Simultaneous expander and deep inferior epigastric perforator reconstruction: indications and alloderm sling technique for protecting the anastomosis
Abstract
Aim: Autologous tissue is considered the “gold standard” for breast reconstruction today. However, little is known about deep inferior epigastric perforator (DIEP) flap reconstruction in combination with tissue expander (TE)/implant. The authors describe a series of combined DIEP flap/TE reconstruction, including its indications and technique to ensure protection of the pedicle during the expansion process.
Methods: Between January 2009 and December 2012, patients undergoing immediate DIEP with TE reconstruction were retrospectively reviewed. Oncologic, comorbid conditions, intraoperative, postoperative expansion, complications, and technique data points were collected. Photographs were taken postoperatively and patient’s satisfaction surveys were obtained to assess overall satisfaction.
Results: Five patients underwent immediate DIEP flap/TE reconstruction utilizing our alloderm sling technique. There were no complications to the pedicle, flap, expander, or mastectomy skin perioperatively or postoperatively. All patients describe being very satisfied, often with improved breast volume and projection as compared to their preoperative appearance.
Conclusion: The results of this study suggest that DIEP flap/TE reconstruction is safe, in particular when utilizing the alloderm sling technique, and should be considered in patients who lack sufficient abdominal tissue, have existing breast asymmetries, or do not desire the scar deformity of latissimus dorsi.
Keywords
Introduction
The overall goal of breast reconstruction is to recreate the most naturally appearing and feeling breasts for patients with breast cancer who are treated with mastectomies. Autologous reconstruction with either the transverse rectus abdominus myocutaneous (TRAM) flap, or more recently the deep inferior epigastric perforator (DIEP) flap, is now considered the “gold standard” for breast reconstruction due to its ability to recreate natural and aesthetic results. However, not all women have sufficient abdominal tissue to make an aesthetically appearing breast. Often, in these cases, an alternative technique for breast reconstruction is the latissimus dorsi (LD) flap with an expander/implant. Miller et al.[1] demonstrated that TRAM flap reconstruction can be simultaneously performed with placement of a tissue expander (TE) to provide improved volume and projection in safe manner for patients who have a thin body habitus with medium to large-sized breasts. Donor site and aesthetic outcomes proved to be statistically improved in patients who underwent TRAM/implant reconstruction when compared to LD/implant reconstruction.[2] Figus et al.[3] applied these same principles to the recent advance in autologous reconstruction the development of the DIEP flap. In concordance with the TRAM/implant literature, Figus et al.[3] demonstrated that placement of a sub-pectoral implant and DIEP flap can be safely performed and utilized in patients with insufficient abdominal tissue, in patients who need correction of breast asymmetries, and in patients that necessitate augmented volume and projection because they desire larger breasts. The main concern with the placement of the expander or implant simultaneously with a DIEP flap is potential injury to the pedicle. The authors describe a series of combined DIEP flap/expander reconstruction as well as the use of an alloderm sling to protect the pedicle from any immediate or delayed injury. The study was approved by review board of Yale University.
Methods
Between January 2009 and December 2012, over 250 DIEP flaps were performed, and 91% were bilateral reconstructions. When clinical assessment demonstrated inadequate abdominal tissue to reconstruct the patient’s desired breast size, discussions regarding the simultaneous use of an expander or implant were undertaken. Patients with a high probability of postoperative radiation were not offered the choice of a combined DIEP/expander procedure. However, history of preoperative radiation was not used as exclusion criteria. There were 5 patients who underwent simultaneous DIEP flap and expander/implant placement. These patient’s charts were retrospectively reviewed, and data points were collected. These data points include patient demographics, co-morbid conditions, pre- or postoperative radiation, primary disease, operative details, the final volume of the expander postoperatively, length of follow-up, and complications. All patients had postoperative photos taken 4-12 months postoperatively. Patients were asked to assess their satisfaction with the reconstruction using a four-point scale, with the number 1 defined as dissatisfied and the number 4 as very satisfied.
Operative technique: alloderm sling
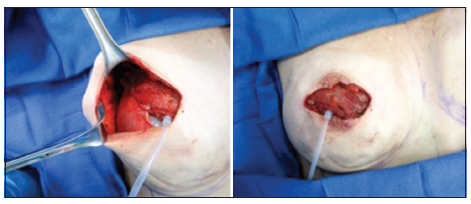
The borders of the breast are outlined preoperatively as is routinely done in expander-only reconstruction. Elevation of DIEP flaps occurs simultaneously while the general surgeons perform the mastectomy. Perforators are isolated, and the inferior epigastric pedicles are dissected and exposed. Once the mastectomies are complete [Figure 1], the subpectoral dissection is undertaken, and sizers are placed. Alloderm is routinely used infero-laterally to recreate the breast pocket and breast borders. The sizer is expanded to the desired final size, and the alloderm is secured in place [Figure 2]. The sizers are then deflated, and a window is created within the medial portion of the pectoralis, which allows access to the internal mammary artery and vein. At this time, dissection of the recipient vessels begins with the removal of the rib over the internal mammary vessels. Once the internal mammary vessels are dissected and exposed, the sizer is then replaced into the subpectoral pocket and re-inflated to the desired breast volume. A piece of alloderm (approximately 4 cm × 5 cm) is shaped to fit the defect between the ribs and the lateral edge of the pectoralis window. The alloderm is first secured to the rib periostium superiorly and inferiorly and is then draped along the lateral border of the pectoralis window [Figure 3]. The sizer is then exchanged with a smooth, round expander/implant, and a small pocket along the infero-lateral breast is dissected for placement of the external port. Saline is infused via the external port, and lateral digital pressure on the expander/implant confirms appropriate position and integrity of the alloderm sling [Figure 4]. The expander/implant is then deflated to allow room for the microvascular anastomosis of the internal mammary vessels to the deep inferior epigastric pedicle. The DIEP flap is then secured in place, the overlying mastectomy skin is approximated, and a subjective amount of saline is infused into the TE/implant. This is all done while ensuring that: (1) the alloderm sling is competent; (2) the DIEP flap appearance and Doppler signal do not change; and (3) the mastectomy skin appears well perfused. As in routine breast reconstruction, the patient is then placed in an upright position, and the appropriate placement of the DIEP flap and expander/implant with resolution of any volume asymmetries is confirmed. Two 10-flat Jackson Pratt drains are placed in each breast: one within the alloderm breast pocket and the other outside of the alloderm infero-laterally. Postoperatively, the patients are placed on DVT prophylaxis until day of discharge and antibiotics until the drains are discontinued. As historically described with TRAM/implant procedures, expansion was initiated 4-6 weeks postoperatively.[4] Permanent smooth, round silicone gel implants were exchanged once the patient’s desired breast volume was met.
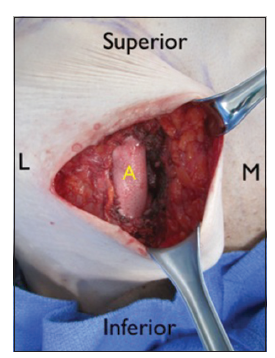
Figure 2. Standard technique of tissue expander reconstruction: placement of subpectoral sizer and securing alloderm inferiolaterally to the released pectoralis muscle edge. The sizer will be replaced with placement of a tissue expander with an external port
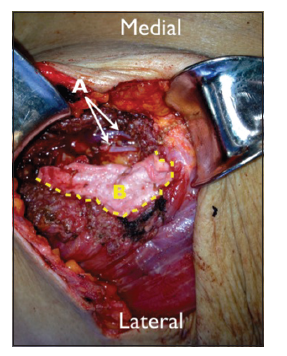
Figure 3. A small window within the pectoralis muscle is made medially and the internal mammary vessesl are dissected and exposed. The alloderm sling is then sutured in place to form the lateral "wall" of the window within the pectoralis or the new medial boundary to the subpectoral expander
Figure 4. (A) Dissected and exposed recipient internal mammary artery and vein; (B) alloderm sling sutured in place to the rib periostium superiorly and inferiorly as well as to the medial edge of the pectoralis. The subpectoral expander is therefore limited from migrating medially towards the internal mammary vessels by the alloderm sling
Results
Five patients underwent combined DIEP/TE reconstruction. The average age was 50 years, and all patients had early disease, few comorbidities, and were not smokers [Table 1]. Four patients had no prior reconstruction, one patient had prior bilateral TE placement with postoperative radiation and subsequent infections that led to significant deformities bilaterally and her desire for secondary reconstruction.
Cohort demographics, comorbid conditions and oncologic characteristics
| Age (years) | Height (m) | Weight (kg) | BMI | Co-morbidities | Smoker | Preoperative radxn | Prior recon | Breast cancer stage | Bilat recon |
|---|---|---|---|---|---|---|---|---|---|
| 41 | 1.54 | 68.03 | 28.3 | None | No | No | No | I | Yes |
| 50 | 1.70 | 58.51 | 20.4 | None | No | No | No | 0 | No |
| 55 | 1.60 | 58.51 | 23.4 | None | No | Yes | Yes | II | Yes |
| 49 | 1.70 | 71.66 | 25.1 | None | No | No | No | I | Yes |
| 50 | 1.65 | 57.15 | 20.7 | HTN, DM | No | No | No | 0 | Yes |
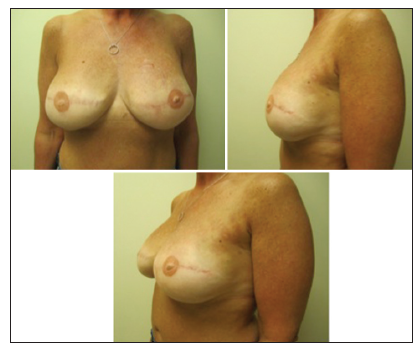
Of the 5 immediate DIEP flap/TE patients, four patients underwent bilateral reconstructions, and 1 patient had a stacked DIEP flap with implant placement for a unilateral defect [Figure 5]. There were no reoperations, episodes of venous congestion, hematomas, partial or total flap losses, seromas, infections, or expander/implant leaks [Table 2]. There were no instances of expander/implant extrusion, migration or palpability. The average final expander size was 325 mL ± 132.5 mL (range: 200-400 mL). All patients have undergone an uneventful expander/implant exchange procedure, and none has necessitated a revision or fat grafting procedures to correct asymmetries. All patients describe being “very satisfied” with their reconstructive result (score 4) with subjective improvement in volume and projection of their breasts. Length of follow-up ranged from 6 to 18 months [Figure 6].
Cohort operative details and complications
| Patient | Expander size | Initial saline infused | Hematoma | Seroma | Injury to flap | Infection | Asymm | Satisfaction score (1-4) |
|---|---|---|---|---|---|---|---|---|
| 1 | 350 mL | 0 mL | No | No | No | No | No | 4 |
| 2 | 250 mL | 150 mL | No | No | No | No | No | 4 |
| 3 | 250 mL | 0 mL | No | No | No | No | No | 4 |
| 4 | 250 mL | 200 mL | No | No | No | No | No | 4 |
| 5 | 275/400 mL | 200/275 mL | No | No | No | No | No | 4 |
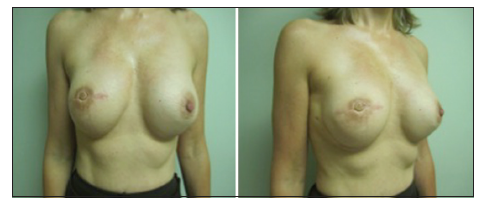
Figure 5. Right stacked deep inferior epigastric perforator (DIEP) flap with immediate subpectoral 250 mL expander and nipple reconstruction. Left breast augmentation with subpectoral 250 mL gel implant. Three months postoperative
Discussion
Plastic surgeons are constantly searching for ways to optimize techniques and perfect results. Koshima et al.[1] was the first to improve upon the TRAM flap design by isolating the abdominal tissue on perforators and sparing the muscle. Not surprisingly, the DIEP flap has since gained widespread popularity and made inroads as the gold standard for autologous reconstruction, providing both a lasting result and breasts that appear and feel natural.
However, not all women who desire autologous reconstruction have sufficient abdominal tissue to recreate an aesthetic appearing breast. Alternative donor sites for autologous reconstruction include the gluteal region, posterior thigh, and medial thigh, however, these sites generally contribute even less tissue than the abdomen. Historically, the standard procedure for thin women desiring autologous reconstruction was a combined LD flap/implant breast reconstruction. Kronowitz et al.[2] recently demonstrated that a superior alternative to the LD flap/implant procedure in this patient population is a combined TRAM/implant procedure. Eighteen TRAM/implant patients demonstrated a higher aesthetic score when compared to the LD/implant group by both the patients and a panel of blinded judges. The overall impression by the blinded judges was that the TRAM flap more accurately “recreated the breast with the implant contributing less to the overall shape” when compared with the LD/implant group. Serletti and Moran[4] corroborated these findings by suggesting that the subcutaneous tissue of the TRAM flap more accurately resembles native breast tissue, and unlike the LD, will not atrophy over time. In fact, any fluctuations in weight will result in volume changes in the TRAM flaps.
In addition to superior aesthetic results, the TRAM/implant group experienced fewer donor site complications when analyzed against the LD/implant group.[2] The scar from an LD flap tends to widen over time, and while it can be concealed behind a bra, the unilateral contour deformity of missing the LD muscle can be apparent.[1] On the other hand, the TRAM or DIEP flap donor site scar does not tend to widen over time, has no contour deformity, and can be easily concealed with most under-garments.
While TRAM/implant procedures offer optimal aesthetic results when compared to standard techniques such as LD/implant, it is technically more challenging. Furthermore, concern lies in potentially injuring the TRAM flap when combined with implant placement. However, multiple authors have demonstrated that in experienced hands, TRAM flap reconstruction can be combined with implant placement without any occurrences of microvascular thrombosis or flap failure.[5,6]
Figus et al.[3] was the first to describe successfully combining DIEP flap reconstruction with immediate implant placement. Fourteen patients were selected as candidates for DIEP/implant reconstruction based on similar criteria to that previously reported in the literature; these patients were then prospectively followed. Ten patients had implants placed subpectorally at the time of the DIEP flap, and 4 patients had the implants placed in a delayed fashion directly under the DIEP flap. Their preferred vessel for anastomosis was the thoracodorsal artery and vein. They did not experience any total flap losses or episodes of microvascular thrombosis, however, they did experience an immediate postoperative infection and hematoma that led to partial flap loss and removal of the implant. In addition, they describe an accidental transection of the internal mammary vessels while placing a delayed implant directly beneath the flap. The aesthetic results were analyzed and revealed “very satisfied” and “excellent” outcomes.[3]
Commentary in response to this data argued against placement of immediate implants or expanders with DIEP flaps for concerns that the implant would either directly or indirectly compromise the vascularity of the flap.[7] This concern for injury to the pedicle, whether immediately or during the expansion, is the basis behind the development of our alloderm sling technique. We propose that this technique can prevent potential injury to the pedicle whether intraoperatively, in a reoperation or any delayed procedures. While total flap loss and microvascular thrombosis events have yet to be described in the literature with combined TRAM/implant procedures, we believe that the alloderm sling technique acts as a safety net to prevent the subpectoral implant/expander from injuring the pedicle. Clearly, this is less of a concern if the preferred recipient vessel is to the thoracodorsal artery and vein,[3] but the alloderm technique may have prevented the reported transection of the inferior mesenteric artery/inferior mesenteric vein (IMA/IMV).
In our cohort, patients desired larger breasts than the overlying skin envelope could maintain and therefore we chose to place smooth, round subpectoral expanders with an external port (the external port was chosen to limit potential injury to the flap during expansion). The subpectoral placement is the standard technique for expander reconstruction, leading to fewer capsular contractures and better concealing the outline of the expander.[1,2,4,8] Initially, saline was not immediately infused into the expander for fear that it could indirectly injure the flap through pressure. However, in our subsequent cases various amounts of saline were infused into the expander and changes in the implantable Doppler, flap, and overlying mastectomy skin was directly visualized. If any of these variables were negatively affected by the expansion, the volume was decreased. In our cohort, there were no reoperations, partial or total flap losses, hematomas, infections, implant failures, or asymmetries. In contrast to previous reports, our cohort demonstrated no seromas in relation to the initial expansion of the expander at the time of surgery.[3] We do, however, place drains within the expander pocket and continue them until the output is less than 30 mL for 24 h. Furthermore, the cohort described is the initial experiences with the described technique, and thus is currently too small to make translatable conclusions.
Our data supports the proven safety of combined TRAM and DIEP/implant procedures as well as the excellent aesthetic results achieved with this procedure.[1,2,4,8] Furthermore, there is evidence that combining an implant with autologous tissue appears to reduce implant related complications in previously irradiated breasts.[6] If a patient has a unilateral defect, stacking two DIEP flaps on top of one another can often provide sufficient tissue to recreate a single breast. However, in cases where the patient desires larger breasts and the contralateral breast needs augmentation [Patient 4, Table 2 and Figure 4], combining a stacked DIEP flap with an expander/implant is an option. This technique achieves the volume and projection the patient desires by utilizing an implant, and gives a natural feel and appearance by utilizing an overlying DIEP flap. As suggested by Figus et al.,[3] an implant/expander can be combined with a DIEP flap to address preoperative breast asymmetries. In our cohort, one patient demonstrated these asymmetries [Patient 5, Table 2], and a 275 mL expander was placed in one side and a 400 mL expander in the other to provide a more symmetric appearance. A noted alternative to simultaneous augmentation with DIEP flaps is to address any asymmetries is fat grafting. However, the advantage of using an implant is to correct the asymmetry immediately and eliminate the need for multiple revisional surgeries.
Additional reconstructive techniques being used in patients with inadequate abdominal tissue include the superior gluteal artery perforator (SGAP) flap, transverse upper gracilis (TUG) flap or the profunda femoris artery perforator (PAP) flap. These techniques, however, tend to be more complex and time intensive. The dissection of the muscle for the SGAP is technically difficult and possesses a relatively short vascular pedicle. There is also the possibility for contour deformity and asymmetry of the buttocks, particularly in the case of unilateral breast reconstruction.[9] Although the TUG flap involves a relatively easy dissection, it provides a rather small skin paddle and thin, fat pad, which limits its utility for reconstruction of small to medium-sized breasts. Furthermore, atrophy of the gracilis muscle may cause secondary volume and contour deformities, requiring additional corrections.[10,11] The PAP flap has a relatively long vascular pedicle and the scar may be hidden in the lower buttock crease. However, there may be scar tenderness causing problems with sitting, visibility of the scar in swimwear or underwear, and asymmetrical donor site with unilateral breast reconstruction.[12] Fat grafting is an option for increasing volume, but it requires multiple procedures and often does not allow for large volume augmentation in excess of 150 mL or more. Although this is certainly an option for revision and touch ups, the authors routinely do not use large volume fat grafting to augment the volume of DIEP flap reconstruction.
This cohort has the limitations inherent to any small cohort and retrospective review, which include the difficulties in making generalizations from a small sample size. Despite the potential benefits of combined DIEP/expander reconstruction in patients desiring larger breasts or with insufficient abdominal tissue, women who smoke or have significant co-morbidities may not be appropriate candidates for this technique.
In this retrospective review, we demonstrate that combined DIEP/expander reconstruction is safe and provides excellent long-term aesthetic results. We report our experience to further support the notion that combined DIEP/implant procedures can have superior aesthetic results when compared to many of the alternative procedures in this select group of patients.[2,5,6] In addition, we describe a technique that may assist surgeons in preventing any inadvertent injury to the pedicle when performing simultaneous DIEP flap/expander reconstruction and using the IMA/IMV as the recipient vessels. The alloderm technique may provide plastic surgeons with the confidence to offer patients this technique as an alternative to traditional LD/implant techniques. This technique offers the ability to use an expander in women whose overall breast size is not yet finalized and who soft tissue envelope will not support a sizeable implant.
Financial support and sponsorship
Nil.
Conficts of interest
There are no conflicts of interest.
REFERENCES
1. Koshima I, Moriguchi T, Soeda S, Tanaka H, Umeda N. Free thin paraumbilical perforator-based flaps. Ann Plast Surg 1992;29:12-7.
2. Kronowitz SJ, Robb GL, Youssef A, Reece G, Chang SH, Koutz CA, Ng RL, Lipa JE, Miller MJ. Optimizing autologous breast reconstruction in thin patients. Plast Reconstr Surg 2003;112:1768-78.
3. Figus A, Canu V, Iwuagwu FC, Ramakrishnan V. DIEP flap with implant: a further option in optimising breast reconstruction. J Plast Reconstr Aesthet Surg 2009;62:1118-26.
4. Serletti JM, Moran SL. The combined use of theTRAM and expanders/implants in breast reconstruction. Ann Plast Surg 1998;40:510-4.
5. Tadiparthi S, Alrawi M, Collis N. Two-stage delayed breast reconstruction with an expander and free abdominal tissue transfer: outcomes of 65 consecutive cases by a single surgeon. J Plast Reconstr Aesthet Surg 2011;64:1608-12.
6. Roehl KR, Baumann DP, Chevray PM, Chang DW. Evaluation of outcomes in breast reconstructions combining lower abdominal free flaps and permanent implants. Plast Reconstr Surg 2010;126:349-57.
7. Chia HL, Breitenfeldt N, Canal AC, Malata CM. Implant augmentation after perforator flap breast reconstruction. J Plast Reconstr Aesthet Surg 2010;63:e172-3.
8. Fisher J, Hammond DC. The combination of expanders with autogenous tissue in breast reconstruction. Clin Plast Surg 1994;21:309-20.
10. Arnez ZM, Pogorelec D, Planinsek F, Ahcan U. Breast reconstruction by the free transverse gracilis (TUG) flap. Br J Plast Surg 2004;57:20-6.
11. Wechselberger G, Schoeller T. The transverse myocutaneous gracilis free flap: a valuable tissue source in autologous breast reconstruction. Plast Reconstr Surg 2004;114:69-73.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Craig ES, Shah A, Persing S, Salomon J, Fusi S. Simultaneous expander and deep inferior epigastric perforator reconstruction: indications and alloderm sling technique for protecting the anastomosis. Plast Aesthet Res 2015;2:63-8. http://dx.doi.org/10.4103/2347-9264.153201
AMA Style
Craig ES, Shah A, Persing S, Salomon J, Fusi S. Simultaneous expander and deep inferior epigastric perforator reconstruction: indications and alloderm sling technique for protecting the anastomosis. Plastic and Aesthetic Research. 2015; 2: 63-8. http://dx.doi.org/10.4103/2347-9264.153201
Chicago/Turabian Style
Craig, Elizabeth Stirling, Ajul Shah, Sarah Persing, Jeffrey Salomon, Stefano Fusi. 2015. "Simultaneous expander and deep inferior epigastric perforator reconstruction: indications and alloderm sling technique for protecting the anastomosis" Plastic and Aesthetic Research. 2: 63-8. http://dx.doi.org/10.4103/2347-9264.153201
ACS Style
Craig, ES.; Shah A.; Persing S.; Salomon J.; Fusi S. Simultaneous expander and deep inferior epigastric perforator reconstruction: indications and alloderm sling technique for protecting the anastomosis. Plast. Aesthet. Res. 2015, 2, 63-8. http://dx.doi.org/10.4103/2347-9264.153201
About This Article
Copyright
Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.