5° to 30°.[1,29,30] Stiffness occurs after deep transposition in 5-10% of cases[14,15,31] and is generally due to prolonged immobilization. Following primary deep transposition, the authors permit the patient to remove the orthesis from the 3rd to 7th day postoperatively, for 1-2 h/day to perform active motion. The orthesis is definitively removed 20 days after surgery. In cases of persistent stiffness, adequate rehabilitation, and medical therapy are typically required.
Clinical evaluation
After obtaining a thorough clinical history, it is mandatory to verify the absence of neurological disorders originating from the upper extremities and cervical spine.
The evaluation may then proceed to the elbow with the evaluation of the position and extent of the surgical scar, as well as palpation of the ulnar nerve along its course, which may be inside the epitrochlear-olecranon channel or medial to the epicondyle if nerve transposition was performed. Areas of tenderness and nerve instability during elbow articular motion are carefully investigated. In cases of ulnar neuropathy at the elbow, palpation along the course of the nerve may trigger a Tinel's sign with the characteristic spread of paresthesias along the area innervated by the nerve up to the 4th and 5th fingers or, in the case of antebrachial sensory nerve neuropathies, to the medial part of the elbow and the medial proximal third of the forearm. An important provocative test is the "pressure-flexion test"[32] in which pressure is exerted on the ulnar nerve for 1 min while the elbow is flexed. Sensitivity testing of the cutaneous territories of the ulnar and median nerves of the hand should be performed.[14] The clinical evaluation is completed by motor testing. Advanced neuropathy is indicated by muscular hypotrophy or atrophy of the intrinsic muscles innervated by the ulnar nerve. Typically, atrophy initially involves the first dorsal interosseous muscle and then extends to the hypothenar muscles. Assessment for the griffe deformity at the 4th and 5th fingers (hyperextension of the metacarpophalangeal joint and flexion of the interphalangeal joint), the Froment and Wartenberg signs (abduction of the 5th finger) and the inability to cross the long fingers (crossed finger test) complete the motor testing. When present, muscle impairment represents a negative prognostic factor. In general, the neurological signs are less severe, with no alterations in muscle tone, and identification is based solely on the clinical evaluation of asthenia and/or diminished strength of the intrinsic muscles innervated by the ulnar nerve.
Instrumental evaluation
Electromyography (EMG) is useful in the differential diagnosis to exclude radiculopathies, thoracic outlet syndrome, and median or ulnar nerve compression at the wrist. However, it fails to reveal neuropathies of the small sensory nerves in the elbow area. When preoperative EMG is performed, the results are particularly useful when assessing postoperative symptoms. In cases of recurrence, the neurological symptoms worsen, which correlates well with the conduction values, confirming the indication for surgical revision. Conversely, when worsening of the clinical condition is not confirmed by conduction studies, the indication for revision surgery should be dictated by the severity and persistence of symptoms. Notably, in cases of chronic axonal lesions, the conduction study results may be unchanged from the preoperative values while there is a slight improvement in the clinical condition.[14] Therefore, when faced with worsening symptoms and unchanged conduction studies, it is difficult to determine whether it is more useful to base the treatment decision on the symptoms, which would suggest surgery, or on the conduction studies, which would suggest a watchful waiting approach in the hopes of an eventual late recovery. Among 30 surgical revisions for the recalcitrant cubical tunnel, Gabel and Amadio[15] performed surgery in 9 patients who had normal EMGs, concluding that normal conduction values were not sufficient to exclude surgical revision. Ultrasound (US) examination may also aid surgeons in the decision-making process. In fact, the dynamic and static evaluation of the ulnar nerve may reveal morphological alterations to the nerve trunk and to the surrounding soft tissues. In the authors' experience, magnetic resonance imaging (MRI) offers less information than a well-performed US. In association with the clinical evaluation, these 2 diagnostic tools may assist in the decision-making process. When surgery is postponed, and symptoms do not improve in a short period, revision surgery should be reconsidered. In conclusion, in some cases, particularly in those of primary nerve instability, the pre- and postoperative conduction studies may be negative even in the presence of severe neuropathic symptoms. In these cases, US examination and MRI may aid in identifying areas of mechanical nerve injury that may indicate the need for surgery.
Indications for revision surgery
The persistence or worsening of neuropathic pain, a decrease in cutaneous sensitivity with paresthesias along the territory of the ulnar nerve in the hand, and muscle deficits despite conservative medical treatment are indications for revision surgery,[14,33] especially if they are associated with significant worsening of the conduction study results.
Techniques in revision surgery
The literature[14-16] regarding surgical revision of failed ulnar nerve decompression at the elbow is limited to a few retrospective studies and case reports.[26,29,34] According to these reports, superficial anterior transposition is the most commonly employed technique for primary surgery and presents a failure rate of 60-80%.[35,36]
The goal of revision surgery is essentially to debride the nerve of its surrounding fibrosis that is causing the compression and kinking. Neurolysis has an important role in the revision of failed surgery of the ulnar nerve at the elbow. However, neurolysis cannot be used as an isolated technique because simple scar excision activates a fibrotic reaction that, within a brief time interval, will compress the nerve again, leading to failure.[15] The removal of external perineural fibrosis is the primary indication for neurolysis.[37-39] When the fibrosis extends within the nerve, among the fascicles, internal neurolysis should be considered. However, in such cases, damage to the vascular supply of the internal nerve may occur, and severe nerve scarring may develop, jeopardizing the attainment of a good result even in cases of anterior nerve transposition.[15]
Medial epicondylectomy is not considered a satisfactory choice for revision surgery, as demonstrated by poor results in all of the cases treated by Goldberg et al.[40] These results may be due to the fibrotic and hypovascular tissue in which the nerve remains following the procedure.[19]
Anterior nerve transposition (subcutaneous, intramuscular, and submuscular) is the most commonly used revision technique after a failed nerve decompression,[14,19] in cases of nerve instability (nerve subluxation or luxation), after medial epicondylectomy, and following a failed anterior transposition.
Among nerve transpositions, subcutaneous transposition yields unpredictable results when used in revision surgery, and, for this reason, it is rarely used by surgeons. If the nerve is moved from the cubital channel to reduce mechanical stress, it is transposed to a relatively hypovascular area[13,15] where it is more exposed to direct trauma.[19] Gabel and Amadio[15] noted 12 poor results in 17 cases, whereas Caputo and Watson[34] reported a 50% rate of poor results using this technique.
Intramuscular transposition is rarely used in revision surgery, with only two cases described in the literature, both of which yielded unsatisfactory results.[15,18]
Submuscular transposition is widely used in revision surgery.[41-43] With this technique, good results may be achieved following failed simple decompressions, medial epicondylectomy, and failed superficial transpositions.[14-16] If performed using the proper technique, the results of anterior submuscular transposition are superior to those obtained with other techniques. In contrast, if this technique is employed for the treatment of failed submuscular transposition, the results are not satisfactory.[15] In such cases, division of the epitrochlear muscular bridge and superficial transposition of the nerve with associated external neurolysis yields good results.[34,44]
The following techniques are not effective and are rarely used: (1) the relocation of the nerve in the cubital tunnel has rarely been used by surgeons, as it is an ineffective method of treating recalcitrant ulnar nerve compression;[14,22,33] (2) the results of nerve isolation with synthetic material, such as silicon or polymeric substances, are unsatisfactory;[32,41] and (3) wrapping the ulnar nerve with autologous saphenous vein has been described with good results.[45,46] Additional studies are needed prior to declaring this technique as an effective method for the treatment of failed ulnar nerve decompression.
The authors prefer nerve isolation by means of muscle flaps or fat tissue[16,47] integrated with anterior submuscular transposition in cases of extensive perineural and soft tissue fibrotic reactions.
Technique preferred by the authors
Anterior deep transposition is the method of choice for many surgeons when revision ulnar nerve surgery is necessary. When approaching a revision surgery for recalcitrant ulnar nerve compression, it can be difficult to locate the area of nerve compromise. For this reason, a thorough exploration of all of the possible areas of compression is necessary, starting from the proximal arcade of Struthers to the deep septum between the FCU and the flexor-pronator group.[3,14,15] The skin incision in revision cases is generally longer than the initial incision, both in the proximal and distal directions. In the subcutaneous tissue, the identification of small regional sensory nerves may be difficult because they are frequently incorporated in the scar tissue from previous surgeries. It is not uncommon to find that one or more of these nerves have been severed. Possible neuromas must be removed,[3,14,19] and proximal nerve stumps must be cauterized and positioned in good-quality soft tissue, such as the triceps muscle.[3,14,19] In cases in which the ulnar nerve is entrapped in firm, fibrous scar tissue, it is advisable to begin the exploration proximal to the region of the previous incision to identify the nerve in a healthy area. Progressing distally, the nerve is then released from the scar. Depending on the technique used during the first surgery, the following three different situations may be encountered: (1) the nerve has been decompressed and is still in the epitrochlear-olecranon channel; (2) the nerve is outside of the epitrochlear-olecranon channel because dynamic nerve instability has occurred with recurrent anterior subluxation during elbow flexion, or because it has been transposed anteriorly in the subcutaneous tissue; or (3) the nerve is outside of the epitrochlear-olecranon channel because it has been transposed anteriorly under the flexor-pronator muscles. Regardless of where the nerve is located, the presence of scar tissue is a consistent pattern, which increases both the difficulty of the dissection and the risk of nerve damage. In these cases, identification of the nerve distal to the cubital channel at the FCU muscle entrance is recommended. From there, dissection proceeds in a distal to proximal direction. Once the nerve and potential compression areas have been released, the following different anatomical situations may be encountered: (1) the ulnar nerve was previously decompressed only and is still located in the epitrochlear-olecranon channel. Proceed with anterior submuscular transposition with myotomy of the epitrochlear muscles using the Z-lengthening technique.[16] It is of paramount importance that excision of the medial intermuscular septum and a complete opening of the distal septum between the FCU and the flexor-pronator muscle group are performed. If the transposition has been accomplished properly, the nerve will lie in its new location without areas of compression or kinking; (2) the ulnar nerve was previously transposed anteriorly and superficially, but there is currently severe fibrosis that renders nerve debridement difficult. If the intermuscular septum was not released during the previous surgery, the nerve passes over the septum, which dislocates the nerve from beneath, creating compression. In other cases, the nerve may be found atop the epitrochlear bone as a consequence of an erroneous transposition or of a failure of the soft tissue anchorage. This situation creates tension along the nerve, resulting in acute angulation and kinking of the nerve at Osborne’s arcade or at the deep distal septum at the level of the FCU. External neurolysis and submuscular transposition are performed as described in section A [Figures 2-5]; and (3) the ulnar nerve was previously already transposed. Surgery then commences with identification of the nerve proximal and distal to the scarred area, isolation of the nerve from the point of fibrosis up to the entrance in the epitrochlear muscles, and decompression of the arcade of Struthers, the intermuscular septum proximally, and the deep flexor-pronator septum distally. Release of the nerve at the entrance, exits, and beneath the muscular channel is then performed. The nerve is generally found to be entrapped by fibrotic tissue[44] in the new muscular channel and neurolysis in necessary. When neurolysis alone is insufficient for the release of the nerve or when the muscular channel has become fibrotic and does not provide adequate vascularization of the nerve, the muscular bridge is opened, and the nerve is transferred superficially.[34]
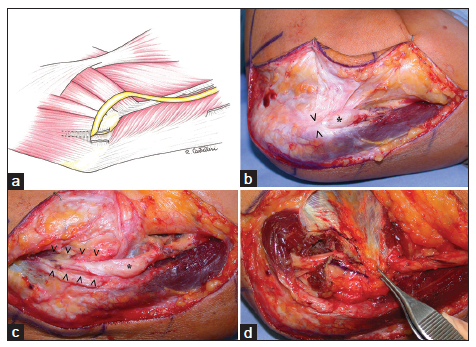
Figure 2. When decompression is insufficient, the nerve kinks at Osborne's arcade and is compressed by the intermuscular septum when transposed anteriorly. (a) The patient underwent two surgeries for simple nerve decompression. The nerve is dislocated anterior to the epitrochlear bone, presenting with a pseudoneuroma bulging (*) proximal to the compression area at Osborne's arcade level (>) which had not been previously released (<)(b) following decompression at zones 4 and 5 (refer to Figure 1) (<) and external neurolysis (*), nerve transposition may be performed; (c) anterior submuscular transposition using a muscle Z-lengthening procedure
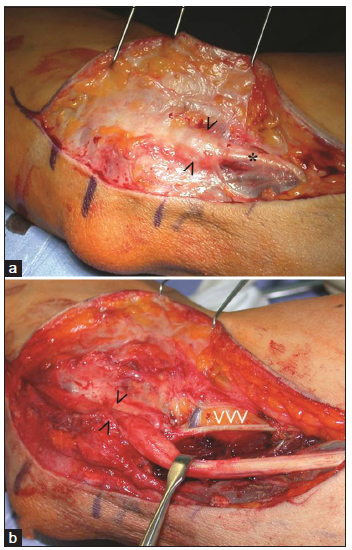
Figure 3. Failed nerve decompression treated with superficial anterior transposition. (a) The ulnar nerve (*) is fibrotic (<>), swollen, and hard to palpation; (b) the intermuscular septum (white arrows) and the distal deep septum in zone 5 (black arrows) were not released during the initial surgery
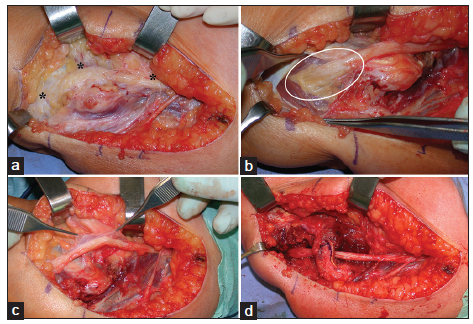
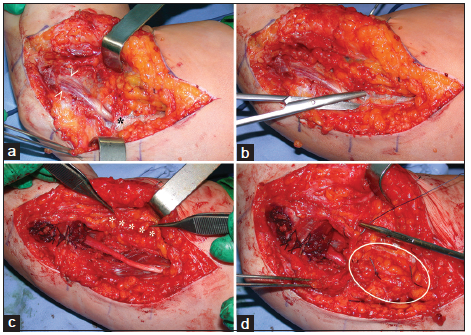
Figure 4. Failed superficial nerve transposition. (a) The nerve is taut over the epitrochlear bone (*) and kinks at the deep septum (zone 5) for an insufficient transposition; (b) nerve decompression by the release of the flexor carpi ulnaris and deep septum. The circled area shows the region of ischemic injury to the nerve secondary to compression; (c) after external neurolysishasbeen performed, the nerve is prepared for anterior submuscular transposition; (d) following transposition, the nerve lies in a soft and vascular tissue bed without tension
Figure 5. Failed superficial nerve transposition. (a and b) Arcade of Struthers (*) was not released during the first surgery, causing nerve kinking and compression. At the epitrochlear level, the nerve appears to lie within good tissue without tension (>); (c) following debridement, the nerve lies in a soft and vascular tissue bed without tension. An adipofascial flap is harvested (*); (d) anterior submuscular transposition with muscular Z-lengthening is performed. The adipofascial flap protects the nerve in the proximal epitrochlear region
Postoperative treatment
A brachial-metacarpal plaster cast is applied for 20 days, with the elbow at 100°-120°of extension. By the 3rd postoperative day, the patient is allowed to temporarily remove the plaster to perform careful active elbow flexion and extension movements. From the 7th day, the patient begins active careful supination with the elbow at 60°-90° of flexion. From the 15th day, supination with the elbow extended is permitted. The plaster is definitively removed 20 days postoperatively, and the patient is then placed under the care of a therapist.
Future directions
Ulnar nerve anatomy at the elbow region and pathophysiology of the compression syndrome are well-recognized. Nonetheless, failure following nerve decompression alone or with associated anterior nerve transposition still occurs. The failure to recognize dynamic ulnar nerve instability, idiopathic or induced after in situ nerve decompression, represents the most frequent procedural error leading to surgical failure. A thorough understanding of the mechanisms causing ulnar nerve compression and injury would reduce the rate of recurrence.
Conclusion
The results of revision surgery following recalcitrant ulnar nerve compression at the elbow are inferior to those obtained after primary surgery, particularly in patients over the age of 50 years, when conduction studies show muscle denervation, or when there is a history of multiple surgeries.[15] To avoid the misinterpretation that the partial resolution of preoperative symptoms is a failure of treatment, it is mandatory that the patient be completely informed prior to surgery.[16] Chronic axonal degeneration is frequently associated with marginal improvement of neuropathic pain, tenderness in the compression area, and hand dysesthesias,[14,16] without a high rate of complete restoration of sensitivity and muscle strength. Nonetheless, the clinical advantages remain relevant provided that an expert surgeon performs the revision surgery.
Financial support and sponsorship
Nil.
Conficts of interest
There are no conflicts of interest.
REFERENCES
1. Dawson DM, Hallett M, Millender LH. Entrapment Neuropathies. 1st ed. Boston: Little Brown; 1983. p. 88.
2. Amadio PC. Anatomical basis for a technique of ulnar nerve transposition. Surg Radiol Anat 1986;8:155-61.
3. Mackinnon SE, Novak CB. Operative Findings in reoperation of patients with cubitaltunnel syndrome. Hand (N Y) 2007;2:137-43.
4. Dellon AL. Musculotendinousvariation about the median humeral epicondyle. J Hand Surg Br 1986;11:175-81.
5. O'Driscoll SW, Horii E, Carmichael SW, Morrey BF. The cubital tunnel and ulnar neuropathy. J Bone Jiont Surg Br 1991;73B:613-7.
6. Childress HM. Recurrent ulnar-nerve dislocation at the elbow. Clin Orthop Relat Res 1975;108:168-73.
7. Osborne GV. The surgical treatment of tardy ulnar neuropathy. J Bone Joint Surg Br 1957;39B:782.
8. King T, Morgan FP. Late results of removing the medial humeral epicondyle for traumatic neuritis. J Bone Joint Surg Br 1959;41B:51-5.
9. Curtiss BF. Traumatic ulnar neuritis: transposition of the nerve. J Nerv Ment Dis 1898;25:480-4.
10. Adson AW. The surgical treatment of progressive ulnar paralysis. Minn Med 1918;1:455-60.
11. Learmont JR. A technique for transplanting the ulnar nerve. Surg Gynecol Obstetr 1942;75:792-3.
12. Panas J. Upon a rare case of ulnar nerve palsy. Arch Gen Med 1878;2:5-22. (in French)
13. Novak BC, Mackinnon SE. Selection of operative procedures for cubital tunnel syndrome. Hand (N Y) 2009;4:50-4.
14. Ruchelsman DE, Lee SK, Posner MA. Failed surgery for ulnar nerve compression at the elbow. Hand Clin 2007;23:359-71.
15. Gabel GT, Amadio PC. Reoperation for failed decompression of the ulnar nerve in the region of the elbow. J Bone Joint Surg Am 1990;72A:213-9.
16. Vogel BR, Nossaman BC, Rayan GM. Revision anterior submuscular transposition of the ulnar nerve for failed subcutaneous transposition. Br J Plast Surg 2004;57:311-6.
17. Puckett BN, Gaston RG, Lourie GM. A novel technique for the treatment of recurrent cubital tunnel syndrome: ulnar nerve wrapping with a tissue engineered bio scaffold. J Hand Surg Eur Vol 2011;36:130-4.
18. Gay JR, Love JG. Diagnosis and treatment of tardy paralysis of the ulnar nerve: based on a study of 100 cases. J Bone Joint Surg Am 1947;29:1087-97.
19. Jackson LC, Hotchkiss RN. Cubital tunnel surgery, complications and treatment of failures. Hand Clin 1996;12:449-56.
21. Spinner M. Nerve decompression. In: Morrey BF, editor. Master Techniques in Orthopaedic Surgery: The Elbow. New York: Raven Press; 1994. pp. 183-206.
22. Antoniadis G, Richter HP. Pain after surgery for ulnar neuropathy at the elbow: a continuing challenge. Neurosurgery 1997;41:585-9.
23. Heithoff SJ. Cubitaltunnel syndrome does not require transposition of the ulnar nerve. J Hand Surg Am 1999;24A:898-905.
24. Lowe JB 3rd, Maggi SP, Mackinnon SE. The position of crossing branches of the medial ante brachial cutaneous nerve during cubital tunnel surgery in humans. Plast Reconstr Surg 2004;114:692-6.
25. Mackinnon SE, Dellon AL, Hudson AR, Hunter DA. A primate model for chronic nerve compression. J Reconstr Microsurg 1985;1:185-95.
26. Rogers MR, Bergfield TG, Aulicino PL. The failed ulnar nerve transposition. Etiology and treatment. Clin Orthop Relat Res 1991;269:193-200.
27. Watchmaker GP, Lee G, Mackinnon SE. Intraneuraltopography of the ulnar nerve in the cubital tunnel facilitates anterior transposition. J Hand Surg Am 1994;19:915-22.
28. O'Driscoll SW, Jaloszynski R, Morrey BF, An KN. Origin of the medial ulnar collateral ligament. J Hand Surg Am 1992;17:164-8.
29. Broudy AS, Leffert RD, Smith RJ. Technical problems with ulnar nerve transposition at the elbow: findings and results of reoperation. J Hand Surg Am 1978;3:85-9.
30. Leffert RD. Anterior submusculartransposition of the ulnar nerves by Lear month technique. J Hand Surg Am 1982;7:147-55.
31. Janes PC, Mann RJ, Farmworth TK. Submuscular transposition of the ulnar nerve. Clin Orthop Relat Res 1989;238:225-32.
32. Novak CB, Lee GW, Mackinnon SE, Lay L. Provocative testing for cubital tunnel syndrome. J Hand Surg Am 1994;19:817-20.
33. Filippi R, Charalampaki P, Reisch R, Koch D, Grunert P. Recurrent cubital tunnel syndrome. Etiology and treatment. Minim Invasive Neurosurg 2001;44:197-201.
34. Caputo AE, Watson HK. Subcutaneous anterior transposition of the ulnar nerve for failed decompression of cubital tunnel syndrome. J Hand Surg Am 2000;25:544-51.
35. Mowlavi A, Andrews K, Lille S, Verhulst S, Zook EG, Milner S. The management of cubital tunnel syndrome: a meta-analysis of clinical studies. Plast Reconstr Surg 2000;106:327-34.
36. Dellon AL. Review of treatment results for ulnar nerve entrapment at the elbow. J Hand Surg Am 1989;14A:688-700.
37. Goth D. Animal experiment studies of neurolysis of peripheral nerves. Handchir Mikrochir Plast Chir 1987;19:212-6.
38. Graf P, Haewe W, Biemer E. Vascular supply of the ulnar nerve following neurolysis in the area of the elbow. Handchir Mikrochir Plast Chir 1986;18:204-6.
39. Rydevik B, Lundborg G, Nordborg C. Intraneuraltissue reactions induced by internal neurolysis. An experimental study on the blood-nerve barrier, connective tissues and nerve fibres of rabbit tibial nerve. Scand J Plast Reconstr Surg 1976;10:3-8.
40. Goldberg BJ, Light TR, Blair SJ. Ulnar neuropathy at the elbow: results of medial epicondilectomy. J Hand Surg Am 1989;14:182-8.
41. Posner MA. Compressive ulnar neuropathies at the elbow: II. Treatment. J Am Acad Orthop Surg 1998;6:289-97.
44. Dagregorio G, Saint-Cast Y. Simple neurolysis for failed anterior submuscular transposition of the ulnar nerve at the elbow. Int Orthop 2004;28:342-6.
45. Vardakas DG, Varitimidis SE, Sotereanos DG. Findings of exploration of a vein-wrapped ulnar nerve; report of a Case. J Hand Surg Am 2001;26:60-3.
46. Varitimidis SE, Riano F, Sotereanos DG. Recalcitrant post-surgical neuropathy of the ulnar nerve at the elbow: treatment with autogenous saphenous vein wrapping. J Reconstr Microsurg 2000;16:273-7.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Vigasio A, Marcoccio I, Morandini E. Recalcitrant cubital tunnel syndrome. Plast Aesthet Res 2015;2:176-82. http://dx.doi.org/10.4103/2347-9264.160881
AMA Style
Vigasio A, Marcoccio I, Morandini E. Recalcitrant cubital tunnel syndrome. Plastic and Aesthetic Research. 2015; 2: 176-82. http://dx.doi.org/10.4103/2347-9264.160881
Chicago/Turabian Style
Vigasio, Adolfo, Ignazio Marcoccio, Eleonora Morandini. 2015. "Recalcitrant cubital tunnel syndrome" Plastic and Aesthetic Research. 2: 176-82. http://dx.doi.org/10.4103/2347-9264.160881
ACS Style
Vigasio, A.; Marcoccio I.; Morandini E. Recalcitrant cubital tunnel syndrome. Plast. Aesthet. Res. 2015, 2, 176-82. http://dx.doi.org/10.4103/2347-9264.160881
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.