The role of radiotherapy in the treatment of oral cavity cancer
Abstract
Radiotherapy plays a critical role in the treatment of oral cavity squamous cell carcinoma as monotherapy in early stage cancer or combined with surgery and/or chemotherapy in advances ones. Recent developments in the imaging of cancer and radiation technology have allowed developing more precise delivery of treatment with recent data demonstrating improvement in survival and lessening of adverse toxics effects of radiation. This review will focus in the recent advances and current state-of-the-art in radiation oncology both external beam radiotherapy and brachytherapy. As complexity of cancer treatments increases a close coordination between head-neck surgeons and radiation oncologist is needed due to a significant proportion of patients will be treated with combined modality therapy.
Keywords
Introduction
Although surgery is the recommended treatment for oral cavity squamous cell carcinoma (OCSCC),[1] radiotherapy (RT) plays a capital role in the treatment of OCSCC either exclusively or as adjuvant after surgery.
RT may be administered using two techniques, which, in turn, are likely to be combined together in the specific case of OCSCC: external beam radiotherapy (EBRT) and brachytherapy (BT). Usually patients with early stage disease are treated exclusively radical radiotherapy; however, patients with unresectable or advanced disease will receive radiotherapy plus chemotherapy or targeted therapy with monoclonal antibodies against epidermal growth factor receptor (EGFR) in order to enhance the cytotoxic effect of radiation.
The present manuscript is a revision of most important manuscripts concerning a large and extended bibliography has been performed in order to elucidate the current role of RT in the treatment of patients with squamous cell carcinoma of the oral cavity.
Radiotherapy techniques overview
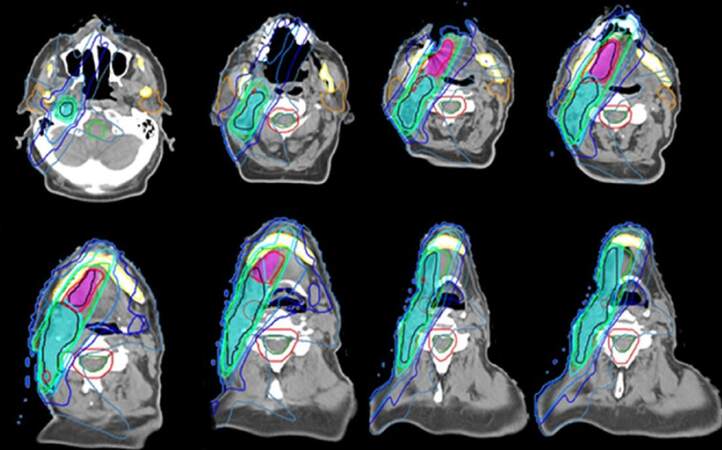
Currently standard EBRT is based on the assessment of target volumes to irradiate and organs at risk to protect in 3D-computed tomography (CT) simulation plus multimodal images (e.g., positron emission tomography-CT, magnetic resonance imaging).[2-6] Delivery of treatment should be based on intensity modulated radiation therapy[7] (IMRT) which involves the use of multiple computer-aided beams of inhomogeneous radiation, allow dose shaping the spatial shape of treatment volume, improving the coverage of target area and the protection of healthy tissue [Figure 1]. When using IMRT different treatment volumes (e.g., macroscopic tumor vs. elective nodal levels) receive a different dosage during the same fraction, without increasing the number of RT sessions, so the intensity of treatment is adjusted to each volume of interest by dose gradients.[8] IMRT compared with traditional 2D-EBRT has been shown to improve toxicity[9] and survival[10] in patients with head neck cancer.
Figure 1. Postoperative intensity modulated radiation therapy plan for an oral tongue squamous cell carcinoma pT2 pN1 M0. High dose encompass risk volumes (blue: ipsilateral nodal bed. purple: tumor bed) while sparing healthy organ: parotids glands (orange) spinal cord (green) mandible and larynx (courtesy of Dr. Enrique Miragall from Fundación ERESA)
Traditionally BT implant has been performed with low dose rate (LDR) by inserting iridium needles (192Ir) mainly; this technique has been gradually displaced by the so-called high dose rate (HDR) BT [Figure 2] due to its advantages of radiation protection of medical personnel, better dose distribution and shorter duration of treatment.[11] However, the accelerated treatment and high dose per fraction used in HDR could lead to a decrease in the therapeutic ratio because of the risk of complications in extreme cases.[12] Liu et al.[13] conducted a meta-analysis to compare HDR BT vs. LDR BT in the treatment of OCSCC. No statistically significant difference was found in the odds ratio (OR) between the group of patients treated with LDR or HDR in terms of local recurrence OR = 1.12, mortality OR = 1.01, and complications grade
3-4 OR = 0.86.
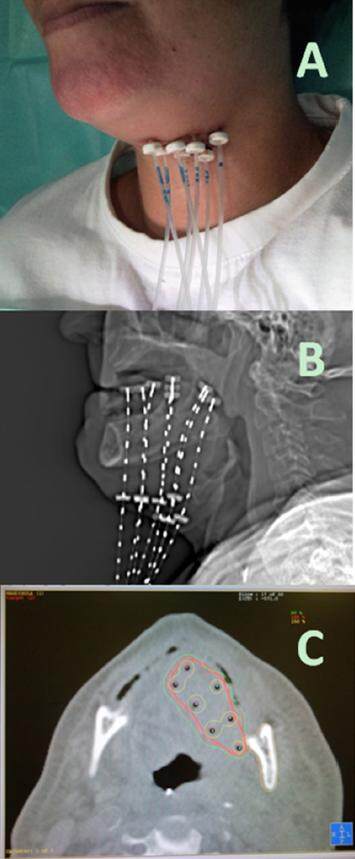
Figure 2. High dose rate brachytherapy for oral tongue carcinoma. (A) showing external outward appearance of percutaneous catheters for after loading technique; (B) digital radiographic reconstruction of the implant for planning purposes; (C) computed tomography axial view showing high isodoses lines covering tumor bed but sparing contralateral tongue, mandible and lips (courtesy of Dr. José Luis Guinot from Instituto Valenciano de Oncología)
The equivalent fractionation and total dosing between LDR and HDR is unknown. Neither the Groupe Européen de Curiethérapie-European Society for Radiotherapy and Oncology (GEC-ESTRO)[11] nor the American Brachytherapy Society[14] came to publish a consensus, although they recommended not to exceed a dose 6 Gy per fraction. In the comparative meta-analysis of Liu et al.,[13] the mean dose administered was 66.17 Gy in LDR group and 50.75 Gy in the HDR. Radiobiological studies suggest that the optimal dose for exclusive HDR is about 50 Gy[15,16] consistent with data from Liu et al.[13] GEC-ESTRO has published recommendations[17] for the calculation of equivalent doses between different protocols and BT techniques.
The main indication for combining EBRT and BT is the need to irradiate the cervical lymph node chains when the risk of involvement is significant due to the primary site,[18] tumor thickness greater than 4 mm[19] and stage cT2-T3.
Stages I-II
In treating early OCSCC the best results were obtained when BT is part of the treatment, either exclusively or as tumor overdose after EBRT.[11] Evidence supporting this practice is based entirely on retrospective series. Even with the advent of IMRT, BT administration is advantageous in terms of shaping and uniformity of dose[20] and tumor control.[21] Table 1 summarizes the results of selected series of OCSCC patients treated with radical BT with or without EBRT.[12,22-47] In the case of floor of mouth stage cT1 local control is 93-95% and 72-88% for stage cT2. Local control in cancer of mobile tongue is achieved in 79-97% for stage I and 65-95% for stage II.
Radical brachytherapy for oral cavity squamous cell carcinoma only, not including other head and neck sites
| Studies | No. of patients | Site | Technique | Radiotherapy schedule | 5-year local control (%) | 5-year survival (%) |
|---|---|---|---|---|---|---|
| Lau et al.[12] 1996 | 27 | Tongue | HDR | BT only, 45.5 Gy @6.5 Gy | 53 | 92 |
| Leung et al.[22] 2002 | 19 | Tongue | HDR | BT only, 45-63 Gy (median 55 Gy, ten fractions) | 94.7 (4-year) | NS |
| Martínez-Monge et al.[23] 2009 | 8 | Oral cavity | HDR | EBRT 45 + BT 16 Gy @4 Gy | 86 (7-year) | 52.3 (7-year) |
| Guinot et al.[24] 2010 | 33 | Tongue | HDR | EBRT 55 + BT 18 Gy @3 Gy | 79 | 74 |
| 17 | Tongue | HDR | BT only 44 Gy @4 Gy | |||
| Inoue et al.[28] 2001 | 25 | Tongue | HDR | BT only 60 Gy @6 Gy | 87 | |
| 26 | LDR | BT only 70 Gy | 84 | |||
| Yamazaki et al.[29] 2003 | 58 | Tongue | HDR | BT only 60 Gy @6 Gy | 84 | |
| 341 | LDR | BT only 70 Gy | 80 | |||
| Yamazaki et al.[30] 2007 | 80 | Tongue | HDR | EBRT 37 Gy + BT 36-60 Gy | 85 | |
| 217 | LDR 226Ra | EBRT 29 Gy + BT 59-94 Gy | 74 | |||
| 351 | LDR 192Ir | EBRT 29 Gy + BT 59-94 Gy | 72 | |||
| Kakimoto et al.[32] 2011 | 14 | Tongue (T3) | HDR | EBRT 30 Gy + 60 Gy | 71 (2-year) | |
| 61 | LDR | EBRT 30 Gy + 72 Gy | 62 | |||
| Akiyama et al.[33] 2012 | 17 | Tongue | HDR | BT only 54 Gy @6 Gy | 88 | |
| 34 | BT only 60 Gy @6 Gy | 88 | ||||
| Donath et al.[34] 1995 | 13 | Oral cavity | HDR | BT only 45-50 Gy @4.5-5 Gy | 92 | |
| Inoue et al.[35] 1998 | 16 | Floor or Mouth | HDR | EBRT 30-40 Gy + BT 36-48 Gy @6 Gy | 94 | |
| 41 | LDR 198Au | EBRT 30-40 Gy + BT 65-85 Gy | 69 | |||
| Matsumoto et al.[36] 2013 | 67 | Tongue | HDR | EBRT 20 Gy + BT 50 Gy | 94 | 88.7 |
| Khalilur et al.[37] 2011 | 125 | Tongue | LDR | 70 Gy | 86 | |
| Vedasoundaram et al.[38] 2014 | 33 | Bucal mucosa | HDR | BT only 38.5 Gy @3.5 Gy EBRT 50 Gy + BT 21 Gy @3.5 Gy | 92.3 | |
| Lee et al.[39] 2014 | 16 | Oral cavity | HDR | BT only 50 Gy @5 Gy EBRT 50 Gy +BT 35 Gy @5 Gy | 84 (3-year) | 70 |
| Tuček et al.[40] 2014 | 20 | Tongue | HDR | BT only 54 gy @3 Gy | 85 | 75 |
| Oota et al.[25] 2006 | 433 | Tongue | LDR | BT only 70 Gy EBRT 35 Gy + BT 60 Gy | 85.6 | |
| Pernot et al.[41] 1996 | 552 | Tongue | LDR | BT only 66 - 75 Gy | 90.5 | 71.5 |
| 207 | FOM | |||||
| Lefebvre et al.[42] 1994 | 429 | OC | LDR | BT only 66 Gy | 90 | |
| Mazeron et al.[43] 1991 | 279 | Tongue & FOM | LDR | BT only 60-70 Gy | 87-93 | |
| Marsiglia et al.[44] 2002 | 160 | FOM | LDR | 88-93 | 76 | |
| Dearnaley et al.[45] 1991 | 149 | Tongue & FOM | LDR | BT only | 90 | |
| Fujita et al.[46] 1999 | 207 | Tongue | LDR | EBRT 30 Gy + BT 50-60 Gy BT only 65-70 Gy | 82.2 | |
| Bachaud et al.[27] 1994 | 94 | Tongue & FOM | LDR | EBRT 48 Gy + BT 26 Gy BT only 66 Gy | 61 | |
| Ihara et al.[47] 2005 | 117 | Tongue | LDR | EBRT 30 Gy + BT 65 Gy BT only 70 Gy | 59.2 | 54 |
Stages III-IV
Usually the treatment of advanced cancer of OCSCC has been included in the group of “advanced head and neck cancer” (AHNC) because of this the indications, techniques and results from clinical trials are fully applicable.
Radiotherapy alone
Modification of EBRT fractionation allows to intensify radiation dose by means of two way: (a) increase in the total dose with hyperfractionation; and (b) shorten the duration of using accelerated fractionation radiotherapy.
Two meta-analyses of randomized trials[48,49] comparing conventional fractionation EBRT (CF-EBRT) against modified fractionation EBRT (MF-EBRT) were published. Bourhis et al.[48] analyzes all clinical trials for all locations of the head and neck (12.6% of cases OCSCC), however data are presented separately depending on location; Glenny et al.[49] examined trials for oral cavity and oropharynx cancer only.
Bourhis et al.[48] found a statistically significant benefit in terms of overall survival (OS) HR = 0.92 in favor of MF-EBRT as well as an improvement in locoregional control (LRC) HR = 0.82. Hyperfractionated EBRT was also significantly better in terms of OS than accelerated EBRT, with an absolute benefit of 8% at 5 years.
Glenny et al.[49] reported that MF-EBRT, reduces overall mortality, HR = 0.86, and increased LRC HR = 0.79. Trials included as "purely hyperfractionated" also showed a significant gain in OS compared with the accelerated fractionation HR = 0.78.
Radiotherapy and chemotherapy combination
Pignon et al.[50] performed a meta-analysis on benefit of chemotherapy (CMT) added to EBRT in head and neck cancer (MACH-NC). Overall improvement in OS was demonstrated when chemotherapy is added to radiation. Maximum benefit was found when CMT is administered concurrently with EBRT: 5-year OS 8% improvement. The benefit of CRT is applicable to all locations of the head and neck.[51]
Two randomized trials have investigated whether the addition of chemotherapy to MF-EBRT is superior to CRT (CF-EBRT) or MF-EBRT alone.
The French Group of Radiation Oncology of Head and Neck Cancer (GORTEC)[52] randomized patients into three arms: accelerated EBRT alone, CF-EBRT plus CMT or accelerated EBRT plus CMT. No statistically significant difference was found between the treatment groups at 3-year OS: 32.2% vs. 37.6% vs. 34.1%, nor distant metastasis (DM). However, both locoregional failure (LCF) (49.9% vs. 41.7% vs. 45.4%) and progression-free survival (PFS) (32.2% vs. 37.6% vs. 34.1%) were significantly lower in the accelerated EBRT arm. Mucosal acute toxicity and the need for feeding tube were significantly higher in patients treated with MF-EBRT.
In the second study by the Radiation Therapy Oncology Group (RTOG)[53] patients were randomized to MF-EBRT alone or FM-EBRT plus CMT. No statistically significant difference was found in 8-year OS (48% in both arms) LRF (37% vs. 39%) PFS (42% vs. 41%) or DM (15% vs. 13 %) No statistically significant differences in toxicity were found either. In conclusion, no advantage in combining MF-EBRT and CMT have been proved so far.
Target therapy
EGFR over expression leads to decreased survival and increased risk of local and regional recurrence in head and neck cancer.[54] The inhibition of EGFR by monoclonal antibodies (cetuximab) associated with EBRT in patients with non-operated AHNC showed an increase 5-year OS (46% vs. 36%) and LRC (47% vs. 34%) compared with EBRT alone.[55] Notably in this trial did not include patients with OCSCC therefore clinical benefit in this group of patients is presently unknown.
Nowadays, the standard of treatment for non-operable AHNC, including OCSCC, is EBRT plus CMT despite the fact that its benefit in OS and LRC probability equals of the hyperfractionated-EBRT. The reasons that have led to this situation are basically two: (1) logistics, due to the consumption of resources and the drawbacks associated with treating patients twice a day, for 7-8 weeks; and (2) the development of high conformation techniques as IMRT, which allow to exploit the different sensitivity to radiation of the tumor and healthy tissues using a single fraction per day with a shorter overall time of treatment, usually 5-6 weeks.
Postoperative radiation therapy
Adjuvant EBRT
The value of postoperative radiotherapy (PORT) for AHNC, was established by Fletcher and Evers[56] and Marcus et al.[57] in 1970’s. The evidence that proves the usefulness of PORT has been based on retrospective studies of large groups of patients. Due to the inherent bias in such kind of studies the survival benefit of PORT is not fully confirmed, although there are no doubts about the gain in LRC.
Lundahl et al.[58] performed a retrospective, matched-pair analysis to compare surgery alone vs. surgery plus PORT. They found significant improvement in LRC and OS in the PORT group.
Lavaf et al.[59] and Kao et al.[60] analyzed patients with AHNC stage III-IV treated with surgery alone or surgery plus PORT from Surveillance Epidemiology End Results (SEER) data base. In multivariate analysis the survival benefit of PORT vs. surgery alone at 5-year was significant in both non-locally advanced tumors with lymph node metastasis (51.6% vs. 40.6%) as in the case of locally advanced tumors with lymph node metastasis (35.3 % vs. 25.2%). Overall PORT significantly improved OS by 11% and cancer-specific survival by 8.6%. They showed a greater reduction in the risk of death in stage N2b-N3 compared to N1-N2a (HR = 0.62, 0.78 and 0.82 respectively). The magnitude of the reduction was larger for tumors of the oropharynx, hypopharynx and larynx compared to oral cavity (HR = 0.72, 0.66 and 0.62 respectively) Patients with lymph node metastasis and any tumor sites, all benefited from the administration of PORT although the gain is greater in high-risk disease.
Whereas PORT is not routinely indicated in patients with HNSCC stage pT1-2 pN1[61] because there is not definitive data supporting that approach. Moergel et al.[62] published a meta-analysis of studies in order to elucidate the role of PORT in patients pN1 with oral cavity and oropharynx primaries. Any firm conclusions could be drawn due to the heterogeneity of the studies, although it was evident more mortality (not significant) in the group treated with PORT (44% vs. 34%). Shrime[63] analyzed the benefit of PORT in patients with OCSCC pT1-2 pN1. PORT improved OS at 5 years [41.4% vs. 54.2% (P < 0.001)] of note PORT improved OS in T2 tongue and floor of mouth subgroup [52.3% vs. 37.9% (P = 0.002) and 39.9% vs. 17.7% (P = 0.003), respectively] but not significantly in T1 subgroup.
The hypothesis that early nodal metastases may express a more aggressive biology supports adjuvant therapy in stage III.[64]
Risk factors for locoregional recurrence
Extracapsular extension (ECE) in cervical lymph node metastases and the involvement of surgical resection margins (ISRM) are the most important prognostic factors for risk of LRC and death.
RTOG[65] stratified patients treated with PORT into 3 risk groups according to the presence of ECE, 2 or more lymph nodes with metastasis or ISRM. Group I were those with no more than 2 nodes affected without ECE; group II included patients with more than 2 positive lymph nodes or ECE, negative margins; group III comprised patients with ISRM. Significant difference was found in the rate of loco-regional recurrence at 5 years between groups I, II and III of 17%, 27% and 67% respectively and median OS at 5.6 years, 2 years and 1.5 years, respectively.
Langendijk et al.[66] conducted a multivariate analysis to define different prognostic groups based on pathologic features a series of 801 patients with AHNC treated with PORT. The final model identified 6 prognostic factors and grouped the patients into 3 risk groups [Table 2]. This model was validated by the Dutch Head and Neck Oncology Cooperative Group (DHNOCG) in a multicenter study.[67]
Risk groups definition according multivariate analysis (recursive partitioning analysis) by Langendijk
| RPA class | Definition | VUMC series | VUMC series | ||
|---|---|---|---|---|---|
| LRC 5-year | OS 5-year | LRC 5-year | OS 5-year | ||
| Class I (intermediate risk) | Free margins without ECE | 92% | 67% | 82% | 60% |
| Class II (high risk) | T1, T2, T4 tumors with close or positive surgical margins; One lymph node metastasis with ECE | 78% | 50% | 82% | 50% |
| Class III (very high risk) | T3 tumors with close or positive surgical margins; Multiple lymph node metastases with extranodal spread; N3 neck | 58% | 37% | 63% | 36% |
Nowadays, there is consensus[68] to identify patients at high risk of recurrence after surgery who benefit from PORT: (1) major criteria: ECC or ISRM; and (2) minor criteria: inadequate surgical margins (< 5 mm), ≥ 2 lymph nodes metastases (N2b-N3), stage pT3-T4 even with negative margins, in primary oral cavity, metastases in levels IV and V, presence of PNI or LVI.
Perineural infiltration
One of most controversial point is the value of PORT when there is PNI but the absence of other factors associated with risk of recurrence. Neither in the analysis of Jonkman et al.[66] or its further validation,[67] PNI was found to be an independent prognostic factor. Bur et al.[69] after a systematic review on the potential benefit of PORT in patients with PNI concluded that there is insufficient evidence to recommend PORT routinely in these cases. The author suggests that in case of infiltration of cranial nerves or multiple PNI, PORT might be justified. PNI is associated with increased risk of nodal recurrence, therefore it is recommended to treat the neck in this scenario.
Time factor in PORT
Evidence exists suggesting that the risk of LRC is higher in patients with AHNC when receiving PORT more than 6 weeks after surgery,[70] OR: 2.89. Further work[71] confirmed elevated RR 1.28 on LRC and decrease in OS (RR: 1.16) per month of delay. The waiting list to start radiotherapy has negative effect on the prognosis according to a Dutch national study.[72]
The accelerated repopulation during radiotherapy is a cause of treatment failure, that can be increased by the undue prolongation of radiation therapy.[73] González Ferreira et al.[74] found an loss in LRC of 1-1.2% per extra-day or 12-14% per extra-week. Prolongation of radiotherapy negatively interferes LRC and OS even in case of CRT.[74]
Finally, the overall treatment time (OTT) from the day of surgery to the end of PORT showed prognostic significance for the LRC and OS in a randomized trial when the entire duration of treatment was greater than 13 weeks.[75] No other randomized studies have been published that would confirm this finding, a retrospective series found no prognostic association in the OTT with LRC neither OS.[76]
Intensification of adjuvant treatment
The value of dose escalation with PORT as a function of risk of recurrence has been explored in 2 prospective randomized trials. Peters and Withers[77] showed the benefit of a dose of 63 Gy in 1.8 Gy fractions in patients with ECE, positive or inadequate surgical margins. Ang et al.[75] published the results of a multicenter trial that randomized 151 patients with high-risk criteria (ECE and 2 or more additional criteria) between accelerated concomitant boost radiotherapy 63 Gy in 5 weeks or the same dose in conventional fractionation in 7 weeks. The accelerated treatment showed significantly improvement in LRC and OS when the interval between surgery and the start of PORT was not stretched or if the duration of the whole treatment (surgery plus PORT) no exceeded 13 weeks. Role of accelerated PORT is not firmly established, a confirmatory phase III Dutch trial (POPART CKTO 2003-11) is currently in recruitment period.
A meta-analysis[78] on the benefit of postoperative CRT confirmed the reduction in RR of LRC (RR = 0.59) and death (RR = 0.80) and improvement in median survival (from 22-32 months to 40-72 months). The authors state that the patients included in those trials were under 70 years and with good performance status, so the impact of the CRT in patients aged 70 or older with associated co-morbidities is unknown.[50,78] A pooled analysis[79] of 2 phase III trials from RTOG[80] and the European Organization for Research and Treatment of Cancer (EORTC)[81] on the role of the postoperative CRT in adjuvant treatment of the SCCHN, confirmed that patients with ECE or ISMR were those who most benefit obtained with the administration of PORT chemoradiation in terms of risk reduction in LRC (48%) in time to progression (23%) and mortality (30%). Other pathological features commonly used to define patients at risk of relapse) were not so decisive influencing LRC, OS, neither benefit of CRT. However a updating of the RTOG 9501 trial[82] found no significant difference between patients treated with PORT alone and those treated CRT regarding LRC (28.8% vs. 22.3%, P = 0.1), DFS (19.1% vs. 20.1%, P = 0.25) or OS (27% vs. 29.1%, P = 0.31); an unplanned analysis on the subgroup of patients with ECE or ISMR showed that the combined treatment improved LRC (33.1% vs. 21%, P = 0.02) and DFS (12.3% vs. 18.4%, P = 0.05) but not OS (19.6% vs. 27.1%, P = 0.07).
On the technical aspects of PORT
PORT administration is a particular challenge from the point of view of the radiation oncologist. Anatomy distortion due to tumor resection, the presence of reconstruction flaps, prosthetic material and the position of scars may influence routes of dissemination and hamper assessing volumes at risk to irradiate. Due the narrow conformation of dose to the target volume by IMRT, failure to design an adequate treatment volume will leave untreated areas of unrecognized risk; on the contrary excessively large volumes lead to higher radiation exposure of healthy tissue regions with consequent toxicity.[83,84] Close collaboration between the radiation oncologist and head and neck surgeon is imperative when interpreting the pathological findings and surgical technique used; the engagement with radiologist and pathologist will be necessary in most cases. There is currently no international consensus on standard volumes for PORT irradiation in AHNC, but there are some guidelines published.[85-88].
Adjuvant brachytherapy
In the specific case of OCSCC, PORT can be performed in fully or partly by BT reaching an equivalent dose of 60-66 Gy (LDR or HDR) on the tumor bed when surgical margins are infiltrated (stages pT1-T3) EBRT is administered alone when cervical nodes are at risk or primary surgical bed is not amenable for BT. Adjuvant BT results are summarized in Table 3.[89-93] While in early-stage OCSCC treated with radical RT adding BT plays a critical role in cure and local control, it is not the case of adjuvant setting (early nor advanced stage OCSCC) as either LRC and OS are equivalent between PORT-EBRT or PORT-BT. Table 4 shows recent published studies on patients with advanced OCSCC treated with PORT IMRT-based.[94-103]
Adjuvant brachytherapy for oral cavity squamous cell carcinoma
| Studies | No. of patients | Site | Technique | RT schedule | 5-year local control (%) | 5-year-overall survival (%) |
|---|---|---|---|---|---|---|
| Goineau et al.[89] 2015 | 112 | Tongue | LDR | EBRT: 60-66 Gy + BT 50-55 Gy | 76 | 56 |
| Petera et al.[90] 2015 | 30 | Tongue | HDR | BT only 54 Gy @3 Gy | 85.4 (3-year) | 73 (3-year) |
| FOM | ||||||
| Lapeyre et al.[91] 2004 | 82 | Tongue | LDR | EBRT 48 Gy + BT 24 Gy BT only 60 Gy | 81 | 80 |
| FOM | ||||||
| Pernot et al.[92] 1995 | 97 | Tongue | LDR | NS | 84 | 79 |
| FOM | ||||||
| Fietkau et al.[93] 1991 | 50 | Tongue | LDR | EBRT 55 Gy + BT 24.5 Gy | 94 (crude) | 84 (crude) |
| FOM |
Postoperative intensity modulated radiation therapy for oral cancer
| Studies | No. of patients | Site | RT schedule | Loco-regional control | Overall survival |
|---|---|---|---|---|---|
| Chan et al.[94] 2013 | 180 | Oral | 83 (2-year) | 65 (2-year) | |
| Hoffman et al.[95] 2015 | 18 | Oral cavity | 66 Gy IMRT with SIB | 78 (5-year) | 77 (5-year) |
| Sher et al.[96] 2011 | 30 | Oral | 64.13 Gy IMRT sequential boosting | 91 (2-year) | 85 (2-year) |
| Gomez et al.[97] 2011 | 35 | Oral | 60 Gy IMRT SIB | 77 (3-year) | 74 (3-year) |
| Chakraborty et al.[98] 2015 | 75 | Oral | IMRT volumetric | 88.9 (2-year) | 80.5 (2-year) |
| Studer et al.[99] 2012 | 99 (R0-1) 17 (R2) | Oral (primary + recurrent) | 70 Gy IMRT SIB | 80 (4-year) 35 (4-year) | 79 (4-year) 30 (4-year) |
| Collan et al.[100] 2010 | 40 | Oral | 58 Gy IMRT sequential boosting | 87.5 (5-year) | 75 (5-year) |
| Geretschläger et al.[101] 2012 | 53 | Oral | 66 Gy IMRT sequential boosting | 79 (3-year) | 73 (3-year) |
| Yao et al.[102] 2007 | 55 (5 p definitive RT) | Oral | 66 Gy IMRT SIB | 82 (3-year) | 82 (3-year) |
| Daly et al.[103] 2011 | 37 (7 definitive RT) | Oral | 66 Gy IMRT SIB | 53 (3-year) | 60 (3-year) |
Acknowledgments
The photographs illustrating in this article was kindly provided by Dr. Enrique Miragall from Fundación ERESA [Figure 1] and Dr. José Luis Guinot from Instituto Valenciano de Oncología [Figure 2].
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. National Comprehensive Cancer Network. Fort Washington PA. c2015. NCCN Practice Guidelines in Oncology: Head and Neck Cancers, version 1. 2015 [updated 2015 Dec 05; cited 2016 Feb 22] Available from http://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf.
2. Unger K, Riaz N, Chen A, Lee NY. Oral Cavity Cancer. In: Lee NY, Liade JL, editors. Target Volume Delineation and Field Setup. A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy. Berlin-Heidelberg: Springer-Verlag; 2013. pp. 35-44.
3. Grégoire V, Ang K, Budach W, Grau C, Hamoir M, Langendijk JA, Lee A, Le QT, MaingonP, Nutting C, O'Sullivan B, Porceddu SV, Lengele B. Delineation of the neck node levels for head and neck tumors: a 2013 update. DAHANCA, EORTC, HKNPCSG, NCIC CTG, NCRI, RTOG, TROG consensus guidelines. Radiother Oncol 2014;110:172-81.
4. Liu SH, Chao KS, Leu YS, Lee JC, Liu CJ, Huang YC, Chang YF, Chen HW, Tsai JT, Chen YJ. Guideline and preliminary clinical practice results for dose specification and target delineation for postoperative radiotherapy for oral cavity cancer. Head Neck 2015;37:933-9.
5. Leclerc M, Lartigau E, Lacornerie T, Daisne JF, Kramar A, Grégoire V. Primary tumor delineation based on (18)FDG PET for locally advanced head and neck cancer treated by chemo-radiotherapy. Radiother Oncol 2015;116:87-93.
6. Brouwer CL, Steenbakkers RJ, Bourhis J, Budach W, Grau C, Grégoire V, van Herk M, Lee A, Maingon P, Nutting C, O'Sullivan B, Porceddu SV, Rosenthal DI, Sijtsema NM, Langendijk JA. CT-based delineation of organs at risk in the head and neck region: DAHANCA, EORTC, GORTEC, HKNPCSG, NCIC CTG, NCRI, NRG Oncology and TROG consensus guidelines. Radiother Oncol 2015;117:83-90.
7. Chao KSC, Mohan R, Marinetti TD, Dong L. Intensity-Modulated Radiation Treatment Techniques and Clinical Applications. In: Halperin EC, Wazer DE, Perez CA, Brady LW, editors. Perez and Brady's Principles and Practice of Radiation Oncology. 6th ed. Philadelphia: Lippincott Williams and Wilkins; 2013. pp. 221-45.
8. Orlandi E, Palazzi M, Pignoli E, Fallai C, Giostra A, Olmi P. Radiobiological basis and clinical results of the simultaneous integrated boost (SIB) intensity modulated radiotherapy (IMRT) for head and neck cancer: a review. Crit Rev Oncol Hematol 2010;73:111-25.
9. Marta GN, Silva V, de Andrade Carvalho H, de Arruda FF, Hanna SA, Gadia R, da Silva JL, Correa SF, Vita Abreu CE, Riera R. Intensity-modulated radiation therapy for head and neck cancer: systematic review and meta-analysis. Radiother Oncol 2014;110:9-15.
10. Beadle BM, Liao KP, Elting LS, Buchholz TA, Ang KK, Garden AS, Guadagnolo BA. Improved survival using intensity-modulated radiation therapy in head and neck cancers: a SEER-Medicare analysis. Cancer 2014;120:702-10.
11. Mazeron JJ, Ardiet JM, Haie-Méder C, Kovács G, Levendag P, Peiffert D, Polo A, Rovirosa A, Strnad V. GEC-ESTRO recommendations for brachytherapy for head and neck squamous cell carcinomas. Radiother Oncol 2009;91:150-6.
12. Lau HY, Hay JH, Flores AD, Threlfall WJ. Seven fractions of twice daily high dose-rate brachytherapy for node-negative carcinoma of the mobile tongue results in loss of therapeutic ratio. Radiother Oncol 1996;39:15-8.
13. Liu Z, Huang S, Zhang D. High dose rate versus low dose rate brachytherapy for oral cancer--a meta-analysis of clinical trials. PLoS One 2013;8:e65423.
14. Nag S, Cano ER, Demanes DJ, Puthawala AA, Vikram B, American Brachytherapy Society. The American Brachytherapy Society recommendations for high-dose-rate brachytherapy for head-and-neck carcinoma. Int J Radiat Oncol Biol Phys 2001;50:1190-8.
15. Petera J, Matula P, Paluska P, Sirak I, Macingova Z, Kasaova L, Frgala T, Hodek M, Vosmik M. High dose rate versus low dose rate brachytherapy in the treatment of tongue carcinoma - a radiobiological study. Neoplasma 2009;56:163-8.
16. Yamazaki H, Inoue T, Yoshida K, Yoshioka Y, Furukawa S, Kakimoto N, Shimizutani K, Inoue T. Brachytherapy for early oral tongue cancer: low dose rate to high dose rate. J Radiat Res 2003;44:37-40.
17. Van Limbergen E, Joiner M, Van der Kogel A, Dörr W. Radiobiology of LDR, HDR, PDR and VLDR Brachytherapy. In: Van Limbergen E, Pötter R, Hoskin P, Baltas D, editors. The GEC ESTRO Handbook of Brachytherapy. 2nd ed (ebook version 1 12/11/2015). Available from: http://www.estro.org/search?type=Activities&q=radiobiology+ldr+hdr&alloptions=on.
18. Grégoire V, Coche E, Cosnard G, Hamoir M, Reychler H. Selection and delineation of lymph node target volumes in head and neck conformal radiotherapy. Proposal for standardizing terminology and procedure based on the surgical experience. Radiother Oncol 2000;56:135-50.
19. Huang SH, Hwang D, Lockwood G, Goldstein DP, O'Sullivan B. Predictive value of tumor thickness for cervical lymph-node involvement in squamous cell carcinoma of the oral cavity: A meta-analysis of reported studies. Cancer 2009;15:1489-97.
20. Sresty NV, Ramanjappa T, Raju AK, Muralidhar KR, Sudarshan G. Acquisition of equal or better planning results with interstitial brachytherapy when compared with intensity-modulated radiotherapy in tongue cancers. Brachytherapy 2010;9:235-8.
21. Studer G, Brown M, Bredell M, Graetz KW, Huber G, Linsenmeier C, Najafi Y, Riesterer O, Rordorf T, Schmid S, Glanzmann C. Follow up after IMRT in oral cavity cancer: update. Radiat Oncol 2012;7:84.
22. Leung TW, Wong VY, Kwan KH, Ng TY, Wong CM, Tung SY, Leung LC, O SK. High dose rate brachytherapy for early stage oral tongue cancer. Head Neck 2002;24:274-81.
23. Martínez-Monge R, Gómez-Iturriaga A, Cambeiro M, Garrán C, Montesdeoca N, Aristu JJ, Alcalde J. Phase I-II trial of perioperative high-dose-rate brachytherapy in oral cavity and oropharyngeal cancer. Brachytherapy 2009;8:26-33.
24. Guinot JL, Santos M, Tortajada MI, Carrascosa M, Estellés E, Vendrell JB, Muelas R, Chust ML, Mengual JL, Arribas L. Efficacy of high-dose-rate interstitial brachytherapy in patients with oral tongue carcinoma. Brachytherapy 2010;9:227-34.
25. Oota S, Shibuya H, Yoshimura R, Watanabe H, Miura M. Brachytherapy of stage II mobile tongue carcinoma. Prediction of local control and QOL. Radiat Oncol 2006;1:21.
26. Fujita M, Hirokawa Y, Kashiwado K, Akagi Y, Kashimoto K, Kiriu H, Matsuura K, Ito K. Interstitial brachytherapy for stage I and II squamous cell carcinoma of the oral tongue: factors influencing local control and soft tissue complications. Int J Radiat Oncol Biol Phys 1999;44:767-75.
27. Bachaud JM, Delannes M, Allouache N, Benchalal M, Alzieu C, David JM, Serrano E, Daly-Schveitzer NJ. Radiotherapy of stage I and II carcinomas of the mobile tongue and/or floor of the mouth. Radiother Oncol 1994;31:199-206.
28. Inoue T, Inoue T, Yoshida K, Yoshioka Y, Shimamoto S, Tanaka E, Yamazaki H, Shimizutani K, Teshima T, Furukawa S. Phase III trial of high- vs. low-dose-rate interstitial radiotherapy for early mobile tongue cancer. Int J Radiat Oncol Biol Phys 2001;51:171-5.
29. Yamazaki H, Inoue T, Yoshida K, Yoshioka Y, Furukawa S, Kakimoto N, Shimizutani K, Inoue T. Brachytherapy for early oral tongue cancer: low dose rate to high dose rate. J Radiat Res 2003;44:37-40.
30. Yamazaki H, Inoue T, Yoshida K, Yoshioka Y, Furukawa S, Kakimoto N, Shimizutani K. Comparison of three major radioactive sources for brachytherapy used in the treatment of node negative T1-T3 oral tongue cancer: influence of age on outcome. Anticancer Res 2007;27:491-7.
31. Ihara N, Shibuya H, Yoshimura R, Oota S, Miura M, Watanabe H. Interstitial brachytherapy and neck dissection for Stage III squamous cell carcinoma of the mobile tongue. Acta Oncol 2005;44:709-16.
32. Kakimoto N, Inoue T, Inoue T, Murakami S, Furukawa S, Yoshida K, Yoshioka Y, Yamazaki H, Tanaka E, Shimizutani K. Results of low- and high-dose-rate interstitial brachytherapy for T3 mobile tongue cancer. Radiother Oncol 2003;68:123-8.
33. Akiyama H, Yoshida K, Shimizutani K, Yamazaki H, Koizumi M, Yoshioka Y, Kakimoto N, Murakami S, Furukawa S, Ogawa K. Dose reduction trial from 60 Gy in 10 fractions to 54 Gy in 9 fractions schedule in high-dose-rate interstitial brachytherapy for early oral tongue cancer. J Radiat Res 2012;53:722-6.
34. Donath D, Vuong T, Shenouda G, MacDonald B, Tabah R. The potential uses of high-dose-rate brachytherapy in patients with head and neck cancer. Eur Arch Otorhinolaryngol 1995;252:321-4.
35. Inoue T, Inoue T, Yamazaki H, Koizumi M, Kagawa K, Yoshida K, Shiomi H, Imai A, Shimizutani K, Tanaka E, Nose T, Teshima T, Furukawa S, Fuchihata H. High dose rate versus LDR interstitial radiotherapy for carcinoma of the floor of mouth. Int J Radiat Oncol Biol Phys 1998;41:53-8.
36. Matsumoto K, Sasaki T, Shioyama Y, Nakamura K, Atsumi K, Nonoshita T, Ooga S, Yoshitake T, Uehara S, Hirata H, Honda H. Treatment outcome of high-dose-rate interstitial radiation therapy for patients with stage I and II mobile tongue cancer. Jpn J Clin Oncol 2013;43:1012-7.
37. Khalilur R, Hayashi K, Shibuya H. Brachytherapy for tongue cancer in the very elderly is an alternative to external beam radiation. Br J Radiol 2011;84:747-9.
38. Vedasoundaram P, Prasanna AK, Ks R, Selvarajan G, Sinnatamby M, Ramapandian S, Kandasamy S. Role of high dose rate interstitial brachytherapy in early and locally advanced squamous cell carcinoma of buccal mucosa. Springerplus 2014;3:590.
39. Lee SU, Cho KH, Moon SH, Choi SW, Park JY, Yun T, Lee SH, Lim YK, Jeong CY. Clinical outcome of high-dose-rate interstitial brachytherapy in patients with oral cavity cancer. Radiat Oncol J 2014;32:238-46.
40. Tuček L, Petera J, Sirák I, Vošmik M, Doležalová H, Brokešová S, Hodek M, Kašaová L, Paluska P. Hyperfractionated high-dose rate brachytherapy in the treatment of oral tongue cancer. Rep Pract Oncol Radiother 2011;16:243-7.
41. Pernot M, Hoffstetter S, Peiffert D, Aletti P, Lapeyre M, Marchal C, Luporsi E, Bey P, Nancy VL. Role of interstitial brachytherapy in oral and oropharyngeal carcinoma: reflection of a series of 1344 patients treated at the time of initial presentation. Otholaryngol Head Neck Surg 1996;115:519-26.
42. Lefebvre JL, Coche-Dequeant B, Buisset E, Mirabel X, Van JT, Prevost B. Management of early oral cavity cancer. Experience of Centre Oscar Lambret. Eur J Cancer B Oral Oncol 1994;30B:216-20.
43. Mazeron JJ, Simon JM, Le Péchoux C, Crook JM, Grimard L, Piedbois P, Le Bourgeois JP, Pierquin B. Effect of dose rate on local control and complications in definitive irradiation of T1-2 squamous cell carcinomas of mobile tongue and floor of mouth with interstitial iridium-192. Radiother Oncol 1991;21:39-47.
44. Marsiglia H, Haie-Meder C, Sasso G, Mamelle G, Gerbaulet A. Brachytherapy for T1-T2 floor of mouth cancers: the Gustave-Roussy Institute experience. Int J Radiat Oncol Biol Phys 2002;52:1257-63.
45. Dearnaley DP, Dardoufas C, A'Hearn RP, Henk JM. Interstitial irradiation for carcinoma of the tongue and floor of mouth: royal marsden hospital experience 1970-1986. Radiother Oncol 1991;21:183-92.
46. Fujita M, Hirokawa Y, Kashiwado K, Akagi Y, Kashimoto K, Kiriu H, Matsuura K, Ito K. Interstitial brachytherapy for stage I and II squamous cell carcinoma of the oral tongue: factors influencing local control and soft tissue complications. Int J Radiat Oncol Biol Phys 1999;44:767-75.
47. Ihara N, Shibuya H, Yoshimura R, Oota S, Miura M, Watanabe H. Interstitial brachytherapy and neck dissection for Stage III squamous cell carcinoma of the mobile tongue. Acta Oncol 2005;44:709-16.
48. Bourhis J, Overgaard J, Audry H, Ang KK, Saunders M, Bernier J, Horiot JC, Le Maître A, Pajak TF, Poulsen MG, O'Sullivan B, Dobrowsky W, Hliniak A, Skladowski K, Hay JH, Pinto LH, Fallai C, Fu KK, Sylvester R, Pignon JP; Meta-Analysis of Radiotherapy in Carcinomas of Head and neck (MARCH) collaborative group. Hyperfractionated or accelerated radiotherapy in head and neck cancer: a meta-analysis. Lancet 2006;368:843-54.
49. Glenny AM, Furness S, Worthington HV, Conway DI, Oliver R, Clarkson JE, Macluskey M, Pavitt S, Chan KK, Brocklehurst P, CSROC Expert Panel. Interventions for the treatment of oral cavity and oropharyngeal cancer: radiotherapy. Cochrane Database Syst Rev 2010;(12):CD006387.
50. Pignon JP, le Maître A, Maillard E, Bourhis J; MACH-NC Collaborative Group. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. Radiother Oncol 2009;92:4-14.
51. Blanchard P, Baujat B, Holostenco V, Bourredjem A, Baey C, Bourhis J, Pignon JP; MACH-CH Collaborative group. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): a comprehensive analysis by tumour site. Radiother Oncol 2011;100:33-40.
52. Bourhis J, Sire C, Graff P, Grégoire V, Maingon P, Calais G, Gery B, Martin L, Alfonsi M, Desprez P, Pignon T, Bardet E, Rives M, Geoffrois L, Daly-Schveitzer N, Sen S, Tuchais C, Dupuis O, Guerif S, Lapeyre M, Favrel V, Hamoir M, Lusinchi A, Temam S, Pinna A, Tao YG, Blanchard P, Aupérin A. Concomitant chemoradiotherapy versus acceleration of radiotherapy with or without concomitant chemotherapy in locally advanced head and neck carcinoma (GORTEC 99-02): an open-label phase 3 randomised trial. Lancet Oncol 2012;13:145-53.
53. Nguyen-Tan PF, Zhang Q, Ang KK, Weber RS, Rosenthal DI, Soulieres D, Kim H, Silverman C, Raben A, Galloway TJ, Fortin A, Gore E, Westra WH, Chung CH, Jordan RC, Gillison ML, List M, Le QT. Randomized phase III trial to test accelerated versus standard fractionation in combination with concurrent cisplatin for head and neck carcinomas in the Radiation Therapy Oncology Group 0129 trial: long-term report of efficacy and toxicity. J Clin Oncol 2014;32:3858-66.
54. Martinez-Useros J, Garcia-Foncillas J. The challenge of blocking a wider family members of EGFR against head and neck squamous cell carcinomas. Oral Oncol 2015;51:423-30.
55. Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, Sur RK, Raben D, Baselga J, Spencer SA, Zhu J, Youssoufian H, Rowinsky EK, Ang KK. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol 2010;11:21-8.
56. Fletcher GH, Evers WT. Radiotherapeutic management of surgical recurrences and postoperative residuals tumors of the head and neck. Radiology 1970;95:185-8.
57. Marcus RB Jr, Million RR, Cassissi NJ. Postoperative irradiation for squamous cell carcinomas of the head and neck: analysis of time-dose factors related to control above the clavicles. Int J Radiat Oncol Biol Phys 1979;5:1943-49.
58. Lundahl RE, Foote RL, Bonner JA, Suman VJ, Lewis JE, Kasperbauer JL, McCaffrey TV, Olsen KD. Combined neck dissection and postoperative radiation therapy in the management of the high-risk neck: a matched-pair analysis. Int J Radiat Oncol Biol Phys 1998;40:529-34.
59. Lavaf A, Genden EM, Cesaretti JA, Packer S, Kao J. Adjuvant radiotherapy improves overall survival for patients with lymph node-positive head and neck squamous cell carcinoma. Cancer 2008;112:535-43.
60. Kao J, Lavaf F, Teng MS, Huang D, Genden EM. Adjuvant radiotherapy and survival for patients with node-positive head and neck cancer: an analysis by primary site and nodal stage. Int J Radiat Oncol Biol Phys 2008;71:362-70.
61. Hu KS, Quon H, Harrison LB. Management of the neck: radiation therapy and chemotherapy. In: Harrison LB, Sessions RB, Hong WK, editors. Head and Neck Cancer. A Multidisciplinary Approach. 3rd ed. Philadelphia: Lippincot-Williams & Wilkins; 2009. pp. 204-31.
62. Moergel M, Meurer P, Ingel K, Wendt TG, Al-Nawas B. Effectiveness of Postoperative Radiotherapy in Patients with Small Oral and Oropharyngeal Squamous Cell Carcinoma and Concomitant Ipsilateral Singular Cervical Lymph Node Metastasis (pN1). Strahlenther Onkol 2011;187:337-43.
63. Shrime MG, Gullane PJ, Dawson L, Kim J, Gilbert RW, Irish JC, Brown DH, Goldstein DP. The impact of adjuvant radiotherapy on survival in T1-2N1 squamous cell carcinoma of the oral cavity. Arch Otolaryngol Head Neck Surg 2010;136:225-8.
64. Leemans CR, Tiwari T, Nauta JJ, van der Waal L, Snow GB. Regional Lymph node involvement and its signficance in the development of distan metastases in head and neck carcinoma. Cancer 1993;71:452-6.
65. Cooper JS, Pajak TF, Forastiere A, Jacobs J, Fu KK, Ang KK, Laramore GE, Al-Sarraf M. Precisely defining high-risk operable head and neck tumors based on RTOG #83-03 and #88-24: Targets for postoperative radiochemotherapy? Head Neck 1998;20:588-94.
66. Langendijk JA, Slotman BJ, van der Waal I, Doornaert P, Berkof J, Leemans CR. Risk group definition by recursive partitioning analysis of patients with squamous cell head and neck carcinoma treated with surgery and postoperative radiotherapy. Cancer 2005;104:1408-17.
67. Jonkman A, Kaanders JH, Terhaard CH, Hoebers FJ, van den Ende PL, Wijers OB, Verhoef LC, de Jong MA, Leemans CR, Langendijk JA. Multicenter validation of recursive partitioning analysis classification for patients with squamous cell head and neck carcinoma treated with surgery and postperative radiotherapy. Int J Radiat Oncol Biol Phys 2007;68:119-25.
68. Expert Panel on Radiation Oncology-Head and Neck, Salama JK, Saba N, Quon H, Garg MK, Lawson J, McDonald MW, Ridge JA, Smith RV, Yeung AR, Yom SS, Beitler JJ. ACR appropriateness criteria® adjuvant therapy for resected squamous cell carcinoma of the head and neck. Oral Oncol 2011;47:554-9.
69. Bur AM, Lin A, Weinstein GS. Adjuvant radiotherapy for early head and neck squamous cell carcinoma with perineural invasion: a systematic review. Head Neck 2016;38 Suppl 1:E2350-7.
70. Huang J, Barbera L, Brouwers M, Browman G, Mackillop WJ. Does delay instarting treatment affect the outcomes of radiotherapy? a systematic review. J Clin Oncol 2003;21:555-63.
71. Chen Z, King W, Pearcey R, Kerba M, Mackillop WJ. The relationship between waiting time for radiotherapy and clinical outcomes: a systematic review of the literature. Radiother Oncol 2008;87:3-16.
72. van Harten MC, Hoebers FJ, Kross KW, van Werkhoven ED, van den Brekel MW, van Dijk BA. Determinants of treatment waiting times for head and neck cancer in the Netherlands and their relation to survival. Oral Oncol 2015;51:272-8.
73. Peters LJ, Withers HR. Applying radiobiological principles to combined modality treatment of head and neck cancer - the time factor. Int J RadiatOncol Biol Phys 1997;39:831-6.
74. González Ferreira JA, Jaén Olasolo J, Azinovic I, Jeremic B. Effect of radiotherapy delay in overall treatment time on local control andsurvival in head and neck cancer: review of the literature. Rep Pract Oncol Radiother 2015;20:328-39.
75. Ang KK, Trotti A, Brown BW, Garden AS, Foote RL, Morrison WH, Geara FB, Klotch DW, Goepfert H, Peters LJ. Randomized trial addressing risk features and time factors of surgery plus radiotherapy in advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys 2001;51:571-8.
76. Langendijk JA, de Jong MA, Leemans CR, de Bree R, Smeele LE, Doornaert P, Slotman BJ. Postoperative radiotherapy in squamous cell carcinoma of the oral cavity: the importance of the overall treatment time. Int J Radiat Oncol Biol Phys 2003;57:693-700.
77. Peters LJ, Goepfert H, Ang KK, Byers RM, Maor MH, Guillamondegui O, Morrison WH, Weber RS, Garden AS, Frankenthaler RA, Oswald MJ, Brown BW. Evaluation of the dose for postoperative radioation therapy of head and neck cancer: first report of a prospective randomized trial. Int J Radiat Oncol Biol Phys 1993;26:3-11.
78. Winquist E, Oliver T, Gilbert R. Postoperative chemoradiotherapy for advanced squamous cell carcinoma of the head and neck: a systematic review with meta-analysis. Head Neck 2007;29:38-46.
79. Bernier J, Cooper JS, Pajak TF, van Glabbeke M, Bourhis J, Forastiere A, Ozsahin EM, Jacobs JR, Jassem J, Ang KK, Lefèbvre JL. Defining risk levels in locally advanced head and neck cancers: a comparative analysis of concurrent postoperative radiation plus chemotherapy trials of EORTC (#22931) and RTOG (#9501). Head Neck 2005;27:843-50.
80. Cooper JS, Pajak TF, Forastiere AA, Jacobs J, Campbell BH, Saxman SB, Kish JA, Kim HE, Cmelak AJ, Rotman M, Machtay M, Ensley JF, Chao KS, Schultz CJ, Lee N, Fu KK; Radiation Therapy Oncology Group 9501/Intergroup. PostpeRative concurrent radiotherapy and chemotherapy for high risk squamous cell carcinoma of the head and neck. N Engl J Med 2004;350:1937-44.
81. Bernier J, Domenge C, Ozsahin M, Matuszewska K, Lefèbvre JL, Greiner RH, Giralt J, Maingon P, Rolland F, Bolla M, Cognetti F, Bourhis J, Kirkpatrick A, van Glabbeke M, European Organization for Research and Treatment of Cancer Trial 22931. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N Engl J Med 2004;350:1945-52.
82. Cooper JS, Zhang Q, Pajak TF, Forastiere AA, Jacobs J, Saxman SB, Kish JA, KimHE, Cmelak AJ, Rotman M, Lustig R, Ensley JF, Thorstad W, Schultz CJ, Yom SS, Ang KK. Long-term follow-up of the RTOG 9501/Intergroup Phase III trial: postoperative concurrent radiation therapy and chemotherapy in high-risk squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys 2012;84:1198-205.
83. Eisbruch A, Gregoire V. Balancing risk and reward in target delineation for highly conformal radiotherapy in head and neck cancer. Semin Radiat Oncol 2009;19:43-52.
84. Chen AM, Farwell DG, Luu Q, Chen LM, Vijayakumar S, Purdy JA. Marginal misses after postoperative intensity-modulated radiotherapy for head and neck cancer. Int J Radiat Oncol Biol Phys 2011;80:1423-9.
85. Grégoire V, Eisbruch A, Hamoir M, Levendag P. Proposal for the delineation of the nodal CTV in the node-positive and the post-operative neck. Radiother Oncol 2006;79:15-20.
86. Liu SH, Chao KS, Leu YS, Lee JC, Liu CJ, Huang YC, Chang YF, Chen HW, Tsai JT, Chen YJ. Guideline and preliminary clinical practice results for dose specification and target delineation for postoperative radiotherapy for oral cavity cancer. Head Neck 2015;37:933-9.
87. Merlotti A, Alterio D, Vigna-Taglianti R, Muraglia A, Lastrucci L, Manzo R, Gambaro G, Caspiani O, Miccichè F, Deodato F, Pergolizzi S, Franco P, Corvò R, Russi EG, Sanguineti G; Italian Association of Radiation Oncology. Technical guidelines for head and neck cancer IMRT on behalf of the Italian association of radiation oncology - head and neck working group. Radiat Oncol 2014;9:264.
88. Ko HC, Gupta V, Mourad WF, Hu KS, Harrison LB, Som PM, Bakst RL. A contouring guide for head and neck cancers with perineural invasion. Pract Radiat Oncol 2014;4:e247-58.
89. Goineau A, Piot B, Malard O, Ferron C, Lisbona A, Cassagnau E, Delamazure AS, Campion L, Bardet E. Postoperative interstitial brachytherapy for resectable squamous cell carcinoma of the tongue. Brachytherapy 2015;14:71-6.
90. Petera J, Sirák I, Laco J, Kašaová L, Tuček L, Doležalová H. High-dose-rate brachytherapy in early oral cancer with close or positive margins. Brachytherapy 2015;14:77-83.
91. Lapeyre M, Bollet MA, Racadot S, Geoffrois L, Kaminsky MC, Hoffstetter S, Dolivet G, Toussaint B, Luporsi E, Peiffert D. Postoperative brachytherapy alone and combined postoperative radiotherapy and brachytherapy boost for squamous cell carcinoma of the oral cavity, with positive or close margins. Head Neck 2004;26:216-23.
92. Pernot M, Aletti P, Carolus JM, Marquis I, Hoffstetter S, Maaloul F, Peiffert D, Lapeyre M, Luporsi E, Marchal C, Noël A, Bey P. Indications, techniques and results of postoperative brachytherapy in cancer of the oral cavity. Radiother Oncol 1995;35:186-92.
93. Fietkau R, Grabenbauer GG, Iro H, Müller RG, Farmand M, Atendorf-Hofmann A, Sauer R. Interstitial and percutaneous radiotherapy after limited surgery in carcinoma of the oral cavity. Strahlenther Onkol 1991;167:591-8.
94. Chan AK, Huang SH, Le LW, Yu E, Dawson LA, Kim JJ, Cho BC, Bayley AJ, Ringash J, Goldstein D, Chan K, Waldron J, O'Sullivan B, Cummings B, Hope AJ. Postoperative intensity-modulated radiotherapy following surgery for oral cavity squamous cell carcinoma: patterns of failure. Oral Oncol 2013;49:255-60.
95. Hoffmann M, Saleh-Ebrahimi L, Zwicker F, Haering P, Schwahofer A, Debus J, Huber PE, Roeder F. Long term results of postoperative Intensity-Modulated Radiation Therapy (IMRT) in the treatment of Squamous Cell Carcinoma (SCC) located in the oropharynx or oral cavity. Radiat Oncol 2015;10:251.
96. Sher DJ, Thotakura V, Balboni TA, Norris CM Jr, Haddad RI, Posner MR, Lorch J, Goguen LA, Annino DJ, Tishler RB. Treatment of oral cavity squamous cell carcinoma with adjuvant or definitive intensity-modulated radiation therapy. Int J Radiat Oncol Biol Phys 2011;81:e215-22.
97. Gomez DR, Zhung JE, Gomez J, Chan K, Wu AJ, Wolden SL, Pfister DG, Shaha A, Shah JP, Kraus DH, Wong RJ, Lee NY. Intensity-modulated radiotherapy in postoperative treatment of oral cavity cancers. Int J Radiat Oncol Biol Phys 2009;73:1096-103.
98. Chakraborty S, Patil VM, Babu S, Muttath G, Thiagarajan SK. Locoregional recurrences after post-operative volumetric modulated arc radiotherapy (VMAT) in oral cavity cancers in a resource constrained setting: experience and lessons learned. Br J Radiol 2015;88:20140795.
99. Studer G, Brown M, Bredell M, Graetz KW, Huber G, Linsenmeier C, Najafi Y, N Riesterer O, Rordorf T, Schmid S, Glanzmann C. Follow up after IMRT in oral cavity cancer: update. Radiat Oncol 2012;7:84.
100. Collan J, Lundberg M, Vaalavirta L, Bäck L, Kajanti M, Mäkitie A, Tenhunen M, Saarilahti K. Patterns of relapse following surgery and postoperative intensity modulated radiotherapy for oral and oropharyngeal cancer. Acta Oncol 2011;50:1119-25.
101. Geretschläger A, Bojaxhiu B, Crowe S, Arnold A, Manser P, Hallermann W, Aebersold DM, Ghadjar P. Outcome and patterns of failure after postoperative intensity modulated radiotherapy for locally advanced or high-risk oral cavity squamous cell carcinoma. Radiat Oncol 2012;7:175.
102. Yao M, Chang K, Funk GF, Lu H, Tan H, Wacha J, Dornfeld KJ, Buatti JM. The failure patterns of oral cavity squamous cell carcinoma after intensity-modulated radiotherapy-the university of iowa experience. Int J Radiat Oncol Biol Phys 2007;67:1332-41.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Cabrera-Rodríguez JJ. The role of radiotherapy in the treatment of oral cavity cancer. Plast Aesthet Res 2016;3:158-66. http://dx.doi.org/10.20517/2347-9264.2016.13
AMA Style
Cabrera-Rodríguez JJ. The role of radiotherapy in the treatment of oral cavity cancer. Plastic and Aesthetic Research. 2016; 3: 158-66. http://dx.doi.org/10.20517/2347-9264.2016.13
Chicago/Turabian Style
Cabrera-Rodríguez, Joaquín J.. 2016. "The role of radiotherapy in the treatment of oral cavity cancer" Plastic and Aesthetic Research. 3: 158-66. http://dx.doi.org/10.20517/2347-9264.2016.13
ACS Style
Cabrera-Rodríguez, JJ. The role of radiotherapy in the treatment of oral cavity cancer. Plast. Aesthet. Res. 2016, 3, 158-66. http://dx.doi.org/10.20517/2347-9264.2016.13
About This Article
Special Issue
Copyright
Author Biographies

Data & Comments
Data
 Cite This Article 37 clicks
Cite This Article 37 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.